Palytoxin
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.162.538 |
| Chemical and physical data | |
| Formula | C129H223N3O54 |
| Molar mass | 2680.13 g.mol-1 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
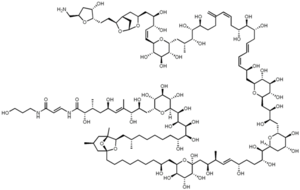
Palytoxin is a complex marine natural product containing 71 asymmetric centers. Palytoxin, isolated from soft coral, is considered to be one of the most toxic non-peptide substances known, second only to maitotoxin in terms of toxicity towards mice. Palytoxin was originally isolated in 1971 in Hawaii from the seaweed-like coral, "Limu make o hana (Seaweed of Death from Hana)"[1]. Later, in 1982 its full chemical structure was published by Prof. Daisuke Uemura and coworkers at Nagoya University[2][3][4]. Professor Yoshito Kishi's group at Harvard University first synthesized palytoxin in 1994[5][6]. This feat is still considered today by many to be the greatest synthetic accomplishment ever, due to its complexity in structure.
Palytoxin targets the sodium-potassium pump protein by binding to the molecule such that the molecule is locked in a position where it allows passive transport of both the sodium and potassium ions, thereby destroying the ion gradient that is essential for most cells.
Typical symptoms of palytoxin poisoning are angina-like chest pains, asthma-like breathing difficulties, tachycardia, unstable blood pressure, hemolysis (destruction of red blood cells), and an electrocardiogram showing an exaggerated T wave. The onset of symptoms is rapid, and death usually follows just minutes after.
Animal studies have shown that vasodilators, such as papaverine and isosorbide dinitrate, can be used as antidotes. The animal experiments only showed benefit if the antidotes were injected into the heart immediately following exposure.[7] Treatment in humans is symptomatic and supportive.
Footnotes
- ^ Clayden, J., Greeves, N. (2000), pages 19-21
- ^ Chemical Society of Japan, et al. (2005). "CSJ Award-2005 Prof. Daisuke Uemura" Retrieved on 24 July, 2007 from http://www.chemistry.or.jp/csj-en/membership/awards/achieve/2005-uemura.html Chemical Soc. of Japan, Prof. D. Uemura
- ^ Chemical Society of Japan, et al. (2005), -- "Its structural determination presented many difficulties. Dr. Uemura elucidated its planar structure in 1981 by repeatedly carrying out site-specific oxidative degradation and determined the structure of the degraded products using a sample that was originally isolated from Palythoa tuberculosa of Okinawa[n] origin."
- ^ Stereochemistry of Palytoxin. 4. Complete Structure, J. K. Cha, W. J. Christ, J. M. Finan, H. Fujioka, Y. Kishi, L. L. Klein, S. S. Ko, J. Leder, W. W. McWhorter, Jr., K. -P. Pfaff, M. Yonaga, D. Uemura, and Y. Hirata, J. Am. Chem. Soc., 104, 7369-7371 (1982)
- ^ "Total Synthesis of Palytoxin Carboxylic Acid and Palytoxin Amide," R.W. Armstrong, J.-M. Beau, S.H. Cheon, W.J. Christ, H. Fujioka, W.-H. Ham, L.D. Hawkins, H. Jin, S.H. Kang, Y. Kishi, M.J. Martinelli, W.W. McWhorter, Jr., M. Mizuno, M. Nakata, A.E. Stutz, F.X. Talamas, M. Taniguchi, J.A. Tino, K. Ueda, J. Uenishi, J.B. White, and M. Yonaga, J. Am. Chem. Soc., 111, 7530 (1989)
- ^ "Synthesis of Palytoxin from Palytoxin Carboxylic Acid", E.M. Suh and Y. Kishi, J. Am. Chem. Soc., 116, 11205 (1994).
- ^ Wiles J, Vick J, Christensen M (1974). "Toxicological evaluation of palytoxin in several animal species". Toxicon. 12 (4): 427–33. doi:10.1016/0041-0101(74)90011-7. PMID 4155146.
{{cite journal}}: CS1 maint: multiple names: authors list (link)
References
- Nelson, David L. and Cox, Michael M.. "Lehninger Principles of Biochemistry, 4th Edition". Published by Freeman in 2005. p.399 mentions palytoxin.
- Clayden, R., Greeves, N. (2000) "Organic chemistry" Oxford publishing
- CBWInfo
