Bacillus mycoides
This article has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these messages)
|
| Bacillus mycoides | |
|---|---|
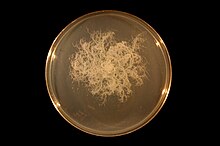
| |
| Bacillus mycoides growing clockwise on an agar plate. | |
| Scientific classification | |
| Kingdom: | |
| Division: | |
| Class: | |
| Order: | |
| Family: | |
| Genus: | |
| Species: | mycoides
|
| Binomial name | |
| Bacillus mycoides | |
Bacillus mycoides is a bacterium of the genus Bacillus.
Structure and compounds
Bacillus mycoides cells are usually larger than 3 micrometers. It forms chains of cells. It can form acid from glucose, this form is non motile. It's cell body does not swell when sporulating; Using Voges–Proskauer test, B. mycoides produces a positive result. It can also hydrolyse starch. B. mycoides is a common soil organism.[1] When grown on solid media B. mycoides forms spreading colonies with a repeating spiral pattern. The direction of curvature of the pattern in a given strain is known as its chirality and is a hereditary trait <ref>Di Franco, C., Beccari, E., Santini, T., Pisaneschi, G., & Tecce, G. 2002. Colony shape as a genetic trait in the pattern-forming Bacillus mycoides. BMC Microbiol. 2, 33.<ref>. B. mycoides has the unusual property of being able to respond to mechanical force and surface structure variations in the media on which it is growing <ref>Stratford JP, Woodley MA, Park S (2013) Variation in the Morphology of Bacillus mycoides Due to Applied Force and Substrate Structure. PLoS ONE 8(12): e81549. doi:10.1371/journal.pone.0081549.<ref>
Uses
Bacillus mycoides are found in common pesticides and are used to inhibit the growth of harmful bacteria and fungi. There seems to be no negative effects on humans or the environment.Citation needed
Nitrogen Cycle
Bacillus mycides is an ammonifying bacterium, and can convert peptone (protein, an organic nitrogen source) into ammonia. In water, ammonia becomes ammonium. This product is essential for nitrifying bacteria such as Nitrosococcus, which use the ammonia under aerobic conditions to make nitrite.
References
