From Wikipedia, the free encyclopedia
Jump to navigation Jump to search
Infobox drug/sandbox (edit · t · history · diff · links · /test · Source · e · t · hist · links · /subpages · /doc · /doc edit )
/testcases2 -- titles, licence, EMA/testcases3 -- pregcat, legal, licence, PLLR, ATC; Wikidata/testcases4 -- chem formula, mab/testcases5 -- identifiers, second id's/testcases6 -- all up/testcases7images -- images/testcases8 -- type, titles/testcases9 -- order variants, container_only/testcases10 -- pharmacokinetic, localINN (2017) has (data page) -- is a redirect/testcases11 -- hormone, gene therapy (2018) , has (data page)/testcases-FDA -- FDA 2023/testcases-warning -- warning box(es)
Side by side comparison {{Infobox drug }} {{Infobox drug/sandbox }}
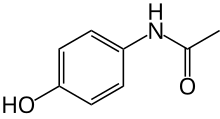
Paracetamol Pronunciation Paracetamol: Trade names Tylenol , Panadol , others [1] Other names N -acetyl-para -aminophenol (APAP), acetaminophen (USAN US AHFS /Drugs.com Monograph MedlinePlus a681004 License data
Pregnancy Routes of Oral (by mouth), rectal , intravenous (IV)Drug class Analgesics and antipyretics ATC code Legal status Bioavailability 63–89%[4] : 73 Protein binding negligible to 10–25% in overdose[5] Metabolism Predominantly in the liver[6] Metabolites APAP gluc , APAP sulfate , APAP GSH , APAP cys , AM404, NAPQI [7] Onset of action Pain relief onset by route :oral – 37 minutes[8] Intravenous – 8 minutes[8] Elimination half-life 1.9–2.5 hours[5] Excretion Urine[5]
N -(4-hydroxyphenyl)acetamide
CAS Number PubChem CID PubChem SID IUPHAR/BPS DrugBank ChemSpider UNII KEGG ChEBI ChEMBL PDB ligand Formula C 8 H 9 N O 2 Molar mass −1 3D model (JSmol ) Density 1.263 g/cm3 Melting point 169 °C (336 °F) [9] [10] Solubility in water 7.21 g/kg (0 °C)[11] 8.21 g/kg (5 °C)[11] 9.44 g/kg (10 °C)[11] 10.97 g/kg (15 °C)[11] 12.78 g/kg (20 °C)[11] ~14 mg/ml (20 °C)
InChI=1S/C8H9NO2/c1-6(10)9-7-2-4-8(11)5-3-7/h2-5,11H,1H3,(H,9,10)
Y Key:RZVAJINKPMORJF-UHFFFAOYSA-N
Y (verify)
Paracetamol Pronunciation Paracetamol: Trade names Tylenol , Panadol , others [1] Other names N -acetyl-para -aminophenol (APAP), acetaminophen (USAN US AHFS /Drugs.com Monograph MedlinePlus a681004 License data Pregnancy Routes of Oral (by mouth), rectal , intravenous (IV)Drug class Analgesics and antipyretics ATC code Legal status Bioavailability 63–89%[4] : 73 Protein binding negligible to 10–25% in overdose[5] Metabolism Predominantly in the liver[6] Metabolites APAP gluc , APAP sulfate , APAP GSH , APAP cys , AM404, NAPQI [7] Onset of action Pain relief onset by route :oral – 37 minutes[8] Intravenous – 8 minutes[8] Elimination half-life 1.9–2.5 hours[5] Excretion Urine[5]
N -(4-hydroxyphenyl)acetamide
CAS Number PubChem CID PubChem SID IUPHAR/BPS DrugBank ChemSpider UNII KEGG ChEBI ChEMBL PDB ligand Formula C 8 H 9 N O 2 Molar mass −1 3D model (JSmol ) Density 1.263 g/cm3 Melting point 169 °C (336 °F) [9] [10] Solubility in water 7.21 g/kg (0 °C)[11] 8.21 g/kg (5 °C)[11] 9.44 g/kg (10 °C)[11] 10.97 g/kg (15 °C)[11] 12.78 g/kg (20 °C)[11] ~14 mg/ml (20 °C)
InChI=1S/C8H9NO2/c1-6(10)9-7-2-4-8(11)5-3-7/h2-5,11H,1H3,(H,9,10)
Y Key:RZVAJINKPMORJF-UHFFFAOYSA-N
Y (verify)
References
^ International Drug Names ^ "Acetaminophen Use During Pregnancy" . Drugs.com . 14 June 2019. Archived from the original on 9 March 2020. Retrieved 25 February 2020 .^ "Regulatory Decision Summary – Acetaminophen Injection" . Health Canada Archived from the original on 7 June 2022. Retrieved 7 June 2022 .^ Working Group of the Australian and New Zealand College of Anaesthetists and Faculty of Pain Medicine (2015). Schug SA, Palmer GM, Scott DA, Halliwell R, Trinca J (eds.). Acute Pain Management: Scientific Evidence ISBN 978-0-9873236-7-5 the original (PDF) on 31 July 2019. Retrieved 28 October 2019 . ^ a b c Forrest JA, Clements JA, Prescott LF (1982). "Clinical pharmacokinetics of paracetamol". Clin Pharmacokinet . 7 (2): 93–107. doi :10.2165/00003088-198207020-00001 . PMID 7039926 . S2CID 20946160 . ^ "Codapane Forte Paracetamol and codeine phosphate product information" (PDF) . TGA eBusiness Services . Alphapharm Pty Limited. 29 April 2013. Archived from the original on 6 February 2016. Retrieved 10 May 2014 .^ "Acetaminophen Pathway (therapeutic doses), Pharmacokinetics" . Archived from the original on 4 March 2016. Retrieved 13 January 2016 .^ a b Pickering G, Macian N, Libert F, Cardot JM, Coissard S, Perovitch P, Maury M, Dubray C (September 2014). "Buccal acetaminophen provides fast analgesia: two randomized clinical trials in healthy volunteers" . Drug Design, Development and Therapy . 8 : 1621–1627. doi :10.2147/DDDT.S63476 PMC 4189711 PMID 25302017 . In postoperative conditions for acute pain of mild to moderate intensity, the quickest reported time to onset of analgesia with APAP is 8 minutes9 for the iv route and 37 minutes6 for the oral route. ^ Karthikeyan M, Glen RC, Bender A (2005). "General Melting Point Prediction Based on a Diverse Compound Data Set and Artificial Neural Networks". Journal of Chemical Information and Modeling . 45 (3): 581–590. doi :10.1021/ci0500132 . PMID 15921448 . ^ "melting point data for paracetamol" . Lxsrv7.oru.edu. Archived from the original on 30 June 2012. Retrieved 19 March 2011 .^ a b c d e Granberg RA, Rasmuson AC (1999). "Solubility of paracetamol in pure solvents". Journal of Chemical & Engineering Data 44 (6): 1391–95. doi :10.1021/je990124v .
Side by side comparison {{Infobox drug }} {{Infobox drug/sandbox }}
Brincidofovir Trade names Tembexa Other names CMX001; Cidofovir-HDP; hexadecyloxypropyl-cidofovir License data Routes of By mouth ATC code Legal status
({[(2S)-1-(4-amino-2-oxo-1,2-dihydropyrimidin-1-yl)-3-hydroxypropan-2-yl]oxy}methyl)[3-(hexadecyloxy)propoxy]phosphinic acid
CAS Number PubChem CID DrugBank ChemSpider UNII KEGG ChEMBL Formula C 27 H 52 N 3 O 7 P Molar mass −1 3D model (JSmol )
CCCCCCCCCCCCCCCCOCCCOP(=O)(O)CO[C@H](CO)Cn1ccc(N)nc1=O
InChI=1S/C27H52N3O7P/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-19-35-20-16-21-37-38(33,34)24-36-25(23-31)22-30-18-17-26(28)29-27(30)32/h17-18,25,31H,2-16,19-24H2,1H3,(H,33,34)(H2,28,29,32)/t25-/m0/s1
Key:WXJFKKQWPMNTIM-VWLOTQADSA-N
Brincidofovir Trade names Tembexa Other names CMX001; Cidofovir-HDP; hexadecyloxypropyl-cidofovir License data Routes of By mouth ATC code Legal status
({[(2S)-1-(4-amino-2-oxo-1,2-dihydropyrimidin-1-yl)-3-hydroxypropan-2-yl]oxy}methyl)[3-(hexadecyloxy)propoxy]phosphinic acid
CAS Number PubChem CID DrugBank ChemSpider UNII KEGG ChEMBL Formula C 27 H 52 N 3 O 7 P Molar mass −1 3D model (JSmol )
CCCCCCCCCCCCCCCCOCCCOP(=O)(O)CO[C@H](CO)Cn1ccc(N)nc1=O
InChI=1S/C27H52N3O7P/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-19-35-20-16-21-37-38(33,34)24-36-25(23-31)22-30-18-17-26(28)29-27(30)32/h17-18,25,31H,2-16,19-24H2,1H3,(H,33,34)(H2,28,29,32)/t25-/m0/s1
Key:WXJFKKQWPMNTIM-VWLOTQADSA-N
Side by side comparison {{Infobox drug }} {{Infobox drug/sandbox }}
Albendazole Trade names Albenza, Valbazen, Zentel, others AHFS /Drugs.com Monograph MedlinePlus a610019 License data Pregnancy Routes of By mouth ATC code Legal status
AU :S4 (Prescription only)CA ℞-only UK :POM (Prescription only)US :℞-only (for humans; veterinary suspension and paste are both OTC)[1]
Bioavailability <5%[2] Protein binding 70%[2] Metabolism Hepatic [2] Elimination half-life 8-12 hours[2] Excretion Bile (humans)
Methyl [5-(propylthio)-1H -benzoimidazol-2-yl]carbamate
CAS Number PubChem CID DrugBank ChemSpider UNII KEGG ChEBI ChEMBL NIAID ChemDB Formula C 12 H 15 N 3 O 2 S Molar mass −1 3D model (JSmol ) Melting point 208 to 210 °C (406 to 410 °F)
CCCSc2ccc1nc(NC(=O)OC)[nH]c1c2
InChI=1S/C12H15N3O2S/c1-3-6-18-8-4-5-9-10(7-8)14-11(13-9)15-12(16)17-2/h4-5,7H,3,6H2,1-2H3,(H2,13,14,15,16)
Y Key:HXHWSAZORRCQMX-UHFFFAOYSA-N
Y (verify)
Albendazole Trade names Albenza, Valbazen, Zentel, others AHFS /Drugs.com Monograph MedlinePlus a610019 License data Pregnancy Routes of By mouth ATC code Legal status
AU :S4 (Prescription only)CA ℞-only UK :POM (Prescription only)US :℞-only (for humans; veterinary suspension and paste are both OTC)[1]
Bioavailability <5%[2] Protein binding 70%[2] Metabolism Hepatic [2] Elimination half-life 8-12 hours[2] Excretion Bile (humans)
Methyl [5-(propylthio)-1H -benzoimidazol-2-yl]carbamate
CAS Number PubChem CID DrugBank ChemSpider UNII KEGG ChEBI ChEMBL NIAID ChemDB Formula C 12 H 15 N 3 O 2 S Molar mass −1 3D model (JSmol ) Melting point 208 to 210 °C (406 to 410 °F)
CCCSc2ccc1nc(NC(=O)OC)[nH]c1c2
InChI=1S/C12H15N3O2S/c1-3-6-18-8-4-5-9-10(7-8)14-11(13-9)15-12(16)17-2/h4-5,7H,3,6H2,1-2H3,(H2,13,14,15,16)
Y Key:HXHWSAZORRCQMX-UHFFFAOYSA-N
Y (verify)
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.