18-Crown-6

| |
| |
| Names | |
|---|---|
| IUPAC name
1,4,7,10,13,16-hexaoxacyclooctadecane
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.037.687 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H24O6 | |
| Molar mass | 264.315 g/mol |
| Density | 1.237 g/cm3 |
| Melting point | 37 to 40 °C (99 to 104 °F; 310 to 313 K) |
| Boiling point | 116 °C (241 °F; 389 K) (0.2 Torr) |
| Related compounds | |
Related compounds
|
Dibenzo-18-crown-6 Triglyme |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
18-Crown-6 is an organic compound with the formula [C2H4O]6 and the IUPAC name of 1,4,7,10,13,16-hexaoxacyclooctadecane. It is a white, hygroscopic crystalline solid with a low melting point.[1] Like other crown ethers, 18-crown-6 functions as a ligand for some metal cations with a particular affinity for potassium cations (binding constant in methanol: 106 M−1). The point group of 18-crown-6 is S6. The dipole moment of 18-crown-6 varies in different solvent and under different temperature. Under 25 °C, the dipole moment of 18-crown-6 is 2.76 ± 0.06 D in cyclohexane and 2.73 ± 0.02 in benzene.[2] The synthesis of the crown ethers led to the awarding of the Nobel Prize in Chemistry to Charles J. Pedersen.
Synthesis
This compound is prepared by a modified Williamson ether synthesis in the presence of a templating cation:[3] It can be also prepared by the oligomerization of ethylene oxide:[1]
- (CH2OCH2CH2Cl)2 + (CH2OCH2CH2OH)2 + 2 KOH → (CH2CH2O)6 + 2 KCl + 2 H2O
It can be purified by distillation, where its tendency to supercool becomes evident. 18-Crown-6 can also be purified by recrystallisation from hot acetonitrile. It initially forms an insoluble solvate.[3] Rigorously dry material can be made by dissolving the compound in THF followed by the addition of NaK to give [K(18-crown-6)]Na, an alkalide salt. Crystallographic analysis reveals a relatively flat molecule but one where the oxygen centres are not oriented in the idealized 6-fold symmetric geometry usually shown.[4] The molecule undergoes significant conformational change upon complexation.
Reactions
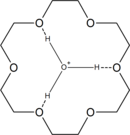
18-Crown-6 has a high affinity for the hydronium ion H3O+, as it can fit inside the crown ether. Thus, reaction of 18-crown-6 with strong acids gives the cation [H3O⊂18-crown-6]+. For example, interaction of 18-crown-6 with HCl gas in toluene with a little moisture gives an ionic liquid layer with the composition [H3O⊂18-crown-6]+[HCl2]−•3.8 C6H5Me, from which the solid [H3O⊂18-crown-6]+[HCl2]− can be isolated on standing. Reaction of the ionic liquid layer with two molar equivalents of water gives the crystalline product (H5O2)[H3O⊂18-crown-6]Cl2.[1]
Applications
18-Crown-6 binds to a variety of small cations, using all 6 oxygens as donor atoms. Crown ethers can be used in the laboratory as phase transfer catalysts.[5] For example, potassium permanganate dissolves in benzene In the presence of 18-crown-6, giving the so-called "purple benzene", which can be used to oxidize diverse organic compounds.[1]
Various substitution reactions are also accelerated in the presence of 18-crown-6, which suppresses ion-pairing. The anions thereby become naked nucleophiles. For example, using 18-crown-6, potassium acetate is a more powerful nucleophile in organic solvents:[1]
- [K(18-crown-6)+]OAc− + C6H5CH2Cl → C6H5CH2OAc + [K(18-crown-6)+]Cl−
The first electride salt using 18-crown-6 that has been synthesized and examined with X-ray crystallography is [Cs(18-crown-6)2]+•e− in 1983. This highly air- and moisture-sensitive solid has a sandwich molecular structure, where the electron is trapped within nearly spherical lattice cavities. However, the shortest electron-electron distance is too long (8.68 Å) to make this material a conductor of electricity.[1]
References
- ^ a b c d e f Jonathan W. Steed; Jerry L. Atwood (2009). Supramolecular Chemistry, 2nd edition. Wiley. ISBN 978-0-470-51233-3.
- ^ Caswell, Lyman R.; Savannunt, Diana S. (January 1988). "Temperature and solvent effects on the experimental dipole moments of three crown ethers". J Heterocyclic Chem. 25 (1): 73–79. doi:10.1002/jhet.5570250111.
- ^ a b George W. Gokel, Donald J. Cram, Charles L. Liotta, Henry P. Harris, and Fred L. Cook (1988). "18-Crown-6". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 6, p. 301. - ^ J. D. Dunitz, P. Seiler "1,4,7,10,13,16-Hexaoxacyclooctadecane" Acta Crystallogr. (1974). B30, 2739. doi:10.1107/S0567740874007928.
- ^ Liotta, C. L.; Berknerin, J. "18-Crown-6" in Encyclopedia of Reagents for Organic Synthesis (Ed: L. Paquette) 2004, J. Wiley & Sons, New York. doi:10.1002/047084289X.rc261

