2'-Fucosyllactose

| |
| Names | |
|---|---|
| IUPAC name
α-L-Fucopyranosyl-(1→2)-β-D-galactopyranosyl-(1→4)-D-glucose
| |
| Systematic IUPAC name
(2R,3R,4R,5R)-4-{[(2S,3R,4S,5R,6R)-4,5-Dihydroxy-6-(hydroxymethyl)-3-{[(2S,3S,4R,5S,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxy}oxan-2-yl]oxy}-2,3,5,6-tetrahydroxyhexanal | |
| Other names
2′-FL
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C18H32O15 | |
| Molar mass | 488.439 g·mol−1 |
| Density | 1.681 g/cm3 |
| 240.0 g/L (in water) | |
| Acidity (pKa) | 11.9 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
2′-Fucosyllactose (2′-FL) is a fucosylated neutral trisaccharide composed of L-fucose, D-galactose, and D-glucose units. It is the most prevalent human milk oligosaccharide (HMO) naturally present in human breast milk, making up about 30% of all of HMOs.[1] It was first discovered in the 1950s in human milk. The oligosaccharide's primary isolation technique has been in use since 1972.[2]
Structure
[edit]2′-FL consists of an L-fucose monomer in the α stereochemical configuration linked at the first carbon to a monomer of D-galactose in the β stereochemical configuration at the second carbon, which is in turn linked at the first carbon to a monomer of D-glucose (which may be in either the α or β configuration) at the fourth carbon.
Production
[edit]The compound may be biosynthesized in quantity using E. coli.[3][4][5][6]
Metabolism
[edit]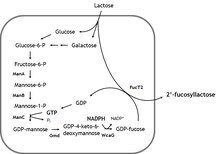
This section needs expansion. You can help by adding to it. (November 2012) |
Uses
[edit]As with many other oligosaccharides, a characteristic of 2′-FL is its ability to protect against infectious diseases[7] by preventing epithelial-level adhesion of toxins and pathogens.[8] 2′-FL stimulates the growth of certain bifidobacteria and upregulation of receptors[citation needed] which collectively lend to toxic and pathogenic protection;[citation needed] this is most prevalent in infants. Among the pathogens that 2′-FL is known to protect against are Campylobacter jejuni, Salmonella enterica (serotype Typhimurium), and Helicobacter pylori.[8]
References
[edit]- ^ Vandenplas, Yvan; Berger, Bernard; Carnielli, Virgilio Paolo; Ksiazyk, Janusz; Lagström, Hanna; Sanchez Luna, Manuel; Migacheva, Natalia; Mosselmans, Jean-Marc; Picaud, Jean-Charles; Possner, Mike; Singhal, Atul; Wabitsch, Martin (2018-08-24). "Human Milk Oligosaccharides: 2′-Fucosyllactose (2′-FL) and Lacto-N-Neotetraose (LNnT) in Infant Formula". Nutrients. 10 (9): 1161. doi:10.3390/nu10091161. ISSN 2072-6643. PMC 6164445. PMID 30149573.
- ^ "Showing metabocard for 2-Fucosyllactose (HMDB02098)". Human Metabolome Database. Retrieved 19 October 2012.
- ^ "U of I microbial engineer synthesizes 2-fucosyllactose to protect babies from pathogens". News Medical. Retrieved 19 October 2012.
- ^ Chin, Young-Wook; Kim, Ji-Yeong; Lee, Won-Heong; Seo, Jin-Ho (2015). "Enhanced production of 2′-fucosyllactose in engineered Escherichia coli BL21star (DE3) by modulation of lactose metabolism and fucosyltransferase". Journal of Biotechnology. 210: 107–115. doi:10.1016/j.jbiotec.2015.06.431. PMID 26193630.
- ^ Chin, Young-Wook; Seo, Nari; Kim, Jae-Han; Seo, Jin-Ho (2016). "Metabolic engineering of Escherichia coli to produce 2'-fucosyllactose via Salvage pathway of guanosine 5'-diphosphate (GDP)-L-fucose". Biotechnology and Bioengineering. 113 (11): 2443–2452. doi:10.1002/bit.26015. PMID 27217241. S2CID 45579939.
- ^ Chin, Young-Wook; Kim, Ji-Yeong; Kim, Jae-Han; Jung, Sang-min; Seo, Jin-Ho (2016). "Improved production of 2'-fucosyllactose in engineered Escherichia coli by expressing putative α-1,2-fucosyltransferase, WcfB from Bacteroides fragilis". Journal of Biotechnology. 257: 192–198. doi:10.1016/j.jbiotec.2016.11.033. PMID 27919692.
- ^ Taylor, Diane E.; Rasko, David A.; Sherburne, Richard; Ho, Clinton; Jewel, Laurence D. (1998). "Lack of correlation between Lewis antigen expression by Helicobacter pylori and gastric epithelial cells in infected patients". Gastroenterology. 115 (5): 1113–1122. doi:10.1016/S0016-5085(98)70082-4. PMID 9797366.
- ^ a b Lee, Won-Heong; Pathanibul, Panchalee; Quarterman, Josh; Jo, Jung-Hyun; Han, Nam-Soo; Miller, Michael J; Jin, Yong-Su; Seo, Jin-Ho (2012). "Whole cell biosynthesis of a functional oligosaccharide, 2'-fucosyllactose, using engineered Escherichia coli". Microbial Cell Factories. 11: 48. doi:10.1186/1475-2859-11-48. PMC 3442965. PMID 22545760.
