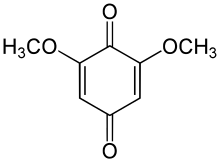
2,6-Dimethoxybenzoquinone
Appearance
| |
| Names | |
|---|---|
| Preferred IUPAC name
2,6-Dimethoxycyclohexa-2,5-diene-1,4-dione | |
| Other names
2,6-Dimethoxy-1,4-benzoquinone; 2,6-DMBQ
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.007.714 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H8O4 | |
| Molar mass | 168.148 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
2,6-Dimethoxybenzoquinone (2,6-DMBQ) is a chemical compound, classified as a benzoquinone, that has been found in Rauvolfia vomitoria[1] and in Tibouchina pulchra.[2]
Toxicity
[edit]At physiological concentrations 2,6-dimethoxybenzoquinone is an antibacterial substance.[3] At higher concentrations there is evidence that it is mutagenic,[4][5] cytotoxic,[5] genotoxic,[6] and hepatotoxic.[7][8][9] Some reports have challenged its mutagenicity[10] and others exclude such a possibility.[11]
References
[edit]- ^ Morris Kupchan, S.; Obasi, Mang E. (1960). "A Note on the Occurrence of 2,6-Dimethoxybenzoquinone in Rauwolfia vomitoria". Journal of the American Pharmaceutical Association (Scientific Ed.). 49 (4): 257–258. doi:10.1002/jps.3030490421. PMID 13853494.
- ^ Jones, Ellery; Ekundayo, Olusegun; Kingston, David G. I. (1981). "Plant Anticancer Agents. XI. 2,6-Dimethoxybenzoquinone as a Cytotoxic Constituent of Tibouchina pulchra". Journal of Natural Products. 44 (4): 493–494. doi:10.1021/np50016a019.
- ^ Nishina, Atsuyoshi; Hasegawa, Kinichi; Uchibori, Tsuyoshi; Seino, Hajime; Osawa, Toshihiko (1991). "2,6-Dimethoxy-p-benzoquinone as an antibacterial substance in the bark of Phyllostachys heterocycla var. Pubescens, a species of thick-stemmed bamboo". Journal of Agricultural and Food Chemistry. 39 (2): 266–269. doi:10.1021/jf00002a009.
- ^ Canonero R; Poggi C Mutagenic activity of 2,6-dimethoxy-1,4-benzoquinone, produced during the nitrosation of dimethophrine, in V 79 cells. Bollettino della Societa italiana di biologia sperimentale (1988), 64(1), 61-8
- ^ a b Brambilla G; Robbiano L; Cajelli E; Martelli A; Turmolini F; Mazzei M Cytotoxic, DNA-damaging and mutagenic properties of 2,6-dimethoxy-1,4-benzoquinone, formed by dimethophrine-nitrite interaction. The Journal of Pharmacology and Experimental Therapeutics (1988), 244(3), 1011-5
- ^ Mazzei M; Roma G; Balbi A; Sottofattori E; Robbiano L Formation of 2,6-dimethoxy-1,4-benzoquinone, a highly genotoxic compound, from the reaction of sodium nitrite with the sympathomimetic drug dimethophrine in acidic aqueous solution. Il Farmaco; edizione scientifica (1988), 43(6), 523-38
- ^ Moore, Gregory A.; Rossi, Luisa; Nicotera, Pierluigi; Orrenius, Sten; O'Brien, Peter J. Quinone toxicity in hepatocytes: studies on mitochondrial calcium release induced by benzoquinone derivatives. Archives of Biochemistry and Biophysics (1987), 259(2), 283-95.
- ^ Siraki, Arno G.; Chan, Tom S.; O'Brien, Peter J. Application of Quantitative Structure-Toxicity Relationships for the Comparison of the Cytotoxicity of 14 p-Benzoquinone Congeners in Primary Cultured Rat Hepatocytes Versus PC12 Cells. Toxicological Sciences (2004), 81(1), 148-159
- ^ Chan, Katie; Jensen, Neil; O'Brien, Peter J. Structure-activity relationships for thiol reactivity and rat or human hepatocyte toxicity induced by substituted p-benzoquinone compounds. Journal of Applied Toxicology (2008), 28(5), 608-620.
- ^ Non-mutagenicity of some wood-related compounds in the bacterial/microsome plate incorporation and microsuspension assays. Mohtashamipur, E., Norpoth, K. International archives of occupational and environmental health. (1984)
- ^ International Journal of Toxicology, 26:253–259, Safety Studies Regarding a Standardized Extract of Fermented Wheat Germ James T. Heimbach JHeimbach LLC, Port Royal, Virginia, USA Gyula Sebestyen Department of Public Health, Faculty of Medicine, Semmelweis University, Budapest, Hungary Gabor Semjen Department of Pharmacology and Toxicology, Faculty of Veterinary Science, Szent Istvan University, Budapest, Hungary Elke Kennepohl Write-Tox Consulting, Spruce Grove, Alberta, Canada
