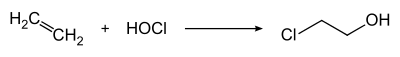2-Chloroethanol
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
2-Chloroethanol[2]
| |||
| Other names | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 3DMet | |||
| 878139 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.003.146 | ||
| EC Number |
| ||
| 25389 | |||
| KEGG | |||
| MeSH | Ethylene+Chlorohydrin | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1135 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C2H5ClO | |||
| Molar mass | 80.51 g·mol−1 | ||
| Appearance | Colourless liquid | ||
| Odor | faint, ether-like | ||
| Density | 1.201 g mL−1 | ||
| Melting point | −62.60 °C; −80.68 °F; 210.55 K | ||
| Boiling point | 127 to 131 °C; 260 to 268 °F; 400 to 404 K | ||
| miscible[3] | |||
| log P | −0.107 | ||
| Vapor pressure | 700 Pa (at 20 °C) | ||
Refractive index (nD)
|
1.441 | ||
| Thermochemistry | |||
Std enthalpy of
combustion (ΔcH⦵298) |
−1.1914 MJ mol−1 | ||
| Hazards | |||
| GHS labelling: | |||
 
| |||
| Danger | |||
| H226, H300, H310, H330 | |||
| P260, P280, P284, P301+P310, P302+P350 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 55 °C (131 °F; 328 K) | ||
| 425 °C (797 °F; 698 K) | |||
| Explosive limits | 5–16% | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
67 mg kg−1 (dermal, rabbit)[citation needed] 72 mg/kg (rat, oral) 81 mg/kg (mouse, oral) 71 mg/kg (rat, oral) 110 mg/kg (guinea pig, oral)[4] | ||
LC50 (median concentration)
|
7.5 ppm (rat, 1 hr) 32 ppm (rat, 4 hr) 260 ppm (guinea pig) 33 ppm (rat, 4 hr) 87 ppm (rat) 115 ppm (mouse)[4] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 5 ppm (16 mg/m3) [skin][3] | ||
REL (Recommended)
|
C 1 ppm (3 mg/m3) [skin][3] | ||
IDLH (Immediate danger)
|
7 ppm[3] | ||
| Related compounds | |||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
2-Chloroethanol is an chemical compound with the formula HOCH2CH2Cl and the simplest chlorohydrin. This colorless liquid has a pleasant ether-like odor. It is miscible with water. The molecule is bifunctional, consisting of both an alkyl chloride and an alcohol functional groups.
Synthesis and applications
2-Chloroethanol is produced by treating ethylene with hypochlorous acid:
2-Chloroethanol was once produced on a large scale as a precursor to ethylene oxide:
This application has been supplanted by the greener direct oxidation of ethylene. Otherwise chloroethanol is used in a number of specialized applications.[6] Several dyes are prepared by the alkylation of aniline derivatives with chloroethanol.[7] It is a building block in the production of pharmaceuticals, biocides and plasticizers. It is also used for manufacture of thiodiglycol. It is a solvent for cellulose acetate and ethyl cellulose, textile printing dyes, in dewaxing, refining of rosin, extraction of pine lignin, and the cleaning of machines.
Environmental aspects
Chloroethanol is a metabolite in the degradation of 1,2-dichloroethane. The alcohol is then further oxidized via chloroacetaldehyde to chloroacetate. This metabolic pathway is topical since billions of kilograms of 1,2-dichloroethane are processed annually as a precursor to vinyl chloride.[8]
Safety
2-Chloroethanol is toxic with an LD50 of 89 mg/kg in rats. Like most organochlorine compounds, chloroethanol combusts to yield hydrogen chloride and phosgene.
In regards to dermal exposure to 2-chloroethanol, the Occupational Safety and Health Administration has set a permissible exposure limit of 5 ppm (16 mg/m3) over an eight-hour time-weighted average, while the National Institute for Occupational Safety and Health has a more protective recommended exposure limit of a 1 ppm (3 mg/m3) exposure ceiling.[9]
It is classified as an extremely hazardous substance in the United States as defined in Section 302 of the U.S. Emergency Planning and Community Right-to-Know Act (42 U.S.C. 11002), and is subject to strict reporting requirements by facilities which produce, store, or use it in significant quantities.[10]
References
- ^ a b c d e f g h i j k l Depositor-supplied synonyms for CID 34
- ^ "2-Chloroethanol". Pubchem. 27 May 2015. Retrieved 27 May 2015.
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0268". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b "Ethylene chlorohydrin". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ http://www.newenv.com/resources/nfpa_chemicals
- ^ Gordon Y. T. Liu, W. Frank Richey, Joanne E. Betso "Chlorohydrins" in Ullmann's Encyclopedia of Industrial Chemistry, 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a06_565
- ^ Roderich Raue and John F. Corbett "Nitro and Nitroso Dyes" in Ullmann's Encyclopedia of Industrial Chemistry, 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a17_383
- ^ 1. Janssen, D. B.; van der Ploeg, J. R. and Pries, F., "Genetics and Biochemistry of 1,2-Dichloroethane Degradation", Biodegradation, 1994, volume 5, pp. 249-57.doi:10.1007/BF00696463
- ^ CDC - NIOSH Pocket Guide to Chemical Hazards
- ^ "40 C.F.R.: Appendix A to Part 355—The List of Extremely Hazardous Substances and Their Threshold Planning Quantities" (PDF) (July 1, 2008 ed.). Government Printing Office. Retrieved October 29, 2011.
{{cite journal}}: Cite journal requires|journal=(help)CS1 maint: postscript (link)