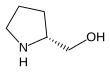
Prolinol
Appearance
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
(R/S) 2-pyrrolidinemethanol
| |||
| Identifiers | |||
| |||
3D model (JSmol)
|
| ||
| ChemSpider | |||
| ECHA InfoCard | 100.157.355 | ||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C5H11NO | |||
| Molar mass | 101.149 g·mol−1 | ||
| Appearance | Liquid | ||
| Density | 1.036 g/mL (liquid) | ||
| Boiling point | 74–76 °C (165–169 °F; 347–349 K) at 2 mmHg | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Irritant | ||
| GHS labelling:[1] | |||

| |||
| Warning | |||
| H315, H319, H335 | |||
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |||
| Flash point | 86 °C (187 °F; 359 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Prolinol is a chiral amino-alcohol that is used as a chiral building block in organic synthesis. It exists as two enantiomers: the D and L forms.
Preparation
[edit]Prolinol is obtained by reduction of the amino acid proline using lithium aluminium hydride.[2][3] Because proline is cheaply available in high optical purity, enantiomerically pure prolinol is also widely available.
Use
[edit]Prolinol is used in broad variety of chemical reactions as chiral ligand, chiral catalyst or chiral auxiliary reagent in the Hajos–Parrish–Eder–Sauer–Wiechert reaction, the Baylis–Hillman reaction, Noyori type reactions and the Michael reaction. [4][5]
See also
[edit]References
[edit]- ^ "D-Prolinol". pubchem.ncbi.nlm.nih.gov. Retrieved 27 December 2021.
- ^ Dickman, D. A.; Meyers, A. I.; Smith, G. A.; Gawley, R. E. "Reduction of α-Amino Acids: L-Valinol". Organic Syntheses; Collected Volumes, vol. 7, p. 530.
- ^ Enders, D.; Fey, P.; Kipphardt, H. "(S)-(–)-1-Amino-2-Methoxymethylpyrrolidine (SAMP) and (R)-(+)-1-Amino-2-Methoxymethylpyrrolidine (RAMP), Versatile Chiral Auxiliaries". Organic Syntheses; Collected Volumes, vol. 8, p. 26.
- ^ Benjamin List (2002). "Proline-catalyzed asymmetric reactions". Tetrahedron. 58 (28): 5573–5590. doi:10.1016/S0040-4020(02)00516-1.
- ^ Shinichi Itsuno; Koichi Ito; Akira Hirao; Seiichi Nakahama (1984). "Asymmetric synthesis using chirally modified borohydrides. Part 2. Enantioselective reduction of ketones with polymeric (S)-prolinol–borane reagent". J. Chem. Soc., Perkin Trans. 1 (12): 2887–2895. doi:10.1039/P19840002887.