Cyanuric triazide
This article needs additional citations for verification. (May 2013) |

| |

| |
| Names | |
|---|---|
| IUPAC name
2,4,6-Triazido-1,3,5-triazine
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C3N12 | |
| Molar mass | 204.117 g·mol−1 |
| Appearance | white crystal |
| Density | 1.73 g·cm−3[1] |
| Melting point | 94 °C (201 °F; 367 K) |
| Boiling point | 150 °C (302 °F; 423 K) Decomposition or explosion |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Explosive substance |
| Flash point | [2] |
| 205 °C (401 °F; 478 K) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cyanuric triazide (C3N12 or (NCN3)3) is described as an environmentally friendly, low toxicity, and organic primary explosive with a detonation velocity of about 7,300 m s−1, and ignition temperature at 205 °C.
Structure
The cyanuric triazide molecule is planar and has three-fold axis symmetry – a triskelion with molecular point group C3h.[3][4] The 1,3,5-triazine (or cyanuric) ring consists of alternating carbon and nitrogen atoms with C–N bond lengths of 1.334 to 1.336 Å. The distance from the center of the ring to each ring carbon atom is 1.286 Å, while the corresponding distance to ring nitrogens is 1.379 Å. Azide groups are linked to the carbon atoms on the cyanuric ring by single bonds with an interatomic distance of 1.399 Å.[5]
Occurrence
This compound is purely synthetic and therefore does not exist in nature.
Synthesis
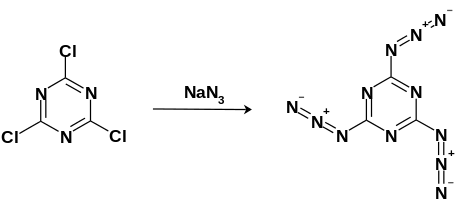
Cyanuric triazide can be synthesized via nucleophilic aromatic substitution by mixing cyanuric trichloride with an excess of sodium azide in refluxing acetone. The white crystals can then be purified via crystallization in −20 °C toluene.[6]
Decomposition reactions
This white polycrystalline solid was found to be stable under standard conditions but is extremely shock sensitive causing it to violently decompose when ground with a mortar. The thermodynamic properties of cyanuric triazide were studied using bomb calorimetry with a combustion enthalpy (H) of 2234 kJ mol−1 under oxidizing conditions and 740 kJ mol−1 otherwise. The former value is comparable to the military explosive RDX, (C3N3)(NO3)3H6, but is not put into use due to its less than favorable stability. Melting point examination showed a sharp melting range to clear liquid at 94–95 °C, gas evolution at 155 °C, orange to brown solution discoloration at 170 °C, orange-brown solidification at 200 °C and rapid decomposition at 240 °C. The rapid decomposition at 240 °C results from the formation of elemental carbon as graphite and the formation of nitrogen gas.[6]
References
- ^ Haynes, W. M.; Lide, D. R. (2012). CRC Handbook of Chemistry and Physics 93rd Ed. CRC Press/Taylor and Francis. ISBN 978-1439880494.
- ^ Mikhail A. Ilyushin, Igor V. Tselinsky And Irina V. Shugalei (2012). "Environmentally Friendly Energetic Materials for Initiation Devices" (PDF). Central European Journal of Energetic Materials. 9 (4): 293–327.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 1291. ISBN 978-0-08-037941-8.
- ^ Housecroft, C. E.; Sharpe, A. G. (2008). Inorganic Chemistry (3rd ed.). Prentice Hall. pp. 94–99. ISBN 978-0-13-175553-6.
- ^ Keßenich, Elmar; Klapötke, Thomas M.; Knizek, Jörg; Nöth, Heinrich; Schulz, Axel (1998). "Characterization, Crystal Structure of 2,4-Bis(triphenylphosphanimino)tetrazolo[5,1-a]-[1,3,5]triazine, and Improved Crystal Structure of 2,4,6-Triazido-1,3,5-triazine". Eur. J. Inorg. Chem. 1998 (12): 2013–2016. doi:10.1002/(SICI)1099-0682(199812)1998:12<2013::AID-EJIC2013>3.0.CO;2-M.
- ^ a b Gillan, Edward G. (2000). "Synthesis of Nitrogen-Rich Carbon Nitride Networks from an Energetic Molecular Azide Precursor". Chemistry of Materials. 12 (12): 3906–3912. doi:10.1021/cm000570y.