Tris(acetylacetonato)cobalt(III)
Appearance
| |
| Names | |
|---|---|
| Other names
Cobalt(III) acetylacetonate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.040.464 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C15H21CoO6 | |
| Molar mass | 356.260 g·mol−1 |
| Appearance | green solid |
| Density | 1.41 g/cm3 |
| Melting point | 213 °C (415 °F; 486 K) |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H302, H317, H334 | |
| P261, P264, P270, P272, P280, P285, P301+P312, P302+P352, P304+P341, P321, P330, P333+P313, P342+P311, P363, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
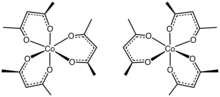
Tris(acetylacetonato)cobalt(III) is the coordination complex with the formula Co(C5H7O2)3. It is a green, diamagnetic solid that is soluble in organic solvents, but not in water. It is prepared by the reaction of cobalt(II) carbonate and acetylacetone in the presence of hydrogen peroxide. The solid is isomorphous with tris(acetylacetonato)iron(III), tris(acetylacetonato)manganese(III), and tris(acetylacetonato)aluminium.[1] These complexes are chiral and often can be resolved into the individual enantiomers. Owing to its solubility in organic solvents, ttris(acetylacetonato)cobalt(III) is used to produce homogeneous catalysts by reduction.[2]
References
- ^ Bryant, Burl E.; Fernelius, W. Conard (1957). "Cobalt(III) Acetylacetonate". Inorganic Syntheses. Vol. 5. pp. 188–189. doi:10.1002/9780470132364.ch53. ISBN 9780470132364.
- ^ Mayo, Peter D.; Tam, William (2002). "Tris(acetoacetonyl)cobalt". Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rn00084. ISBN 0471936235.
