Diethyl ether peroxide
This article needs additional citations for verification. (July 2014) |
| |

| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H10O3 | |
| Molar mass | 106.12 g/mol |
| Density | 1.005 g/cm3 |
| Boiling point | 62 to 64 °C (144 to 147 °F; 335 to 337 K) at 18.7 hPa (reduced pressure) |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Explosive |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Diethyl ether peroxides are a class of organic peroxides that slowly form in diethyl ether upon storage under air, light, or in the presence of metal by autoxidation.[1]
Diethyl ether hydroperoxide
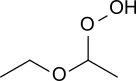
Diethyl ether hydroperoxide (CH3-CH2-O-CH(OOH)-CH3) is a colorless liquid of low viscosity with a pleasant smell. Upon heating it weakly deflagrates, resulting in a fog of acetic acid and water. Diethyl ether hydroperoxide decomposes in the presence of sodium hydroxide and Fe2+-containing salts.
Diethyl ether peroxide, also known as ethylidene peroxide, (-CH(CH3)OO-)n is a polymerization product of diethyl ether hydroperoxide. It is a colorless oily liquid that is an extremely brisant and friction sensitive explosive material. Amounts of less than 5 milligrams can damage chemical apparatuses.[who?] The dangerous properties of ether peroxides are the reason that diethyl ether and other peroxide forming ethers like tetrahydrofuran (THF) or ethylene glycol dimethyl ether (1,2-dimethoxyethane) are avoided in industrial processes.
Tests
Diethyl ether peroxides can be detected with a potassium iodide (KI) solution in acetic acid or potassium iodide / starch paper. A positive test results in the formation of iodine (I2) that causes a yellow or brown color of the ether phase or a dark bluish spot on the paper strip.[2]
Degradation
Ether peroxides can be destroyed by disproportionation to acetaldehyde with Fe2+ or Mn2+ ions or with triphenylphosphine (PPh3). The resulting aldehyde has to be removed to prevent a rapid back-formation of peroxides.
References
- ^ A. Rieche, R. Meister (1936). "Modellversuche zur Autoxidation der Äther". Angewandte Chemie (in German). 49 (5): 106.
- ^ "Peroxide Forming Solvents". Sigma-Aldrich. Retrieved 2014-07-09.


