Paclitaxel trevatide
 | |
| Clinical data | |
|---|---|
| Other names | NG1005; GRN1005 |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C257H308N32O79 |
| Molar mass | 5109.436 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Paclitaxel trevatide (development codes NG1005 and GRN1005) is an experimental chemotherapy drug that is under development by Angiochem Inc, a Canadian biotech company. Phase II clinical trials have completed for several indications, and the company is preparing for phase III trials.[1]
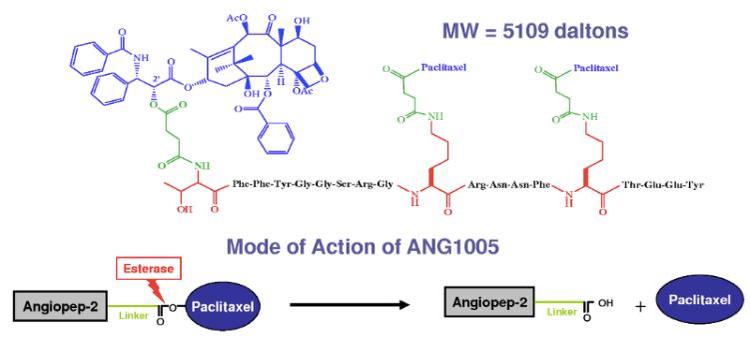
Paclitaxel trevatide is a paclitaxel-Angiopep-2 conjugate. Various Angiopep vectors have been composed and differ by their anti-cancer moieties. This has then been shown to be a prospective cancer therapy drug that can not only be conjugated to paclitaxel but also peptides, monoclonal antibodies, siRNA and many other biological materials. Paclitaxel trevatide has the potential to treat a variety of CNS diseases including glioma.[2] Research has shown reduction in tumor growth in mice and rats with glioblastoma.[3][4][5]
Mechanism of action[edit]
This drug was specifically designed to treat brain tumors. Because of the blood brain barrier (BBB), many cancer therapy drugs are prevented from passing through the brain capillaries into the parenchyma.[6] Paclitaxel is generally prevented from reaching its target in the cell due to the presence of the efflux pump P-glycoprotein (P-gp) at the barrier. This is known as a multidrug resistant-associated protein (MRP1) that causes resistance amongst many organic drugs that are not conjugated to acidic ligands.[7] This receptor is an ATP-driven transporter that will pump drugs, drug metabolites, and endogenous metabolites out of the cell.[citation needed]
Paclitaxel trevatide contains paclitaxel, which stabilizes microtubule polymer formation. Microtubules are composed of polymers consisting of the protein tubulin. Therefore, paclitaxel binds at the site of β-tubulin and induces polymerization, thus protecting the microtubule from disassembling during mitosis. This blocks the progression of mitosis due to a prolonged activation of the microtubule in the mitotic checkpoint, resulting in cell apoptosis or reversion to the G0 phase. However, paclitaxel trevatide will effectively transport across the BBB with approximately a 100-fold higher transport rate compared to a free paclitaxel.[8] Paclitaxel trevatide will cross the capillary medium via receptor mediated transcytosis of the low-density lipoprotein receptor-related protein 1 (LRP1) which is upregulated in some cancers.[9] Ester hydrolyzing enzymes (esterases) then catalyze a highly stereospecific reaction that results in hydrolysis of the paclitaxel trevatide ester to carboxylic acids. This results in the intracellular release of paclitaxel and subsequent action on tubulin.[citation needed]
Concentration has been found to play a key role paclitaxel trevatide's administration. Studies show that a high concentration will suppress microtubule detachment which is necessary for mitosis to occur.[10] The microenvironment of cells may also differ from that of rapidly proliferating cancer cells. This is generally more oxygen deficient and acidic. These conditions will then result in a selective pressure towards cancer cell proliferation. It has been shown that cancer cell lines exposed to this hypoxic or serum-deprived condition will upregulate LRP1 expression, thus leading to an increased uptake of paclitaxel trevatide.[11] However, these receptors are also present in non-cancerous cells, thus presenting a challenge due to non-specific targeting. It is then believed that because these receptors are overexposed in tumors, that receptor binding may be in favor of the cancer cells.[citation needed]
Clinical trials[edit]
In 2008, two phase I clinical trials of paclitaxel trevatide were started; one in patients with advanced cancer and brain metastases,[12] and another in patients with recurrent malignant glioma.[13] Favorable initial tolerability results in brain cancer were reported in March 2009.[14] and more in October 2009 and updated in June 2010.[9] In 2014, paclitaxel trevatide received orphan drug designation from FDA for the treatment of glioblastoma multiform[15] As of January 2016, Angiochem has conducted phase II trials for the treatment of breast cancer with brain metastasis, glioblastoma, and high-grade glioma.[16]
Results from the phase II trial in patients with breast cancer and leptomeningeal carcinomatosis, a fatal form of brain metastasis, were presented at ASCO in 2016.[17]
References[edit]
- ^ "Angiochem Announces Successful End-of-Phase 2 Meeting with FDA for ANG1005 | Angiochem: Peptide-Antibody Conjugates that Cross the Blood Brain Barrier". angiochem.com.
- ^ "ANG1005 - A Promising New Targeted Taxane Derivative". Archived from the original on 27 May 2013. Retrieved 5 June 2013.
- ^ Régina A, Demeule M, Ché C, Lavallée I, Poirier J, Gabathuler R, Béliveau R, Castaigne JP (2008). "Antitumour activity of ANG1005, a conjugate between paclitaxel and the new brain delivery vector Angiopep-2". Br J Pharmacol. 155 (2): 185–197. doi:10.1038/bjp.2008.260. PMC 2538693. PMID 18574456.
- ^ Thomas FC, Taskar K, Rudraraju V, Goda S, Thorsheim HR, Gaasch JA, Mittapalli RK, Palmieri D, Steeg PS, Lockman PR, Smith QR (November 2009). "Uptake of ANG1005, a Novel Paclitaxel Derivative, Through the Blood-Brain Barrier into Brain and Experimental Brain Metastases of Breast Cancer". Pharm Res. 26 (11): 2486–94. doi:10.1007/s11095-009-9964-5. PMC 2896053. PMID 19774344.
- ^ Patel MM, Goyal BR, Bhadada SV, Bhatt JS, Amin AF (2009). "Getting into the brain: approaches to enhance brain drug delivery". CNS Drugs. 23 (1): 35–58. doi:10.2165/0023210-200923010-00003. PMID 19062774. S2CID 26113811.
- ^ Thomas FC, Taskar K, Rudraraju V, et al. (November 2009). "Uptake of ANG1005, a Novel Paclitaxel Derivative, Through the Blood-Brain Barrier into Brain and Experimental Brain Metastases of Breast Cancer". Pharm. Res. 26 (11): 2486–94. doi:10.1007/s11095-009-9964-5. PMC 2896053. PMID 19774344.
- ^ Shao K, Huang R, Li J, Han L, Ye L, Lou J, Jiang C (2010). "Angiopep-2 modified PE-PEG based polymeric micelles for amphotericin B delivery targeted to the brain". J Control Release. 147 (1): 118–26. doi:10.1016/j.jconrel.2010.06.018. PMID 20609375.
- ^ "Influence of glioma tumour microenvironment on the transport of ANG1005 via low-density lipoprotein receptor-related protein 1" (PDF).
- ^ a b "Angiochem Presents Complete Phase 1 /2 Clinical Data for ANG1005: Further Demonstrating Benefits of Targeting LRP-1 Pathway in Cancer". Pharmalive.com. Archived from the original on 2011-07-15. Retrieved 2010-06-08. (paid subscription required)[unreliable medical source?][verification needed]
- ^ Régina A, Demeule M, Ché C, et al. (September 2008). "Antitumour activity of ANG1005, a conjugate between paclitaxel and the new brain delivery vector Angiopep-2". Br. J. Pharmacol. 155 (2). British Journal of Pharmacology: 185–97. doi:10.1038/bjp.2008.260. PMC 2538693. PMID 18574456.
- ^ "nfluence of glioma tumour microenvironment on the transport of ANG1005 via low-density lipoprotein receptor-related protein" (PDF). British Journal of Cancer.
- ^ "A Phase 1, Open-Label, Dose Escalation Study of ANG1005 in Patients With Advanced Solid Tumors and Metastatic Brain Cancer". 3 June 2010. Retrieved 16 August 2010.
- ^ "angiochems-ang1005-demonstrates-preliminary-clinical-safety-and-tolerability-in-brain-cancers" (Press release). CheckOrphan.com. Halsin Partners. Archived from the original on 22 July 2012. Retrieved 16 August 2010.[unreliable medical source?]
- ^ "AngioChem's ANG1005 Shows Promise In The Treatment Of Brain Cancers". 23 October 2008.
- ^ "Angiochem's ANG1005 Received Orphan Drug Designation from FDA for the Treatment of Glioblastoma multiform | Angiochem: Peptide-Antibody Conjugates that Cross the Blood Brain Barrier". angiochem.com.
- ^ "ANG1005 in Patients With Recurrent High-Grade Glioma". clinicaltrials.gov.
- ^ "Angiochem's ANG1005 Shows Clinical Benefit and Prolonged Survival in Phase II Trial | Angiochem: Peptide-Antibody Conjugates that Cross the Blood Brain Barrier". angiochem.com.
External links[edit]
- Mazza M, Uchegbu IF, Schätzlein AG (September 2008). "Cancer and the blood–brain barrier: 'Trojan horses' for courses?". Br J Pharmacol. 155 (2): 149–51. doi:10.1038/bjp.2008.274. PMC 2538701. PMID 18587417.
- "ANG1005 Crosses The Blood-Brain Barrier To Reduce Tumor Size And Is Effective In Resistant Tumors". Medical News Today.
- "AngioChem's ANG1005 demonstrates preliminary clinical safety and tolerability in brain cancers" (Press release). Eurekalert. Halsin Partners. October 2008.