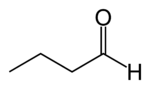
Butyraldehyde
| |

| |
| Names | |
|---|---|
| IUPAC name
butyraldehyde
| |
| Systematic IUPAC name
butanal | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.004.225 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H8O | |
| Molar mass | 72.11 g/mol |
| Density | 0.8 g/ml |
| Melting point | −99 °C |
| Boiling point | 74.8 °C016 |
| 7.6 g/100 mL (20 °C) | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | −7 °C |
| Explosive limits | 2.5–12.5% |
| Related compounds | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Butyraldehyde, also known as butanal, is an organic compound with the formula CH3(CH2)2CHO. This compound is the aldehyde derivative of butane. It is a colourless flammable liquid that smells like sweaty feet. It is miscible with most organic solvents.
Production
Butyraldehyde is produced almost exclusively by the hydroformylation of propylene:
- CH3CH=CH2 + H2 + CO → CH3CH2CH2CHO
Traditionally, hydroformylation was catalyzed by cobalt carbonyl and later rhodium complexes of triphenylphosphine. The dominant technology involves the use of rhodium catalysts derived from the water-soluble ligand Tppts. An aqueous solution of the rhodium catalyst converts the propylene to the aldedyde, which forms a lighter immiscible phase. About 6 billion kilograms are produced annually by hydroformylation.
Butyraldehyde can be produced by the catalytic dehydrogenation of n-butanol. At one time, it was produced industrially by the catalytic hydrogenation of crotonaldehyde, which is derived from acetaldehyde.[2]
Upon prolonged exposure to air, butyraldehyde oxidizes to form butyric acid.
References
- ^ Merck Index, 11th Edition, 1591
- ^ Boy Cornils, Richard W. Fischer, Christian Kohlpaintner "Butanals" in Ullmann's Encyclopedia of Industrial Chemistry, 2000, Wiley-VCH, Weinheim. doi:10.1002/14356007.a04_447
External links

