Cyanuric chloride

| |

| |
| Names | |
|---|---|
| IUPAC name
2,4,6-Trichloro-1,3,5-triazine
| |
| Other names
Trichlorotriazine
s-Triazine trichloride Cyanuryl chloride | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.003.287 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UN number | 2670 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C3Cl3N3 | |
| Molar mass | 184.41 g/mol |
| Appearance | White powder |
| Density | 1.32 g/cm3 |
| Melting point | 154 °C |
| Boiling point | 192 °C |
| hydrolyzes | |
| Solubility in organic solvents | soluble |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Related compounds | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cyanuric chloride is the inorganic compound with the formula (NCCl)3. This colorless solid is the chlorinated derivative of 1,3,5-triazine. It is the trimer of cyanogen chloride.[1] Cyanuric chloride is the main precursor to the popular but controversial herbicide atrazine.
Production
Cyanuric acid is prepared in two steps from hydrogen cyanide via the intermediacy of cyanogen chloride, which is trimerized at elevated temperatures over a carbon catalyst:
- HCN + Cl2 → ClCN + HCl
- 3 ClCN → (ClCN)3
In 2005, approximately 200,000 tonnes were produced.[2]
Industrial Uses
It is estimated that 70% of cyanuric chloride is used in the preparation of the triazine-class pesticides, especially atrazine. Such reactions rely on the easy displacement of the chloride with nucleophiles such as amines:
- (ClCN)3 + 2 RNH2 → (RNHCN)(ClCN)2 + RNH3+Cl-
Other triazine herbicides, such as simazine, anilazine and cyromazine are made in an analogous way.[3]
Cyanuric chloride is also used as a precursor to dyes and crosslinking agents. The largest class of these dyes are the sulfonated triazine-stilbene optical brighteners (OBA) or fluorescent whitening agents (FWA) commonly found in detergent formulas and white paper.[2] Many reactive dyes also incorporate a triazine ring. They are also manufactured by way of the chloride displacement reaction shown above.[3]
Organic synthesis
In one specialized application, cyanuric chloride is employed as a reagent in organic synthesis for the conversion of alcohols and carboxylic acids into alkyl and acyl chlorides, respectively:[4]
It is also used as a dehydrating agent and for the activation of carboxylic acids for reduction to alcohols. Heating with DMF gives "Gold's reagent" Me2NCH=NCH=NMe2+Cl-, which is a versatile source of aminoalkylations and a precursor to heterocycles.[5][6]
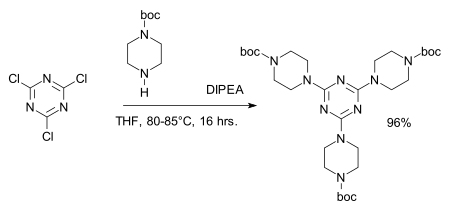
The chloride centers are easily replaced by amines to give melamine derivatives, for example in the synthesis of dendrimers:[7][8]
It is also employed the synthesis of an experimental adenosine receptor ligand.[9]:
References
- ^ Cyanuric chloride at Chemicalland21.com
- ^ a b Klaus Huthmacher, Dieter Most "Cyanuric Acid and Cyanuric Chloride" in Ullmann's Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a08_191.
- ^ a b Ashford's Dictionary of Industrial Chemicals, 3rd edition, 2011, pages 2495-8
- ^ K. Venkataraman, and D. R. Wagle (1979). "Cyanuric chloride : a useful reagent for converting carboxylic acids into chlorides, esters, amides and peptides". Tet. Lett. 20 (32): 3037–3040. doi:10.1016/S0040-4039(00)71006-9.
- ^ Probst, D. A.; Hanson, P. R.; Barda, D. A. "Cyanuric Chloride" in Encyclopedia of Reagents for Organic Synthesis, 2004, John Wiley & Sons. doi:10.1002/047084289X.rn00320
- ^ John T. Gupton; Steven A. Andrews (1990). "β-Dimethylaminomethylenation: N,N-Dimethyl-N'-p-tolylformamidine". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 7, p. 197. - ^ Abdellatif Chouai and Eric E. Simanek (2008). "Kilogram-Scale Synthesis of a Second-Generation Dendrimer Based on 1,3,5-Triazine Using Green and Industrially Compatible Methods with a Single Chromatographic Step". J. Org. Chem. 73 (6): 2357–2366. doi:10.1021/jo702462t. PMID 18307354.
- ^ Reagent: DIPEA, amine protective group: BOC
- ^ WO application 03101980, "1,3,5-TRIAZINE DERIVATIVES AS LIGANDS FOR HUMAN ADENOSINE-A3 RECEPTORS", published 2003-12-11 (Reagent number two: norephedrine, base DIPEA)