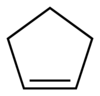
Cyclopentene
Appearance
This article needs additional citations for verification. (June 2012) |
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Cyclopentene
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.005.030 | ||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C5H8 | |||
| Molar mass | 68.11 g/mol | ||
| Density | 0.771 g/cm3 | ||
| Melting point | −135 °C (−211 °F; 138 K) | ||
| Boiling point | 44 to 46 °C (111 to 115 °F; 317 to 319 K) | ||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
| Flash point | −29 °C (−20 °F; 244 K) | ||
| Related compounds | |||
Related compounds
|
Cyclopentadiene Cyclobutene | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Cyclopentene is a chemical compound with the formula Template:Carbon5Template:Hydrogen8.[1] It is a colorless liquid with a petrol-like odor. It is one of the cycloalkenes.
Cyclopentene is produced industrially in large amounts. It is used as a monomer for synthesis of plastics, and in a number of chemical syntheses.
It can be obtained from vinylcyclopropane in the vinylcyclopropane-cyclopentene rearrangement.
References
- ^ "cyclopentene". Retrieved June 15, 2012.