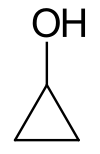
Cyclopropanol
Appearance
| |
| Names | |
|---|---|
| IUPAC name
Cyclopropanol
| |
| Other names
Cyclopropyl alcohol, Hydroxycyclopropane
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.217.724 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C3H6O | |
| Molar mass | 58.080 g·mol−1 |
| Density | 0.917 g/mL[1] |
| Boiling point | 101 to 102 °C (214 to 216 °F; 374 to 375 K)[2] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cyclopropanol is an organic compound with the chemical formula C3H6O. It contains a cyclopropyl group with a hydroxyl group attached to it. The compound is highly unstable due to the three-membered ring, and is susceptible to reactions that open the ring. It is highly prone to rearrangement, undergoing structural isomerization to form propanal.[3][4] This property is useful synthetically: cyclopropanol can be used as a synthon for the homoenolate of propanal. The chemical is also useful as a reagent to introduce a cyclopropyl group into ester, sulfate, and amine linkages. The resulting cyclopropyl-containing compounds have been used in investigations of potential antiviral drugs[5] and of modulators of protein trafficking.[6]
References
- ^ "Small-Ring Compounds. VI. Cyclopropanol, Cyclopropyl Bromide and Cyclopropylamine". J. Am. Chem. Soc. 73 (7): 3176–3179. 1951. doi:10.1021/ja01151a053.
{{cite journal}}: Unknown parameter|authors=ignored (help) - ^ "Enzymatic hydrolysis of cyclopropyl acetate. A facile method for medium- and large-scale preparations of cyclopropanol". Tetrahedron Lett. 28 (24): 2767–2768. 1987. doi:10.1016/S0040-4039(00)96204-X.
{{cite journal}}: Unknown parameter|authors=ignored (help) - ^ "The Reaction of Epichlorohydrin with the Grignard Reagent". J. Am. Chem. Soc. 64 (3): 484–487. 1942. doi:10.1021/ja01255a004.
{{cite journal}}: Unknown parameter|authors=ignored (help) - ^ "The Reaction of Epichlorohydrin with the Grignard Reagent. Some Derivatives of Cyclopropanol". J. Am. Chem. Soc. 65 (9): 1782–1783. 1943. doi:10.1021/ja01249a507.
{{cite journal}}: Unknown parameter|authors=ignored (help) - ^ WO application 2009005677, Cottell, J. J.; Link, J. O. Schroeder, S. D.; Taylor, J.; Tse, W.; Vivian, R. W.; Yang, Z.-Y., "Antiviral compounds", published 2009-01-08
- ^ WO application 2009062118, Bulawa, C. E.; Devit, M.; Elbaum, D., "Modulators of protein trafficking", published 2009-05-14
