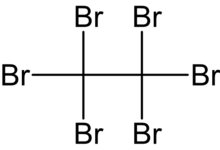
Hexabromoethane
Appearance
| |
| Names | |
|---|---|
| Other names
perbromoethane
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2Br6 | |
| Molar mass | 503.446 g·mol−1 |
| Appearance | yellowish crystals |
| Boiling point | 210–215 °C (410–419 °F; 483–488 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Hexabromoethane (HBE, perbromoethane) is a perbromocarbon with the chemical formula C2Br6. It is a yellowish white crystalline solid.[1] It decomposes to tetrabromoethylene upon heating.[2] Like many other halocarbons, HBE decomposes when exposed to radiation.[3]
See also
[edit]References
[edit]- ^ Holloway, M. D., Holloway, E. (2020). Dictionary of Industrial Terminology.
- ^ Nefedov, OM; Maltsev, AK; Svyatkin, VA (1976),Direct spectroscopic study of the mechanism of thermal decomposition of hexahaloethanes and their silicon and germanium analogs using matrix isolation, Seriya Khimicheskaya
- ^ Iyer, RM; Willard, JE, (1967) Production and Annealing of Br2 in the Radiolysis of Polycrystalline C2Br6: An In Situ Determination, The Journal of Chemical Physics. 46 (9): 3501–3506.
