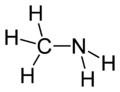
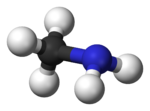
Methylamine
| |||

| |||
| Names | |||
|---|---|---|---|
| IUPAC name
aminomethane, methanamine
| |||
| Other names
monomethylamine
MMA | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.000.746 | ||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| CH5N | |||
| Molar mass | 31.058 g·mol−1 | ||
| Appearance | Colorless Gas | ||
| Density | d40.699 (−10.8 °C) / 0.902 g/cm³, 40w/w% in water | ||
| Melting point | −94 °C (179.15 K) | ||
| Boiling point | −6 °C (267.2 K) | ||
| 108 g/100 mL (20 °C) | |||
| Acidity (pKa) | 10.62[1] | ||
| Viscosity | 0.23 cP at 0 °C | ||
| Structure | |||
| tetrahedral | |||
| 1.31 D (gas) | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Corrosive liquid and gas, inhalation hazard, flammable. | ||
| NFPA 704 (fire diamond) | |||
| Flash point | 8 °C | ||
| Related compounds | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Methylamine is the organic compound with a formula of CH3NH2. This colourless gas is a derivative of ammonia, wherein one H atom is replaced by a methyl group. It is the simplest primary amine. It is sold as a solution in methanol, ethanol, THF, and water, or as the anhydrous gas in pressurized metal containers. Industrially methylamine is sold in its anhydrous form in pressurized railcars and tank trailers. It has a strong odor similar to fish. Methylamine is used as a building block for the synthesis of many other commercially available compounds. Hundreds of millions of kilograms are produced annually.
Production
Methylamine is prepared commercially by the reaction of ammonia with methanol in the presence of a silicoaluminate catalyst. Dimethylamine and trimethylamine are coproduced; the reaction kinetics and reactant ratios determine the ratio of the three products.[2]
- CH3OH + NH3 → CH3NH2 + H2O
In this way, more than 400M kg are produced annually.
In the laboratory, methylamine hydrochloride is readily prepared by treating formaldehyde with ammonium chloride.[3]
The colourless hydrochloride salt can be converted to the amine by the addition of strong base, like NaOH:
- CH3NH2·HCl + NaOH → CH3NH2 + NaCl + H2O
Methylamine was first prepared by Wurtz by the hydrolysis of methyl isocyanate and related compounds.[4]
Reactivity and applications
Methylamine is a good nucleophile as it is highly basic and unhindered, though, as an amine it is considered a weak base. Its use in organic chemistry is pervasive. Some reactions involving simple reagents include: with phosgene to methyl isocyanate, with carbon disulfide and sodium hydroxide to the sodium methyldithiocarbamate, with chloroform and base to methyl isocyanide and with ethylene oxide to methylethanolamines.
Representative commercially significant chemicals produced from methylamine include the pharmaceuticals ephedrine and theophylline, the pesticides carbofuran, carbaryl, and metham sodium, and the solvents N-methylformamide and N-methylpyrrolidone. The preparation of some surfactants and photographic developers require methylamine as a building block.[4]
Liquid methylamine can be used as a solvent analogous to liquid ammonia. It shares some of the properties of liquid ammonia, but is better for dissolving organic substances, in the same way that methanol is better than water.[5] Methylamine can also be used for scavenging H2S from hydrocarbon in refining applications.
Biological chemistry
Methylamine arises naturally as the result of putrefaction and is a substrate for methanogenesis.[6] It serves as a buffering agent in the lumen of the chloroplast in plants, effectively siphoning off protons that are heading for ATP synthase.[citation needed]
Safety
The [[LD50]] (mouse) is 2400 mg/m3. Methylamine is also controlled as a List 1 substance by the United States Drug Enforcement Administration (DEA); the DEA lists methylamine as a precursor (to methamphetamine).
References
- ^ Hall, H.K., J. Am. Chem. Soc., 1957, 79, 5441.
- ^ Corbin D.R.; Schwarz S.; Sonnichsen G.C. (1997). "Methylamines synthesis: A review". Catalysis Today. 37 (2): 71–102. doi:10.1016/S0920-5861(97)00003-5.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Marvel, C. S.; Jenkins, R. L. (1941). "Methylamine Hydrochloride". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 1, p. 347. - ^ a b Karsten Eller, Erhard Henkes, Roland Rossbacher, Hartmut Höke "Amines, Aliphatic" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005.
- ^ H. D. Gibbs (1906). "Liquid methylamine as a solvent, and a study of its chemical reactivity". J. Am. Chem. Soc. 28: 1395–1422. doi:10.1021/ja01976a009.
- ^ Thauer, R. K., "Biochemistry of Methanogenesis: a Tribute to Marjory Stephenson", Microbiology, 1998, 144, 2377-2406.