N-Methylformamide

| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
N-Methylformamide | |
| Systematic IUPAC name
N-Methylmethanamide | |
| Other names
Methylformide
NMF | |
| Identifiers | |
3D model (JSmol)
|
|
| 1098352 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.004.205 |
| EC Number |
|
| KEGG | |
| MeSH | methylformamide |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2H5NO | |
| Molar mass | 59.068 g·mol−1 |
| Appearance | Colourless liquid |
| Odor | fishy, ammoniacal |
| Density | 1.011 g mL−1 |
| Melting point | −4 °C (25 °F; 269 K) |
| Boiling point | 182.6 °C; 360.6 °F; 455.7 K |
| Miscible | |
Refractive index (nD)
|
1.432 |
| Thermochemistry | |
Heat capacity (C)
|
125.2 J K−1 mol−1 |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H312, H360 | |
| P280, P308+P313 | |
| Flash point | 111 °C (232 °F; 384 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
4 g kg−1 (oral, rat) |
| Related compounds | |
Related alkanamides
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
N-Methylformamide (NMF) is a colorless, nearly odorless, organic compound and secondary amide with molecular formula CH3NHCHO, which is a liquid at room temperature. NMF is mainly used as a reagent in various organic syntheses with limited applications as a highly polar solvent.[1]
NMF is closely related to other formamides, notably formamide and dimethylformamide (DMF). However, industrial use and production of NMF are far less than for either of these other formamides. DMF is favored over NMF as a solvent due to its greater stability.[1] Annual production of NMF can be assumed to be significantly less than the production of either formamide (100,000 tons) or DMF (500,000 tons).[1]
Structure and properties
[edit]Because of slow rotation about the N-C(O) bond, NMF exists as two rotamers that can be distinguished by NMR spectroscopy. The two principal resonance structures for one of these rotamers is shown:
This description highlights the partial double bond that exists between the carbonyl carbon and nitrogen, which raises the rotational barrier. Thus, the molecule is not able to freely rotate around its main axis and the (E)-configuration is preferred due to steric repulsion of the larger substituents.[2]
This molecule has been tentatively identified in interstellar space by the ALMA radio telescope. It may have formed on dust grains. This could prove to be a key molecule for interstellar pre-biotic chemistry due to its peptide bond.[3]
Preparation
[edit]NMF is typically prepared by allowing methylamine to react with methyl formate:[1]
- CH3NH2 + HCOOCH3 → HCONHCH3 + CH3OH
A less common alternative to this process is transamidation involving formamide:[1]
- HCONH2 + CH3NH2 → HCONHCH3 + NH3
Uses
[edit]NMF is a specialized solvent in oil refineries. It is a precursor in specialized amidation reactions where formamide would not be suitable. These reactions can generally be categorized by the following equation:
- R-Lg + CH3NHCHO → R-NCH3CHO + H-Lg (where Lg is a leaving group).[1]
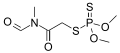
Laboratory uses
[edit]NMF is the precursor to methyl isocyanide, a ligand in coordination chemistry.[4] NMF is used as a solvent in Aluminum Electrolytic Capacitors.
References
[edit]- ^ a b c d e f Bipp, H.; Kieczka, H. "Formamides". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a12_001. ISBN 978-3527306732.
- ^ . doi:10.1002/bip.360360405.
{{cite journal}}: Cite journal requires|journal=(help); Missing or empty|title=(help) - ^ Belloche, A.; et al. (May 2017). "Rotational spectroscopy, tentative interstellar detection, and chemical modeling of N-methylformamide". Astronomy & Astrophysics. 601: 41. arXiv:1701.04640. Bibcode:2017A&A...601A..49B. doi:10.1051/0004-6361/201629724. A49.
- ^ Schuster, R. E.; Scott, J. E.; Casanova, J. Jr. (1966). "Methyl Isocyanide". Organic Syntheses. 46: 75; Collected Volumes, vol. 5, p. 772.

