Osteoblast
| Osteoblast | |
|---|---|
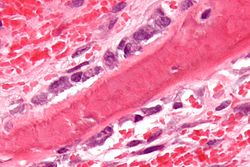 Osteoblasts (blue) rimming a bony spicule (pink - on diagonal of image). In this routinely fixed and decalcified (bone mineral removed) tissue, the osteoblasts have retracted and are separated from each other and from their underlying matrix. In living bone, the cells are linked by tight junctions and gap junctions, and integrated with underlying osteocytes and matrix H&E stain. | |
| Details | |
| Identifiers | |
| Greek | osteoblast |
| MeSH | D010006 |
| TH | H2.00.03.7.00002 |
| FMA | 66780 |
| Anatomical terminology | |
Osteoblast (from the Greek combining forms for "bone", ὀστέο-, osteo- and βλαστάνω, blastanō "germinate") are cells with single nuclei that synthesize bone. However, in the process of bone formation, osteoblastos function in groups of connected cells. Individual cells cannot make bone, and the group of organized osteoblasts together with the bone made by a unit of cells is usually called the osteon; the basis of this is discussed in "Organization and ultrastructure of osteoblasts" below.
Osteoblasts are specialized, terminally differentiated products of mesenchymal stem cells.[1] They synthesize very dense, crosslinked collagen, and several additional specialized proteins in much smaller quantities, including osteocalcin and osteopontin, which compose the organic matrix of bone.
In organized groups of connected cells, osteoblasts produce a calcium and phosphate-based mineral, hydroxyapatite, that is deposited, in a highly regulated manner, into the organic matrix forming a very strong and dense mineralized tissue - the mineralized matrix. This is further discussed in "Mineralization of bone" below. The mineralized skeleton is the main support for the bodies of air breathing vertebrates. It also is an important store of minerals for physiological homeostasis including both acid-base balance and calcium or phosphate maintenance.[2][3]
Definitions of bone-forming and -degrading cells
Osteoblasts are the major cellular component of bone. The functional part of bone, the bone matrix, is entirely extracellular (outside of the cells). The bone matrix consists of protein and mineral. The protein is called the organic matrix; it is synthesized first, and then the mineral is added. The vast majority of the organic matrix is collagen, which provides tensile strength. Then the matrix is mineralized by deposition of a calcium-phosphate-hydroxide salt called hydroxyapatite (alternative name, hydroxylapatite). This mineral is very hard, and provides compressive strength. Thus, the collagen and mineral together are a composite material with excellent tensile and compressive strength, which can bend under a strain and recover its shape without damage. This is called elastic deformation. Forces that exceed the capacity of bone to behave elastically may cause failure, typically bone fractures.
Normally, almost all of the bone matrix, in the air breathing vertebrates, is mineralized by the osteoblasts. Before the organic matrix is mineralized, it is called osteoid. Osteoblasts that are buried in matrix are called osteocytes. During active bone formation, the surface layer of osteoblasts consists of cuboidal cells, called active osteoblasts. When the bone-forming unit is not actively synthesizing bone, the surface osteoblasts are flattened and are called inactive osteoblasts. Osteocytes remain alive and are connected by cell processes to a surface layer of osteoblasts. Osteocytes have important functions in maintenance of the skeleton.
Bone is a dynamic tissue that is constantly being reshaped by osteoblasts, which produce and secrete matrix proteins and transport mineral into the matrix, and osteoclasts, which break down the tissue. The osteoclasts are the only cell type other than osteoblasts and osteocytes that are structural components of bone, although within the hollow inside of bones are many other cell types of the bone marrow. Components of the bone marrow that are essential for osteoblast bone formation include mesenchymal stem cells, the precursors of osteoblasts, and blood vessels which supply oxygen and nutrients for bone formation. Bone is a highly vascular tissue, and active formation of blood vessel cells, also from mesenchymal stem cells, is essential to support the metabolic activity of bone. The balance of bone formation and bone resorption tends to be negative with age, particularly in post-menopausal women,[4] often leading to a loss of bone serious enough to cause fractures, which is called osteoporosis.
Osteogenesis
Osteoblasts arise from mesenchymal stem cells. Mesenchymal stem cells are found in large numbers in the periosteum, the fibrous-like layer on the outside surface of bones, and in the bone marrow. During cellular differentiation of osteoblasts, the developing progenitor cells express the regulatory transcription factor Cbfa1/Runx2, which is also active in chondrocytes. A second important transcription factor required for osteoblastic differentiation is osterix.[5] Osteoprogenitors differentiate under the influence of growth factors, although isolated mesenchymal stem cells in tissue culture form osteoblasts under permissive conditions that include vitamin C and substrates for alkaline phosphatase, a key enzyme that provides high concentrations of phosphate at the site of mineral deposition.[6]
In the living organism, bone development is very complex; in most cases it follows the formation of a first skeleton of cartilage made by chondrocytes, which is then removed and replaced by bone, made by osteoblasts. Key growth factors in skeletal differentiation include bone morphogenetic proteins (BMPs), which determine to a major extent where bone differentiation occurs and where joint spaces are left between bones. The system of cartilage replacement by bone in the living organism has a complex regulatory system. It includes the bone morphogenetic proteins, in particular BMP2, that also regulate early patterning of the skeleton. Other growth factors that are important include transforming growth factor beta (TGF-β), which is part of a superfamily of proteins that include BMPs, which possess common signaling elements in the TGF beta signaling pathway. TGF-β is particularly important in cartilage differentiation, which in most cases precedes osteoblast-mediated bone formation. An additional family of essential bone regulatory factors is the fibroblast growth factors (FGFs), which determine where skeletal elements occur in relation to the skin.
Bone formation is regulated by steroid and protein hormones
Many other regulatory systems are involved in the transition of cartilage to bone and in maintenance of bone, reflecting that the skeleton is a large organ that is formed and degraded throughout life in the air breathing vertebrates, and that the skeleton is important both as a supporting structure and for maintenance of calcium, phosphate, and acid-base status in the whole organism.[7] A particularly important bone-targeted hormonal regulator is parathyroid hormone (PTH). Parathyroid hormone is a protein made by the parathyroid gland under the control of serum calcium activity.[8] PTH also has important systemic functions, including to keep the serum calcium nearly constant regardless of calcium intake. A common misconception is that more dietary calcium results in more calcium in the bones; minor increases in blood calcium do occur after large doses of dietary calcium. However, this is not a significant mechanism supporting osteoblast bone formation, except in the condition, rare in the developed world, of very low dietary calcium; further, abnormally high dietary calcium raises the risk of serious health consequences not directly related to bone mass including heart attacks and strokes.[9] Intermittent PTH stimulation increases osteoblast activity, although PTH is bifunctional and mediates bone matrix degradation at higher concentrations.
The skeleton is also modified for reproduction and in response to nutritional and other hormone stresses; it responds to steroids, including estrogen and glucocorticoids, which are important in reproduction and in regulation of energy metabolism. Since bone is in a central position in physiology, and bone turnover involves major expenditures of energy for synthesis and degradation, many additional central signals including pituitary hormones regulate osteoblasts. Two of these are adrenocorticotropic hormone[10] and follicle stimulating hormone.[11] The physiological role for responses to these, and several other glycoprotein hormones, is not fully understood, although it is likely that ACTH is bifunctional, like PTH, supporting bone formation with periodic spikes of ACTH but causing bone destruction in large concentrations. In mice, mutations that reduce the efficiency of ACTH-induced glucocorticoid production in the adrenals cause the skeleton to become very dense (osteosclerotic bone).[12][13]
Organization and ultrastructure of osteoblasts
In well-preserved bone studied at high magnification with the electron microscope, the individual osteoblasts are shown to be connected by tight junctions, which prevent extracellular fluid from passing between osteoblasts and thus create a bone compartment separate from the general extracellular fluid.[14] The osteoblasts are also connected by gap junctions, very small pores that connect the individual osteoblasts, allowing the cells in one cohort of synthetic cells to function as a unit.[15] The gap junctions also connect deeper layers of cells, which when surrounded by bone are renamed osteocytes, to the surface layer. This was demonstrated directly by injecting low molecular weight fluorescent dyes into osteoblasts and showing that the dye diffused to surrounding and deeper cells in the bone-forming unit,[16] which is also called the osteon. Bone is composed of many of these units, which are separated by impermeable zones with no cellular connections, called cement lines.
Collagen and accessory proteins made by osteoblasts
Almost all of the organic (non-mineral) component of bone is very dense collagen type I,[17] which forms dense crosslinked ropes that give bone its tensile strength, keeping it from pulling apart. By mechanisms still unclear, the osteoblasts secrete layers of oriented collagen, with the layers parallel to the long axis of the bone alternating with layers at right angles to the long axis of the bone every few micrometers. Defects in collagen type I cause the commonest inherited disorder of bone, called osteogenesis imperfecta.[18]
Minor, but important, amounts of small proteins, including osteocalcin and osteopontin, are also secreted in the organic matrix of bone.[19] Osteocalcin is not expressed at significant concentrations except in bone, and thus osteocalcin is a specific marker for bone matrix synthesis.[20] These proteins have long been recognized to link organic and mineral component of bone matrix in ultrastructural studies.[21] However, in mice where expression of osteocalcin or osteopontin were eliminated by targeted disruption of the respective genes (knockout mice), accumulation of mineral was not notably affected, indicating that organization of matrix is not related, in any important way, to mineral transport,[22][23] although the proteins are necessary for maximal matrix strength due to their intermediate localization between mineral and collagen.
The relation of bone to its ancestral precursor, cartilage
The primitive skeleton is cartilage, a solid avascular (without blood vessels) tissue in which individual cartilage-matrix secreting cells, or chondrocytes, occur. Chondrocytes do not have intercellular connections and are not coordinated in units. Cartilage is composed of a network of collagen type II held in tension by water-absorbing proteins, hydrophilic proteoglycans.[24] This is the adult skeleton in cartilaginous fishes such as sharks, and it is retained as the initial skeleton in more advanced classes of animals.
In the air-breathing vertebrates, cartilage is replaced by advanced cellular bone. A transitional tissue is mineralized cartilage. Cartilage mineralizes by massive expression of phosphate-producing enzymes, which cause high local concentrations of calcium and phosphate that precipitate.[25] This mineralized cartilage is not very dense or very strong. In the air breathing vertebrates it is used as a scaffold for formation of cellular bone made by osteoblasts, and then it is removed by osteoclasts, which specialize in degrading mineralized tissue.
The osteoblasts of air-breathing vertebrates, in contrast, produce an advanced type of bone matrix consisting of extremely dense mineral, tiny irregular crystals of the mineral hydroxyapatite, packed around the collagen ropes.[26] This is an extremely strong composite material that allows the skeleton to be shaped mainly as hollow tubes. Reducing the long bones to tubular shells reduces the weight of the skeleton while maintaining strength.
Mineralization of bone
The mechanisms of mineralization are not fully understood. A key step in understanding the process was the discovery by Harold M. Frost in the late 1950s that fluorescent low-molecular weight compounds that bind strongly to bone mineral, such as tetracycline or calcein, when administered for short periods, accumulate in very narrow bands in the new bone.[27] These bands, which run from one side of the contiguous group of bone-forming osteoblasts to the other (see the diagram, Major features of the bone-forming complex), occur at a very narrow (sub-micrometer) mineralization front. On the other hand, at most of the bone surfaces, there is no new bone formation, no tetracycline uptake and no mineral formation at all. This strongly suggests that facilitated or active transport, coordinated across the bone-forming group, is involved in bone formation, and that only cell-mediated mineral formation occurs. That is, there is no uptake of mineral at all due to dietary calcium creating mineral by mass action, in contrast to a popular idea that eating more calcium than is required to maintain health results in more bone mineral.
The mechanism of mineral formation in bone is clearly distinct from the phylogenetically older process by which cartilage is mineralized: tetracycline does not label mineralized cartilage at narrow bands or in specific sites, but diffusely, in keeping with a passive mineralization mechanism.[28]
Since osteoblasts separate bone from the extracellular fluid by tight junctions,[29] it is not surprising that regulated transport is involved. Unlike cartilage, the phosphate and calcium cannot move in or out by passive diffusion, because the tight junctions between osteoblasts isolate the bone formation space. Calcium is transported across osteoblasts by facilitated transport (that is, by passive transporters, which do not pump calcium against a gradient).[30] In contrast, phosphate is actively produced by a combination of secretion of phosphate-containing compounds, including ATP, and by phosphatases that cleave off phosphate to create a high concentration of phosphate at the mineralization front; these include alkaline phosphatase, a membrane-anchored protein that is a characteristic marker that is expressed in large amounts at the apical (secretory) face of active osteoblasts.

At least one more regulated transport process is involved. The stoichiometry of bone mineral basically is that of hydroxyapatite precipitating from phosphate, calcium, and water at slightly alkaline pH. This was established by the mid 20th century:[31]
6 HPO42− + 2 H2O + 10 Ca2+ ⇌ Ca10(PO4)6(OH)2 + 8 H+
In a closed system, thus, as mineral precipitates acid would accumulate, rapidly lowering the pH and stopping further precipitation, unless the acid is removed. In cartilage, there is no barrier to diffusion and acid therefore diffuses away. But in the osteon, where matrix is separated from extracellular fluid by tight junctions, this cannot occur. On the other hand, in the controlled sealed compartment, removing H+ will drive complete mineral precipitation under a wide variety of extracellular conditions, as long as calcium and phosphate are available in the matrix compartment. This stoichiometry-driven reasoning was recognized in the 1950s and 1960s, with experimental evidence supporting an alkaline bone compartment published at that time.[32] On the other hand, the mechanism by which acid transits the barrier layer remains uncertain to this day. However, osteoblasts have massive capacity for Na+/H+ exchange, which has been known since the 1980s; it is now known to function with redundant Na/H exchangers, NHE1 and NHE6.[33] This H+ exchange is a major element in acid removal, although mechanism(s) by which H+ is transported from the matrix space into the barrier osteoblast is not known.
It is noteworthy that in bone removal, mediated by a specialized cell called the osteoclast, a reverse transport mechanism exists that uses acid delivered to the mineralized matrix to drive hydroxyapatite into solution.[34]
Feedback from osteocytes
Feedback from physical activity maintains bone mass, and feedback from osteocytes limits the size of the bone-forming unit. A number of mechanisms regulate bone density including stress on the bone.[35] An important additional mechanism is secretion by osteocytes, buried in the matrix, of sclerostin, an interesting protein that interferes with a pathway that maintains osteoblast activity. Thus, when the osteon reaches a limiting size, it self-inactivates the bone synthesis pathway.[36]
Morphology and histological staining
Hematoxylin and eosin, or H&E, staining, shows that the cytoplasm of active osteoblasts is slightly basophilic due to the presence of a large amount of rough endoplasmic reticulum. This reflects that the active osteoblast produces an enormous quantity of collagen type I, with about 10% of the bone matrix being collagen and the balance mineral.[37] The osteoblast's nucleus is spherical and large. An active osteoblast is also characterized morphologically by a prominent Golgi apparatus that appears histologically as a clear zone adjacent to the nucleus, reflecting that the products of the cell are mostly for transport into the osteoid, the non-mineralized matrix. Active osteoblasts synthesize, are easily labeled by antibodies to, Type-I collagen, and are often labeled using naphthol phosphate and the diazonium dye fast blue to demonstrate alkaline phosphatase enzyme activity directly. The alkaline phosphatase is almost entirely found on the apical (secretory) cell membrane.
-
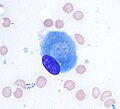
Osteoblast (Wright Giemsa stain, 100x)
-
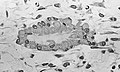
Light micrograph of decalcified cancellous bone displaying osteoblasts actively synthesizing osteoid, containing two osteocytes.
-
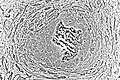
Light micrograph of osteoblasts actively synthesizing osteoid (center).
-
Light micrograph of osteoblasts actively synthesizing rudimentary bone tissue (center).
-
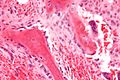
Osteoblasts lining bone (H&E stain).
See also
References
- ^ Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. (1999). "Multilineage potential of adult human mesenchymal stem cells". Science. 284: 143-7. doi:10.1126/science.284.5411.143 PMID 10102814.
- ^ Arnett T. (2003). Regulation of bone cell function by acid-base balance. Proc Nutr Soc. 62: 511-20. PMID 14506899.
- ^ Blair HC, Zaidi M, Huang CL, Sun L. (2008). The developmental basis of skeletal cell differentiation and the molecular basis of major skeletal defects. Biol Rev Camb Philos Soc. 83: 401-15. doi: 10.1111/j.1469-185X.2008.00048.x. PMID 18710437.
- ^ Nicks KM, Fowler TW, Gaddy D. (2010). "Reproductive hormones and bone." Curr Osteoporos Rep. 8: 60-7. doi: 10.1007/s11914-010-0014-3. PMID 20425612.
- ^ Karsenty G. (2008). "Transcriptional control of skeletogenesis". Annu Rev Genomics Hum Genet. 9: 183-96. doi: 10.1146/annurev.genom.9.081307.164437. PMID 18767962.
- ^ Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. (1999). "Multilineage potential of adult human mesenchymal stem cells". Science. 284: 143-7. doi:10.1126/science.284.5411.143 PMID 10102814.
- ^ Blair HC, Sun L, Kohanski RA. (2007). "Balanced regulation of proliferation, growth, differentiation, and degradation in skeletal cells". Ann N Y Acad Sci. 1116:165-73. PMID 17646258.
- ^ Blair HC, Zaidi M, Huang CL, Sun L. (2008). "The developmental basis of skeletal cell differentiation and the molecular basis of major skeletal defects". Biol Rev Camb Philos Soc. 83: 401-15. doi:10.1111/j.1469-185X.2008.00048.x PMID 18710437.
- ^ Reid IR, Bristow SM, Bolland MJ. (2015) Cardiovascular complications of calcium supplements. J Cell Biochem. 116: 494-501. doi:10.1002/jcb.25028 PubMed PMID 25491763.
- ^ Zaidi M, Sun L, Robinson LJ, Tourkova IL, Liu L, Wang Y, Zhu LL, Liu X, Li J, Peng Y, Yang G, Shi X, Levine A, Iqbal J, Yaroslavskiy BB, Isales C, Blair HC. ACTH protects against glucocorticoid-induced osteonecrosis of bone. Proc Natl Acad Sci U S A. 2010 107:8782-7. doi:10.1073/pnas.0912176107 PMID 20421485
- ^ Sun L, Peng Y, Sharrow AC, Iqbal J, Zhang Z, Papachristou DJ, Zaidi S, Zhu LL, Yaroslavskiy BB, Zhou H, Zallone A, Sairam MR, Kumar TR, Bo W, Braun J, Cardoso-Landa L, Schaffler MB, Moonga BS, Blair HC, Zaidi M. FSH directly regulates bone mass. Cell. 2006 125:247-60. PMID 16630814.
- ^ Hoekstra M, Meurs I, Koenders M, Out R, Hildebrand RB, Kruijt JK, Van Eck M, Van Berkel TJ. Absence of HDL cholesteryl ester uptake in mice via SR-BI impairs an adequate adrenal glucocorticoid-mediated stress response to fasting. J Lipid Res. 2008 Apr;49(4):738-45. doi: 10.1194/jlr.M700475-JLR200. PubMed PMID 18204096.
- ^ Martineau C, Martin-Falstrault L, Brissette L, Moreau R. The atherogenic Scarb1 null mouse model shows a high bone mass phenotype. Am J Physiol Endocrinol Metab. 2013 Nov 19. [Epub ahead of print] PubMed PMID 24253048.
- ^ Arana-Chavez VE, Soares AM, Katchburian E. (1995) "Junctions between early developing osteoblasts of rat calvaria as revealed by freeze-fracture and ultrathin ya electron microscopy." Arch Histol Cytol. 1995 58:285-92. PMID 8527235.
- ^ Doty SB. (1981) "Morphological evidence of gap junctions between bone cells." Calcif Tissue Int. 33: 509-12. PMID 6797704.
- ^ Yellowley CE, Li Z, Zhou Z, Jacobs CR, Donahue HJ. (2000) "Functional gap junctions between osteocytic and osteoblastic cells." J Bone Miner Res. 15:209-17. PMID 10703922.
- ^ Reddi AH, Gay R, Gay S, Miller EJ. (1977) "Transitions in collagen types during matrix-induced cartilage, bone, and bone marrow formation". Proc Natl Acad Sci U S A. 74:5589-92. PMID 271986
- ^ Kuivaniemi H, Tromp G, Prockop DJ. (1991) "Mutations in collagen genes: causes of rare and some common diseases in humans". FASEB J. 5: 2052-60. PMID 2010058.
- ^ Aubin JE, Liu F, Malaval L, Gupta AK. (1995) Osteoblast and chondroblast differentiation. Bone. 17(2 Suppl): 77S-83S. PubMed PMID 8579903.
- ^ Delmas PD, Demiaux B, Malaval L, Chapuy MC, Meunier PJ. (1986) "Osteocalcin (or bone gla-protein), a new biological marker for studying bone pathology". Presse Med. 15: 643-6. PMID 2939433.
- ^ Roach HI. (1994) Why does bone matrix contain non-collagenous proteins? The possible roles of osteocalcin, osteonectin, osteopontin and bone sialoprotein in bone mineralisation and resorption. Cell Biol Int. 18: 617-28. PMID 8075622.
- ^ Boskey AL, Gadaleta S, Gundberg C, Doty SB, Ducy P, Karsenty G. (1998) "Fourier transform infrared microspectroscopic analysis of bones of osteocalcin-deficient mice provides insight into the function of osteocalcin". Bone. 23: 187-96. PMID 9737340.
- ^ Thurner PJ, Chen CG, Ionova-Martin S, Sun L, Harman A, Porter A, Ager JW 3rd, Ritchie RO, Alliston T. (2010) "Osteopontin deficiency increases bone fragility but preserves bone mass". Bone. 46: 1564-73. doi:10.1016/j.bone.2010.02.014 PMID 20171304.
- ^ Blair HC, Zaidi M, Schlesinger PH. (2002) "Mechanisms balancing skeletal matrix synthesis and degradation". Biochem J. 364: 329-41. PMID 12023876.
- ^ Blair HC, Zaidi M, Schlesinger PH. (2002) "Mechanisms balancing skeletal matrix synthesis and degradation". Biochem J. 364: 329-41. PMID 12023876.
- ^ Blair HC, Robinson LJ, Huang CL, Sun L, Friedman PA, Schlesinger PH, Zaidi M. (2011) "Calcium and bone disease". Biofactors. 37:159-67. doi: 10.1002/biof.143. PMID 21674636.
- ^ Frost HM. (1969). Tetracycline-based histological analysis of bone remodeling. Calcif Tissue Res. 3: 211-37. PMID 4894738.
- ^ Blair HC, Robinson LJ, Huang CL, Sun L, Friedman PA, Schlesinger PH, Zaidi M. (2011) "Calcium and bone disease". Biofactors. 37:159-67. doi: 10.1002/biof.143. PMID 21674636.
- ^ Arana-Chavez VE, Soares AM, Katchburian E. (1995) "Junctions between early developing osteoblasts of rat calvaria as revealed by freeze-fracture and ultrathin section electron microscopy." Arch Histol Cytol. 1995 58:285-92. PMID 8527235.
- ^ Blair HC, Robinson LJ, Huang CL, Sun L, Friedman PA, Schlesinger PH, Zaidi M. (2011) "Calcium and bone disease". Biofactors. 37:159-67. doi: 10.1002/biof.143. PMID 21674636.
- ^ Neuman, William F.; Neuman, Margaret W. (1958). The Chemical Dynamics of Bone Mineral. Chicago: The University of Chicago Press. ISBN 0-226-57512-8.
- ^ Schartum, S, Nichols G Jr. (1962). Concerning pH gradients between the extracellular compartment and fluids bathing the bone mineral surface and their relation to calcium ion distribution. J Clin Invest. 41:1163-8. PMID 14498063.
- ^ Liu L, Schlesinger PH, Slack NM, Friedman PA, Blair HC. High capacity Na+/H+ exchange activity in mineralizing osteoblasts. J Cell Physiol. 2011 Jun;226(6):1702-12. doi: 10.1002/jcp.22501.
- ^ Blair HC, Teitelbaum SL, Ghiselli R, Gluck S. Osteoclastic bone resorption by a polarized vacuolar proton pump. Science. 1989 245(4920): 855-7 .PMID 2528207.
- ^ Klein-Nulend J, Nijweide PJ, Burger EH. (2003). Osteocyte and bone structure. Curr Osteoporos Rep. 1: 5-10. PMID 16036059.
- ^ Baron R, Rawadi G, Roman-Roman S. (2006). Wnt signaling: a key regulator of bone mass. Curr Top Dev Biol. 76: 103-27. PMID 17118265.
- ^ Neuman, William F.; Neuman, Margaret W. (1958). The Chemical Dynamics of Bone Mineral. Chicago: The University of Chicago Press. ISBN 0-226-57512-8.
Further reading
- William F. Neuman and Margaret W. Neuman. (1958). The Chemical Dynamics of Bone Mineral. Chicago: The University of Chicago Press. ISBN 0-226-57512-8.
- Netter, Frank H. (1987). Musculoskeletal system: anatomy, physiology, and metabolic disorders. Summit, New Jersey: Ciba-Geigy Corporation ISBN 0-914168-88-6.