Mesenchymal stem cell
| Mesenchymal stem cell | |
|---|---|
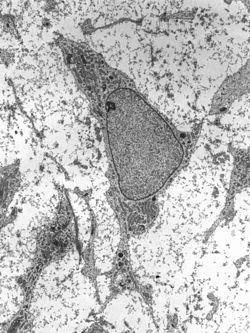 Transmission electron micrograph of a mesenchymal stem cell displaying typical ultrastructural characteristics | |
| Details | |
| Identifiers | |
| Latin | cellula mesenchymatica praecursoria |
| MeSH | D059630 |
| TH | H2.00.01.0.00008 |
| Anatomical terms of microanatomy | |
Mesenchymal stem cells (MSCs), also known as mesenchymal stromal cells or medicinal signaling cells, are multipotent stromal cells that can differentiate into a variety of cell types, including osteoblasts (bone cells), chondrocytes (cartilage cells), myocytes (muscle cells) and adipocytes (fat cells which give rise to marrow adipose tissue).[1][2][3][4]
Structure
[edit]Definition
[edit]Mesenchymal stem cells (MSCs), a term first used (in 1991) by Arnold Caplan at Case Western Reserve University,[5] are characterized morphologically by a small cell body with long, thin cell processes. While the terms mesenchymal stem cell (MSC) and marrow stromal cell have been used interchangeably for many years, neither term is sufficiently descriptive:
- Mesenchyme is embryonic connective tissue that is derived from the mesoderm and that differentiates into hematopoietic and connective tissue, whereas MSCs do not differentiate into hematopoietic cells.[6]
- Stromal cells are connective tissue cells that form the supportive structure in which the functional cells of the tissue reside. While this is an accurate description for one function of MSCs, the term fails to convey the relatively recently discovered roles of MSCs in the repair of tissue.[7]
- The term encompasses multipotent cells derived from other non-marrow tissues, such as placenta,[8] umbilical cord blood, adipose tissue, adult muscle, corneal stroma,[9] or the dental pulp of deciduous (baby) teeth.[10] The cells do not have the capacity to reconstitute an entire organ.
- Evidence shows that MSCs originate from progenitor perivascular cells called pericytes that reside on vessels.[11]
Morphology
[edit]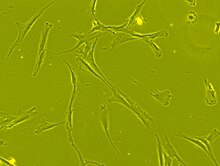

The cell body contains a large, round nucleus with a prominent nucleolus, which is surrounded by finely dispersed chromatin particles, giving the nucleus a clear appearance. The remainder of the cell body contains a small amount of Golgi apparatus, rough endoplasmic reticulum, mitochondria, and polyribosomes. The cells, which are long and thin, are widely dispersed, and the adjacent extracellular matrix is populated by a few reticular fibrils, but is devoid of the other types of collagen fibrils.[12][13] These distinctive morphological features of mesenchymal stem cells can be visualized label-free using live cell imaging.
Classification
[edit]The International Society for Cellular Therapy (ISCT) has proposed a set of standards to define MSCs. A cell can be classified as an MSC if it shows plastic adherent properties under normal culture conditions and has a fibroblast-like morphology. In fact, some argue that MSCs and fibroblasts are functionally identical.[14] The study in Science, "Multilineage Potential of Adult Mesenchymal Stem Cells," describes how MSCs can undergo osteogenic, adipogenic and chondrogenic differentiation ex vivo. As of November 2023, this paper has been cited over 29,000 times. Cultured MSCs also express on their surface CD73, CD90 and CD105, while lacking the expression of CD11b, CD14, CD19, CD34, CD45, CD79a and HLA-DR surface markers.[15]
Location in the body
[edit]MSCs are found throughout the human body.
Bone marrow
[edit]Bone marrow was the original source of MSCs,[16] and is still the most frequently utilized source. These bone marrow stem cells do not contribute to the formation of blood cells, and so do not express the hematopoietic stem cell marker CD34. They are sometimes referred to as bone marrow stromal stem cells.[17]
Cord cells
[edit]The youngest and most primitive MSCs may be obtained from umbilical cord tissue, namely Wharton's jelly and the umbilical cord blood. However, MSCs are found in much higher concentration in the Wharton's jelly compared to cord blood, which is a rich source of hematopoietic stem cells. The umbilical cord is available after a birth. It is normally discarded, and poses no risk for collection. These MSCs may prove to be a useful source of MSCs for clinical applications, due to their primitive properties and fast growth rate.[18]
Adipose tissue
[edit]Adipose-tissue-derived MSCs (AdMSCs), in addition to being easier and safer to isolate than bone-marrow-derived MSCs, can be obtained in larger quantities.[16][19]
Molar cells
[edit]The developing tooth bud of the mandibular third molar is a rich source of MSCs. While they are described as multipotent, it is possible that they are pluripotent. They eventually form enamel, dentin, blood vessels, dental pulp, and nervous tissues. These stem cells are capable of differentiating into chondrocytes, cardiomyocytes, melanocytes, and hepatocyte‐like cells in vitro.[10]
Amniotic fluid
[edit]Stem cells are present in amniotic fluid. As many as 1 in 100 cells collected during amniocentesis are pluripotent mesenchymal stem cells.[20]
Function
[edit]Differentiation capacity
[edit]MSCs have a great capacity for self-renewal while maintaining their multipotency. Recent work suggests that β-catenin, via regulation of EZH2, is a central molecule in maintaining the "stemness" of MSCs.[21] The standard test to confirm multipotency is differentiation of the cells into osteoblasts, adipocytes and chondrocytes as well as myocytes.
MSCs have been seen to even differentiate into neuron-like cells,[22] but doubt remains about whether the MSC-derived neurons are functional.[23] The degree to which the culture will differentiate varies among individuals and how differentiation is induced, e.g., chemical vs. mechanical;[24] and it is not clear whether this variation is due to a different amount of "true" progenitor cells in the culture or variable differentiation capacities of individuals' progenitors. The capacity of cells to proliferate and differentiate is known to decrease with the age of the donor, as well as the time in culture.[25] Likewise, whether this is due to a decrease in the number of MSCs or a change to the existing MSCs is not known.[citation needed]
Immunomodulatory effects
[edit]MSCs have an effect on innate and specific immune cells, and research has shown an ability to suppress tumor growth.[26] MSCs produce many immunomodulatory molecules including prostaglandin E2 (PGE2),[27] nitric oxide,[28] indoleamine 2,3-dioxygenase (IDO), interleukin 6 (IL-6), and other surface markers such as FasL,[29] PD-L1 and PD-L2.[30]
MSCs have an effect on macrophages, neutrophils, NK cells, mast cells and dendritic cells in innate immunity. MSCs are able to migrate to the site of injury, where they polarize through PGE2 macrophages in M2 phenotype which is characterized by an anti-inflammatory effect.[31] Further, PGE2 inhibits the ability of mast cells to degranulate and produce TNF-α.[32][33] Proliferation and cytotoxic activity of NK cells is inhibited by PGE2 and IDO. MSCs also reduce the expression of NK cell receptors - NKG2D, NKp44 and NKp30.[34] MSCs inhibit respiratory flare and apoptosis of neutrophils by production of cytokines IL-6 and IL-8.[35] Differentiation and expression of dendritic cell surface markers is inhibited by IL-6 and PGE2 of MSCs.[36] The immunosuppressive effects of MSC also depend on IL-10, but it is not certain whether they produce it alone, or only stimulate other cells to produce it.[37]
MSC expresses the adhesion molecules VCAM-1 and ICAM-1, which allow T-lymphocytes to adhere to their surface. Then MSC can affect them by molecules which have a short half-life and their effect is in the immediate vicinity of the cell.[28] These include nitric oxide,[38] PGE2, HGF,[39] and activation of receptor PD-1.[40] MSCs reduce T cell proliferation between G0 and G1 cell cycle phases[41] and decrease the expression of IFNγ of Th1 cells while increasing the expression of IL-4 of Th2 cells.[42] MSCs also inhibit the proliferation of B-lymphocytes between G0 and G1 cell cycle phases.[40][43]
Antimicrobial properties
[edit]MSCs produce several antimicrobial peptides (AMPs), including human cathelicidin LL-37,[44] β-defensins,[45] lipocalin 2[46] and hepcidin.[47] These peptides, together with the enzyme indoleamine 2,3-dioxygenase (IDO), are responsible for the broad-spectrum antibacterial activity of MSCs.[48]
Clinical significance
[edit]
Mesenchymal stem cells can be activated and mobilized in reaction to injury and infection. As of May 2024, a search for "mesenchymal stem cells" or "mesenchymal stromal cells" at ClinicalTrials.gov returns more than 1,760 studies featuring MSCs[49] for more than 920 conditions.
Autoimmune disease
[edit]Clinical studies investigating the efficacy of mesenchymal stem cells in treating diseases are in clinical development around the world, particularly treating autoimmune diseases, graft versus host disease, Crohn's disease, multiple sclerosis, systemic lupus erythematosus and systemic sclerosis.[50][51]
Other diseases
[edit]Many of the early clinical successes using intravenous transplantation came in systemic diseases such as graft versus host disease and sepsis. Direct injection or placement of cells into a site in need of repair may be the preferred method of treatment, as vascular delivery suffers from a "pulmonary first pass effect" where intravenous injected cells are sequestered in the lungs.[52]
Further studies into the mechanisms of MSC action may provide avenues for increasing their capacity for tissue repair.[53][54]
Research
[edit]The majority of modern culture techniques still take a colony-forming unit-fibroblasts (CFU-F) approach, where raw unpurified bone marrow or ficoll-purified bone marrow mononuclear cells are plated directly into cell culture plates or flasks. Mesenchymal stem cells, but not red blood cells or hematopoietic progenitors, are adherent to tissue culture plastic within 24 to 48 hours. However, at least one publication has identified a population of non-adherent MSCs that are not obtained by the direct-plating technique.[55]
Other flow cytometry-based methods allow the sorting of bone marrow cells for specific surface markers, such as STRO-1.[56] STRO-1+ cells are generally more homogenous and have higher rates of adherence and higher rates of proliferation, but the exact differences between STRO-1+ cells and MSCs are not clear.[57]
Methods of immunodepletion using such techniques as MACS have also been used in the negative selection of MSCs.[58]
The supplementation of basal media with fetal bovine serum or human platelet lysate is common in MSC culture. Prior to the use of platelet lysates for MSC culture, the pathogen inactivation process is recommended to prevent pathogen transmission.[59]
New research titled Transplantation of human ESC-derived mesenchymal stem cell spheroids ameliorates spontaneous osteoarthritis in rhesus macaques[60] Various chemicals and methods, including low-level laser irradiation, have been used to increase stem cell proliferation.[61]
History
[edit]Scientists Ernest A. McCulloch and James E. Till first revealed the clonal nature of marrow cells in the 1960s.[62][63] In 1970, Arnold Caplan identified certain conditions by which mesodermal cells differentiate into cartilage or myogenic (muscle) tissue and bone.[64]
An ex vivo assay for examining the clonogenic potential of multipotent marrow cells was later reported in the 1970s by Friedenstein and colleagues.[65][66] In this assay system, stromal cells were referred to as colony-forming unit-fibroblasts (CFU-f).
Subsequent experimentation revealed the plasticity of marrow cells and how their fate is determined by environmental cues. Culturing marrow stromal cells in the presence of osteogenic stimuli such as ascorbic acid, inorganic phosphate and dexamethasone could promote their differentiation into osteoblasts. In contrast, the addition of transforming growth factor-beta (TGF-b) could induce chondrogenic markers.[citation needed]
The first clinical trials of MSCs were completed by Osiris Therapeutics in 1995 when a group of 15 patients were injected with cultured MSCs to test the safety of the treatment.[67] The first regulatory approvals for MSCs were granted conditional approval in 2012 in Canada and New Zealand for treating Graft vs. Host Disease (GvHD) and, subsequently, in Japan to treat Crohn’s Disease-related fistula.[68]
Since then, more than 1,500 clinical trials have been conducted to treat numerous conditions.[69]
Controversies
[edit]The term "mesenchymal stem cells" and what constitutes the most scientifically correct meaning for the MSC initialism, has been debated for years. Most mesenchymal cell or "MSC" preps only contain a minority fraction of true multipotent stem cells, with most cells being stromal in nature. Caplan proposed rephrasing MSCs to emphasize their role as "medicinal signaling cells."[70] Within the stem cell field, the shorthand "MSC" has most commonly now come to refer to "mesenchymal stromal/stem cells" because of the heterogeneous nature of the cellular preparations.
There is also growing concern about the marketing and application of unapproved MSCs and mesenchymal stem cells that lack rigorous data to back up these clinical uses into patients by for-profit clinics.[71][72]
See also
[edit]- Bone marrow
- Fibroblast
- Muse cell
- Intramembranous ossification
- Mesenchyme
- Multipotency
- Cord lining
- Bone marrow adipose tissue
- List of human cell types derived from the germ layers
References
[edit]- ^ Tonk CH, Witzler M, Schulze M, Tobiasch E (2020). "Mesenchymal Stem Cells". In Brand-Saberi B (ed.). Essential Current Concepts in Stem Cell Biology. Learning Materials in Biosciences. Cham: Springer International Publishing. pp. 21–39. doi:10.1007/978-3-030-33923-4_2. ISBN 978-3-030-33923-4. S2CID 214523766.
- ^ Ankrum JA, Ong JF, Karp JM (March 2014). "Mesenchymal stem cells: immune evasive, not immune privileged". Nature Biotechnology. 32 (3): 252–60. doi:10.1038/nbt.2816. PMC 4320647. PMID 24561556.
- ^ Mahla RS (2016). "Stem Cells Applications in Regenerative Medicine and Disease Therapeutics". International Journal of Cell Biology. 2016: 6940283. doi:10.1155/2016/6940283. PMC 4969512. PMID 27516776.
- ^ Caplan AI (June 2017). "Mesenchymal Stem Cells: Time to Change the Name!". Stem Cells Translational Medicine. 6 (6): 1445–1451. doi:10.1002/sctm.17-0051. PMC 5689741. PMID 28452204.
- ^ Bianco P, Robey PG, Simmons PJ (April 2008). "Mesenchymal stem cells: revisiting history, concepts, and assays". Cell Stem Cell. 2 (4): 313–319. doi:10.1016/j.stem.2008.03.002. PMC 2613570. PMID 18397751.
- ^ Porcellini A (2009). "Regenerative medicine: a review". Revista Brasileira de Hematologia e Hemoterapia. 31 (Suppl. 2). doi:10.1590/S1516-84842009000800017.
- ^ Valero MC, Huntsman HD, Liu J, Zou K, Boppart MD (2012). "Eccentric exercise facilitates mesenchymal stem cell appearance in skeletal muscle". PLOS ONE. 7 (1): e29760. Bibcode:2012PLoSO...729760V. doi:10.1371/journal.pone.0029760. PMC 3256189. PMID 22253772.
- ^ Wang S, Qu X, Zhao RC (April 2012). "Clinical applications of mesenchymal stem cells". Journal of Hematology & Oncology. 5: 19. doi:10.1186/1756-8722-5-19. PMC 3416655. PMID 22546280.
- ^ Branch MJ, Hashmani K, Dhillon P, Jones DR, Dua HS, Hopkinson A (August 2012). "Mesenchymal stem cells in the human corneal limbal stroma". Investigative Ophthalmology & Visual Science. 53 (9): 5109–16. doi:10.1167/iovs.11-8673. PMID 22736610.
- ^ a b Liu J, Yu F, Sun Y, Jiang B, Zhang W, Yang J, Xu G, Liang A, Liu S (March 2015). "Concise Reviews: Characteristics and Potential Applications of Human Dental Tissue-Derived Mesenchymal Stem Cells". Stem Cells. 33 (3): 627–38. doi:10.1002/stem.1909. PMID 25447379.
- ^ Crisan M, Yap S, Casteilla L, Chen CW, Corselli M, Park TS, Andriolo G, Sun B, Zheng B, Zhang L, Norotte C, Teng PN, Traas J, Schugar R, Deasy BM, Badylak S, Buhring HJ, Giacobino JP, Lazzari L, Huard J, Péault B (September 2008). "A perivascular origin for mesenchymal stem cells in multiple human organs". Cell Stem Cell. 3 (3): 301–13. doi:10.1016/j.stem.2008.07.003. PMID 18786417.
- ^ Netter FH (1987). Musculoskeletal system: anatomy, physiology, and metabolic disorders. Summit, New Jersey: Ciba-Geigy Corporation. p. 134. ISBN 978-0-914168-88-1.
- ^ Brighton CT, Hunt RM (July 1991). "Early histological and ultrastructural changes in medullary fracture callus". The Journal of Bone and Joint Surgery. American Volume. 73 (6): 832–847. doi:10.2106/00004623-199173060-00006. PMID 2071617.
- ^ Hematti P (May 2012). "Mesenchymal stromal cells and fibroblasts: a case of mistaken identity?". Cytotherapy. 14 (5): 516–21. doi:10.3109/14653249.2012.677822. PMID 22458957.
- ^ Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, et al. (1 January 2006). "Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement". Cytotherapy. 8 (4): 315–7. doi:10.1080/14653240600855905. PMID 16923606.
- ^ a b Strioga M, Viswanathan S, Darinskas A, Slaby O, Michalek J (September 2012). "Same or Not the Same? Comparison of Adipose Tissue-Derived Versus Bone Marrow-Derived Mesenchymal Stem and Stromal Cells". Stem Cells and Development. 21 (14): 2724–52. doi:10.1089/scd.2011.0722. PMID 22468918.
- ^ Gregory CA, Prockop DJ, Spees JL (June 2005). "Non-hematopoietic bone marrow stem cells: molecular control of expansion and differentiation". Experimental Cell Research. Molecular Control of Stem Cell Differentiation. 306 (2): 330–5. doi:10.1016/j.yexcr.2005.03.018. PMID 15925588.
- ^ Liau LL, Ruszymah BH, Ng MH, Law JX (January 2020). "Characteristics and Clinical Applications of Wharton's Jelly-Derived Mesenchymal Stromal Cells". Current Research in Translational Medicine. 68 (1): 5–16. doi:10.1016/j.retram.2019.09.001. PMID 31543433. S2CID 202731274.
- ^ Bunnell BA, Flaat M, Gagliardi C, Patel B, Ripoll C (June 2008). "Adipose-derived stem cells: isolation, expansion and differentiation". Methods. Methods in stem cell research. 45 (2): 115–20. doi:10.1016/j.ymeth.2008.03.006. PMC 3668445. PMID 18593609.
- ^ "What is Cord Tissue?". CordAdvantage.com. 30 October 2018.
- ^ Sen B, Paradise CR, Xie Z, Sankaran J, Uzer G, Styner M, et al. (June 2020). "β-Catenin Preserves the Stem State of Murine Bone Marrow Stromal Cells Through Activation of EZH2". Journal of Bone and Mineral Research. 35 (6): 1149–1162. doi:10.1002/jbmr.3975. PMC 7295671. PMID 32022326.
- ^ Jiang Y, Jahagirdar BN, Reinhardt RL, Schwartz RE, Keene CD, Ortiz-Gonzalez XR, et al. (July 2002). "Pluripotency of mesenchymal stem cells derived from adult marrow". Nature. 418 (6893): 41–9. Bibcode:2002Natur.418...41J. doi:10.1038/nature00870. PMID 12077603. S2CID 47162269. (Retracted, see doi:10.1038/s41586-024-07653-0, PMID 38886620, Retraction Watch)
- ^ Franco Lambert AP, Fraga Zandonai A, Bonatto D, Cantarelli Machado D, Pêgas Henriques JA (March 2009). "Differentiation of human adipose-derived adult stem cells into neuronal tissue: does it work?". Differentiation; Research in Biological Diversity. 77 (3): 221–8. doi:10.1016/j.diff.2008.10.016. PMID 19272520.
- ^ Engler AJ, Sen S, Sweeney HL, Discher DE (August 2006). "Matrix elasticity directs stem cell lineage specification". Cell. 126 (4): 677–89. doi:10.1016/j.cell.2006.06.044. PMID 16923388. S2CID 16109483.
- ^ Choudhery MS, Badowski M, Muise A, Pierce J, Harris DT (January 2014). "Donor age negatively impacts adipose tissue-derived mesenchymal stem cell expansion and differentiation". Journal of Translational Medicine. 12 (1): 8. doi:10.1186/1479-5876-12-8. PMC 3895760. PMID 24397850.
- ^ Banerjee K, Chattopadhyay A, Banerjee S (July 2022). "Understanding the association of stem cells in fetal development and carcinogenesis during pregnancy". Advances in Cancer Biology - Metastasis. 4: 100042. doi:10.1016/j.adcanc.2022.100042. ISSN 2667-3940. S2CID 248485831.
- ^ Spaggiari GM, Abdelrazik H, Becchetti F, Moretta L (June 2009). "MSCs inhibit monocyte-derived DC maturation and function by selectively interfering with the generation of immature DCs: central role of MSC-derived prostaglandin E2". Blood. 113 (26): 6576–6583. doi:10.1182/blood-2009-02-203943. PMID 19398717. S2CID 206878089.
- ^ a b Ren G, Zhao X, Zhang L, Zhang J, L'Huillier A, Ling W, et al. (March 2010). "Inflammatory cytokine-induced intercellular adhesion molecule-1 and vascular cell adhesion molecule-1 in mesenchymal stem cells are critical for immunosuppression". Journal of Immunology. 184 (5): 2321–2328. doi:10.4049/jimmunol.0902023. PMC 2881946. PMID 20130212.
- ^ Akiyama K, Chen C, Wang D, Xu X, Qu C, Yamaza T, et al. (May 2012). "Mesenchymal-stem-cell-induced immunoregulation involves FAS-ligand-/FAS-mediated T cell apoptosis". Cell Stem Cell. 10 (5): 544–555. doi:10.1016/j.stem.2012.03.007. PMC 3348385. PMID 22542159.
- ^ Davies LC, Heldring N, Kadri N, Le Blanc K (March 2017). "Mesenchymal Stromal Cell Secretion of Programmed Death-1 Ligands Regulates T Cell Mediated Immunosuppression". Stem Cells. 35 (3): 766–776. doi:10.1002/stem.2509. PMC 5599995. PMID 27671847.
- ^ Kim J, Hematti P (December 2009). "Mesenchymal stem cell-educated macrophages: a novel type of alternatively activated macrophages". Experimental Hematology. 37 (12): 1445–53. doi:10.1016/j.exphem.2009.09.004. PMC 2783735. PMID 19772890.
- ^ Brown JM, Nemeth K, Kushnir-Sukhov NM, Metcalfe DD, Mezey E (April 2011). "Bone marrow stromal cells inhibit mast cell function via a COX2-dependent mechanism". Clinical and Experimental Allergy. 41 (4): 526–34. doi:10.1111/j.1365-2222.2010.03685.x. PMC 3078050. PMID 21255158.
- ^ Kay LJ, Yeo WW, Peachell PT (April 2006). "Prostaglandin E2 activates EP2 receptors to inhibit human lung mast cell degranulation". British Journal of Pharmacology. 147 (7): 707–13. doi:10.1038/sj.bjp.0706664. PMC 1751511. PMID 16432506.
- ^ Spaggiari GM, Capobianco A, Becchetti S, Mingari MC, Moretta L (February 2006). "Mesenchymal stem cell-natural killer cell interactions: evidence that activated NK cells are capable of killing MSCs, whereas MSCs can inhibit IL-2-induced NK-cell proliferation". Blood. 107 (4): 1484–90. doi:10.1182/blood-2005-07-2775. hdl:11567/267587. PMID 16239427.
- ^ Raffaghello L, Bianchi G, Bertolotto M, Montecucco F, Busca A, Dallegri F, et al. (January 2008). "Human mesenchymal stem cells inhibit neutrophil apoptosis: a model for neutrophil preservation in the bone marrow niche". Stem Cells. 26 (1): 151–62. doi:10.1634/stemcells.2007-0416. PMID 17932421. S2CID 32230553.
- ^ Jiang XX, Zhang Y, Liu B, Zhang SX, Wu Y, Yu XD, Mao N (May 2005). "Human mesenchymal stem cells inhibit differentiation and function of monocyte-derived dendritic cells". Blood. 105 (10): 4120–6. doi:10.1182/blood-2004-02-0586. PMID 15692068.
- ^ Ma S, Xie N, Li W, Yuan B, Shi Y, Wang Y (February 2014). "Immunobiology of mesenchymal stem cells". Cell Death and Differentiation. 21 (2): 216–25. doi:10.1038/cdd.2013.158. PMC 3890955. PMID 24185619.
- ^ Ren G, Zhang L, Zhao X, Xu G, Zhang Y, Roberts AI, et al. (February 2008). "Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide". Cell Stem Cell. 2 (2): 141–50. doi:10.1016/j.stem.2007.11.014. PMID 18371435.
- ^ Di Nicola M, Carlo-Stella C, Magni M, Milanesi M, Longoni PD, Matteucci P, et al. (May 2002). "Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli". Blood. 99 (10): 3838–43. doi:10.1182/blood.v99.10.3838. hdl:11379/598065. PMID 11986244. S2CID 5889200.
- ^ a b Augello A, Tasso R, Negrini SM, Amateis A, Indiveri F, Cancedda R, Pennesi G (May 2005). "Bone marrow mesenchymal progenitor cells inhibit lymphocyte proliferation by activation of the programmed death 1 pathway". European Journal of Immunology. 35 (5): 1482–90. doi:10.1002/eji.200425405. PMID 15827960. S2CID 24088675.
- ^ Glennie S, Soeiro I, Dyson PJ, Lam EW, Dazzi F (April 2005). "Bone marrow mesenchymal stem cells induce division arrest anergy of activated T cells". Blood. 105 (7): 2821–7. doi:10.1182/blood-2004-09-3696. PMID 15591115. S2CID 33590543.
- ^ Aggarwal S, Pittenger MF (February 2005). "Human mesenchymal stem cells modulate allogeneic immune cell responses". Blood. 105 (4): 1815–22. doi:10.1182/blood-2004-04-1559. PMID 15494428.
- ^ Corcione A, Benvenuto F, Ferretti E, Giunti D, Cappiello V, Cazzanti F, et al. (January 2006). "Human mesenchymal stem cells modulate B-cell functions". Blood. 107 (1): 367–72. doi:10.1182/blood-2005-07-2657. PMID 16141348.
- ^ Krasnodembskaya A, Song Y, Fang X, Gupta N, Serikov V, Lee JW, Matthay MA (December 2010). "Antibacterial effect of human mesenchymal stem cells is mediated in part from secretion of the antimicrobial peptide LL-37". Stem Cells. 28 (12): 2229–38. doi:10.1002/stem.544. PMC 3293245. PMID 20945332.
- ^ Sung DK, Chang YS, Sung SI, Yoo HS, Ahn SY, Park WS (March 2016). "Antibacterial effect of mesenchymal stem cells against Escherichia coli is mediated by secretion of beta- defensin- 2 via toll- like receptor 4 signalling". Cellular Microbiology. 18 (3): 424–36. doi:10.1111/cmi.12522. PMC 5057339. PMID 26350435.
- ^ Gupta N, Krasnodembskaya A, Kapetanaki M, Mouded M, Tan X, Serikov V, Matthay MA (June 2012). "Mesenchymal stem cells enhance survival and bacterial clearance in murine Escherichia coli pneumonia". Thorax. 67 (6): 533–9. doi:10.1136/thoraxjnl-2011-201176. PMC 3358432. PMID 22250097.
- ^ Alcayaga-Miranda F, Cuenca J, Martin A, Contreras L, Figueroa FE, Khoury M (October 2015). "Combination therapy of menstrual derived mesenchymal stem cells and antibiotics ameliorates survival in sepsis". Stem Cell Research & Therapy. 6: 199. doi:10.1186/s13287-015-0192-0. PMC 4609164. PMID 26474552.
- ^ Meisel R, Brockers S, Heseler K, Degistirici O, Bülle H, Woite C, et al. (April 2011). "Human but not murine multipotent mesenchymal stromal cells exhibit broad-spectrum antimicrobial effector function mediated by indoleamine 2,3-dioxygenase". Leukemia. 25 (4): 648–54. doi:10.1038/leu.2010.310. PMID 21242993.
- ^ "Search of: Mesenchymal stem cells - List Results - ClinicalTrials.gov". clinicaltrials.gov. Retrieved 26 May 2023.
- ^ Figueroa FE, Carrión F, Villanueva S, Khoury M (2012). "Mesenchymal stem cell treatment for autoimmune diseases: a critical review". Biological Research. 45 (3): 269–77. doi:10.4067/S0716-97602012000300008. PMID 23283436.
- ^ Sharma RR, Pollock K, Hubel A, McKenna D (May 2014). "Mesenchymal stem or stromal cells: a review of clinical applications and manufacturing practices". Transfusion. 54 (5): 1418–37. doi:10.1111/trf.12421. PMC 6364749. PMID 24898458.
- ^ Fischer UM, Harting MT, Jimenez F, Monzon-Posadas WO, Xue H, Savitz SI, et al. (June 2009). "Pulmonary passage is a major obstacle for intravenous stem cell delivery: the pulmonary first-pass effect". Stem Cells and Development. 18 (5): 683–92. doi:10.1089/scd.2008.0253. PMC 3190292. PMID 19099374.
- ^ Heirani-Tabasi A, Hassanzadeh M, Hemmati-Sadeghi S, Shahriyari M, Raeesolmohaddeseen M (2015). "Mesenchymal Stem Cells; Defining the Future of Regenerative Medicine". Journal of Genes and Cells. 1 (2): 34–39. doi:10.15562/gnc.15. S2CID 87157970.
- ^ Anderson JD, Johansson HJ, Graham CS, Vesterlund M, Pham MT, Bramlett CS, et al. (March 2016). "Comprehensive Proteomic Analysis of Mesenchymal Stem Cell Exosomes Reveals Modulation of Angiogenesis via Nuclear Factor-KappaB Signaling". Stem Cells. 34 (3): 601–13. doi:10.1002/stem.2298. PMC 5785927. PMID 26782178.
- ^ Wan C, He Q, McCaigue M, Marsh D, Li G (January 2006). "Nonadherent cell population of human marrow culture is a complementary source of mesenchymal stem cells (MSCs)". Journal of Orthopaedic Research. 24 (1): 21–8. doi:10.1002/jor.20023. PMID 16419965. S2CID 28963721.
- ^ Gronthos S, Graves SE, Ohta S, Simmons PJ (December 1994). "The STRO-1+ fraction of adult human bone marrow contains the osteogenic precursors". Blood. 84 (12): 4164–73. doi:10.1182/blood.V84.12.4164.bloodjournal84124164. PMID 7994030.
- ^ Oyajobi BO, Lomri A, Hott M, Marie PJ (March 1999). "Isolation and characterization of human clonogenic osteoblast progenitors immunoselected from fetal bone marrow stroma using STRO-1 monoclonal antibody". Journal of Bone and Mineral Research. 14 (3): 351–61. doi:10.1359/jbmr.1999.14.3.351. PMID 10027900. S2CID 23683884.
- ^ Tondreau T, Lagneaux L, Dejeneffe M, Delforge A, Massy M, Mortier C, Bron D (1 January 2004). "Isolation of BM mesenchymal stem cells by plastic adhesion or negative selection: phenotype, proliferation kinetics and differentiation potential". Cytotherapy. 6 (4): 372–9. doi:10.1080/14653240410004943. PMID 16146890.
- ^ Iudicone P, Fioravanti D, Bonanno G, Miceli M, Lavorino C, Totta P, et al. (January 2014). "Pathogen-free, plasma-poor platelet lysate and expansion of human mesenchymal stem cells". Journal of Translational Medicine. 12: 28. doi:10.1186/1479-5876-12-28. PMC 3918216. PMID 24467837.
- ^ Jiang B, Fu X, Yan L, Li S, Zhao D, Wang X, et al. (2019). "Transplantation of human ESC-derived mesenchymal stem cell spheroids ameliorates spontaneous osteoarthritis in rhesus macaques". Theranostics. 9 (22): 6587–6600. doi:10.7150/thno.35391. PMC 6771254. PMID 31588237.
- ^ Borzabadi-Farahani A (September 2016). "Effect of low-level laser irradiation on proliferation of human dental mesenchymal stem cells; a systemic review". Journal of Photochemistry and Photobiology. B, Biology. 162: 577–582. doi:10.1016/j.jphotobiol.2016.07.022. PMID 27475781.
- ^ Becker AJ, McCulloch EA, Till JE (February 1963). "Cytological demonstration of the clonal nature of spleen colonies derived from transplanted mouse marrow cells". Nature. 197 (4866): 452–4. Bibcode:1963Natur.197..452B. doi:10.1038/197452a0. hdl:1807/2779. PMID 13970094. S2CID 11106827.
- ^ Siminovitch L, Mcculloch EA, Till JE (December 1963). "The distribution of colony-forming cells among spleen colonies". Journal of Cellular and Comparative Physiology. 62 (3): 327–36. doi:10.1002/jcp.1030620313. hdl:1807/2778. PMID 14086156.
- ^ Caplan AI (June 2017). "Mesenchymal Stem Cells: Time to Change the Name!". Stem Cells Translational Medicine. 6 (6): 1445–1451. doi:10.1002/sctm.17-0051. PMC 5689741. PMID 28452204.
- ^ Friedenstein AJ, Deriglasova UF, Kulagina NN, Panasuk AF, Rudakowa SF, Luriá EA, Ruadkow IA (1974). "Precursors for fibroblasts in different populations of hematopoietic cells as detected by the in vitro colony assay method". Experimental Hematology. 2 (2): 83–92. PMID 4455512.
- ^ Friedenstein AJ, Gorskaja JF, Kulagina NN (September 1976). "Fibroblast precursors in normal and irradiated mouse hematopoietic organs". Experimental Hematology. 4 (5): 267–74. PMID 976387.
- ^ Lazarus HM, Haynesworth SE, Gerson SL, Rosenthal NS, Caplan AI (October 1995). "Ex vivo expansion and subsequent infusion of human bone marrow-derived stromal progenitor cells (mesenchymal progenitor cells): implications for therapeutic use". Bone Marrow Transplantation. 16 (4): 557–564. PMID 8528172.
- ^ Galipeau J, Sensébé L (June 2018). "Mesenchymal Stromal Cells: Clinical Challenges and Therapeutic Opportunities". Cell Stem Cell. 22 (6): 824–833. doi:10.1016/j.stem.2018.05.004. PMC 6434696. PMID 29859173.
- ^ Lazarus HM, Haynesworth SE, Gerson SL, Caplan AI (October 1997). "Human bone marrow-derived mesenchymal (stromal) progenitor cells (MPCs) cannot be recovered from peripheral blood progenitor cell collections". Journal of Hematotherapy. 6 (5): 447–455. doi:10.1089/scd.1.1997.6.447. PMID 9368181.
- ^ Caplan AI (February 2019). "Medicinal signalling cells: they work, so use them". Nature. 566 (7742): 39. Bibcode:2019Natur.566R..39C. doi:10.1038/d41586-019-00490-6. PMID 30723355.
- ^ Vigdor N (4 February 2021). "Lawmaker Promoted Stem Cell Therapy for Covid-19 in Fraud Scheme, U.S. Says". The New York Times. ISSN 0362-4331. Retrieved 8 February 2021.
- ^ Office of the Commissioner (9 September 2020). "FDA Warns About Stem Cell Therapies". U.S. Food and Drug Administration.
Further reading
[edit]- Murphy MB, Moncivais K, Caplan AI (November 2013). "Mesenchymal stem cells: environmentally responsive therapeutics for regenerative medicine". Experimental & Molecular Medicine. 45 (11): e54. doi:10.1038/emm.2013.94. PMC 3849579. PMID 24232253.
External links
[edit]- "Mesenchymal stem cells fact sheet". Euro Stem Cell. June 2012. Archived from the original on 27 November 2016. Retrieved 25 June 2012. scientist-reviewed and not too technical
- "Mesenchymal Stem Cell Research". Johns Hopkins University. Archived from the original on 15 October 2017. Retrieved 26 June 2012.
