User:Mr. Ibrahem/Cabergoline
 | |
| Clinical data | |
|---|---|
| Trade names | Cabaser, Dostinex, others |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Routes of administration | By mouth |
| Drug class | Dopamine receptor agonists |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | First-pass effect seen; absolute bioavailability unknown |
| Protein binding | Moderately bound (40–42%); concentration-independent |
| Metabolism | Liver, predominately via hydrolysis of the acylurea bond or the urea moiety |
| Elimination half-life | 63–69 hours (estimated) |
| Excretion | Urine (22%), feces (60%) |
| Identifiers | |
| |
| Chemical and physical data | |
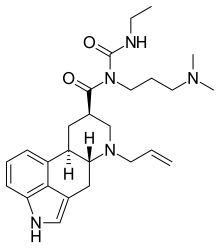
| Formula | C26H37N5O2 |
| Molar mass | 451.615 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Cabergoline, sold under the brand names Dostinex among others, is a medication used for high blood prolactin and less commonly for Parkinson's and to suppress milk production.[1][2] Outside of pregnancy, it is preferred to bromocriptine due to less side effects.[1] It is taken by mouth.[1]
Common side effects include nausea, abdominal pain, headache, sleepiness, and constipation.[1] Other side effects include pulmonary fibrosis and low blood pressure with standing.[1] There is no clear evidence of harm of use during pregnancy, but such use has not been well studied.[5] Carbergoline works by stimulating dopamine receptors and decreasing the release of prolactin.[2]
Cabergoline was patented in 1980 and approved for medical use in 1993.[6] It is avaliable as a generic medication.[2] In the United Kingdom 20 tablets of 1 mg costs the NHS about 65 pounds in 2020.[2] This amount in the US costs about 72 USD.[7]
References[edit]
- ^ a b c d e f g "Cabergoline Monograph for Professionals". Drugs.com. Archived from the original on 27 February 2021. Retrieved 9 November 2020.
- ^ a b c d e BNF 79. London: Pharmaceutical Press. March 2020. p. 432. ISBN 978-0857113658.
- ^ "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 30 October 2020. Retrieved 9 November 2020.
- ^ "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 11 April 2021. Retrieved 9 November 2020.
- ^ "Cabergoline (Dostinex) Use During Pregnancy". Drugs.com. Archived from the original on 24 November 2020. Retrieved 9 November 2020.
- ^ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 533. ISBN 9783527607495. Archived from the original on 2021-08-28. Retrieved 2020-09-19.
- ^ "Cabergoline Prices and Cabergoline Coupons". GoodRx. Retrieved 9 November 2020.
