User:Mr. Ibrahem/Deflazacort
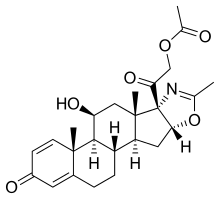 | |
| Clinical data | |
|---|---|
| Trade names | Calcort, Emflaza, others |
| AHFS/Drugs.com | Monograph |
| Routes of administration | By mouth |
| Drug class | Glucocorticoid[1] |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 40% |
| Metabolism | By plasma esterases, to active metabolite |
| Elimination half-life | 1.1–1.9 hours (metabolite) |
| Excretion | Kidney (70%) and fecal (30%) |
| Identifiers | |
| |
| Chemical and physical data | |
| Formula | C25H31NO6 |
| Molar mass | 441.524 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Deflazacort, sold under the trade name Calcort among others, is a glucocorticoid used to treat allergic disorders and inflammatory disorders.[1] It may also be used for Duchenne muscular dystrophy.[2] It is taken by mouth.[1]
Common side effects include weight gain, upper respiratory tract infection, increased hair growth, red skin, and irritability.[2] Other side effects may include infection, adrenal insufficiency, Cushing syndrome, high blood sugar, anaphylaxis, blood clots, and toxic epidermal necrolysis.[2] Deflazacort 6 mg has a similar anti-inflammatory effect as prednisone 5 mg.[1]
Deflazacort was patented in 1965 and approved for medical use in 1985.[3] In the United Kingdom 60 tablets of 6 mg costs the NHS about £16 as of 2021.[1] In the United States this amount costs about 4,100 USD.[4]
References[edit]
- ^ a b c d e f BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 714. ISBN 978-0857114105.
- ^ a b c d "Deflazacort Monograph for Professionals". Drugs.com. Archived from the original on 12 August 2020. Retrieved 22 December 2021.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 486. ISBN 9783527607495. Archived from the original on 2021-08-28. Retrieved 2021-07-01.
- ^ "Emflaza Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 14 August 2020. Retrieved 22 December 2021.
