User:Mr. Ibrahem/Empagliflozin
 | |
| Clinical data | |
|---|---|
| Trade names | Jardiance, others |
| Other names | BI-10773 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a614043 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Sodium-glucose cotransporter-2 (SGLT2) inhibitor[2] |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| Chemical and physical data | |
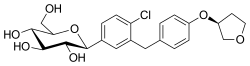
| Formula | C23H27ClO7 |
| Molar mass | 450.91 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Empagliflozin, sold under the brand name Jardiance among others, is a medication used together with diet and exercise to treat type 2 diabetes.[2][6] It is less preferred than metformin and sulfonylureas.[7] It may be used together with other medications such as metformin or insulin.[2][6] It is not recommended for type 1 diabetes.[2] It is taken by mouth.[2]
Common side effects include urinary tract infections, fungal infections of the groin, and joint pains.[2] Rarer but more serious side effects include a skin infection of the groin called Fournier's gangrene and a form of diabetic ketoacidosis with normal blood sugar levels.[2][8] Use in pregnancy and breastfeeding is not recommended.[7] Use is not recommended in those with significant kidney disease, though it may help slow the progression of mild kidney problems.[2][6] Empagliflozin is an inhibitor of the sodium glucose co-transporter-2 (SGLT-2), and works by increasing sugar lost in the urine.[2]
Empagliflozin was approved for medical use in the US and in the European Union in 2014.[2][9][10][11] It is on the World Health Organization's List of Essential Medicines.[12] A month supply in the UK costs the NHS about £36.59 as of 2019[update].[7] In the US, the wholesale cost of this amount is about US$442.[13] In 2017, it was the 228th most commonly prescribed medication in the United States, with more than two million prescriptions.[14][15]
References[edit]
- ^ a b "Empagliflozin (Jardiance) Use During Pregnancy". Drugs.com. 30 August 2018. Archived from the original on 5 August 2019. Retrieved 10 February 2020.
- ^ a b c d e f g h i j "Empagliflozin Monograph for Professionals". Drugs.com. AHFS. Archived from the original on 6 April 2019. Retrieved 21 December 2018.
- ^ "Jardiance 10 mg film-coated tablets - Summary of Product Characteristics (SmPC)". (emc). Archived from the original on 20 September 2020. Retrieved 10 February 2020.
- ^ "Jardiance 25 mg film-coated tablets - Summary of Product Characteristics (SmPC)". (emc). 23 October 2019. Archived from the original on 22 September 2020. Retrieved 10 February 2020.
- ^ "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 17 May 2021. Retrieved 8 September 2020.
- ^ a b c Davies MJ, D'Alessio DA, Fradkin J, Kernan WN, Mathieu C, Mingrone G, Rossing P, Tsapas A, Wexler DJ, Buse JB (December 2018). "Management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD)". Diabetologia. 61 (12): 2461–2498. doi:10.1007/s00125-018-4729-5. PMID 30288571.
- ^ a b c British national formulary: BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 691. ISBN 9780857113382.
- ^ "FDA warns about rare occurrences of a serious infection of the genital area with SGLT2 inhibitors for diabetes". U.S. Food and Drug Administration (FDA). 9 February 2019. Archived from the original on 13 December 2019. Retrieved 18 March 2019.
- ^ "Drug Approval Package: Jardiance (empagliflozin) Tablets NDA #204629". U.S. Food and Drug Administration (FDA). 8 September 2014. Archived from the original on 11 February 2020. Retrieved 10 February 2020.
- ^ "Jardiance: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 11 February 2020. Retrieved 10 February 2020.
- ^ "Jardiance EPAR". European Medicines Agency (EMA). Archived from the original on 28 August 2021. Retrieved 10 February 2020.
- ^ World Health Organization (2023). The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
- ^ "NADAC". Centers for Medicare and Medicaid Services. 27 February 2019. Archived from the original on 6 March 2019. Retrieved 3 March 2019.
- ^ "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ^ "Empagliflozin - Drug Usage Statistics". ClinCalc. Archived from the original on 13 April 2020. Retrieved 11 April 2020.
