User:Mr. Ibrahem/Felbamate
 | |
| Clinical data | |
|---|---|
| Trade names | Felbatol |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a606011 |
| Routes of administration | By mouth (tablets, suspension) |
| Drug class | Anticonvulsant |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | >90% |
| Metabolism | Liver |
| Elimination half-life | 20–23 hours |
| Identifiers | |
| |
| Chemical and physical data | |
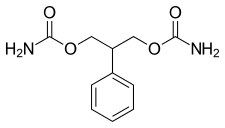
| Formula | C11H14N2O4 |
| Molar mass | 238.24 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Felbamate, sold under the brand name Felbatol, is medication used to treat epilepsy.[1] It is specifically used to treat partial seizures and Lennox–Gastaut syndrome.[1] It is only recommended with safer options are not effective.[1] It is taken by mouth.[1]
Common side effects include loss of appetite, nausea, trouble sleeping, and headache.[1] Side effects may include aplastic anemia, liver failure, Stevens-Johnson syndrome, and suicide.[1] Safety in pregnancy is unclear.[1] How it works is not clear.[1]
Felbamate was approved for medical use in the United States in 1993.[1] It is not approved in the United Kingdom, but can be acquired if other option are not effective.[2] In the United States 30 tablets of 600 mg cost about 95 USD as of 2021.[3]
References
[edit]- ^ a b c d e f g h i j k "Felbamate Monograph for Professionals". Drugs.com. Archived from the original on 6 November 2021. Retrieved 10 December 2021.
- ^ BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 324. ISBN 978-0857114105.
- ^ "Felbamate Prices, Coupons & Patient Assistance Programs". Drugs.com. Retrieved 10 December 2021.
