User:Mr. Ibrahem/Lorlatinib
 | |
| Clinical data | |
|---|---|
| Trade names | Lorbrena, Lorviqua |
| Other names | PF-6463922 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a619005 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Tyrosine kinase inhibitor[1] |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 81% |
| Protein binding | 66% |
| Metabolism | Mainly CYP3A4 and UGT1A4 |
| Elimination half-life | 24 hrs (single dose) |
| Excretion | 48% urine (<1% unchanged), 41% faeces (9% unchanged) |
| Identifiers | |
| |
| Chemical and physical data | |
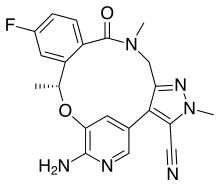
| Formula | C21H19FN6O2 |
| Molar mass | 406.421 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Lorlatinib, sold under the brand name Lorbrena and Lorviqua, is a medication used to treat non-small cell lung cancer (NSCLC).[1] Specifically it is used in anaplastic lymphoma kinase (ALK) positive cases that have failed other treatments.[1] It is taken by mouth.[1]
Common side effects are high cholesterol, high triglycerides, swelling, peripheral neuropathy, problems with memory, tiredness, weight gain, and joint pain.[2] Other side effects may include liver problems, seizures, hallucinations, pneumonitis, and AV block.[1] Use in pregnancy may harm the baby.[1] It is a tyrosine kinase inhibitor of ALK and ROS1.[1]
Lorlatinib was approved for medical use in the United States in 2018 and Europe in 2019.[1][2] In the United Kingdom it costs the NHS about £5,300 a month as of 2021.[3] This amount in the United States is about 18,500 USD.[4]
References[edit]
- ^ a b c d e f g h i j "Lorlatinib Monograph for Professionals". Drugs.com. Archived from the original on 4 March 2021. Retrieved 24 November 2021.
- ^ a b c "Lorviqua". Archived from the original on 14 November 2021. Retrieved 24 November 2021.
- ^ BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 1033. ISBN 978-0857114105.
- ^ "Lorbrena Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 26 January 2021. Retrieved 24 November 2021.
