User:Mr. Ibrahem/Tacalcitol
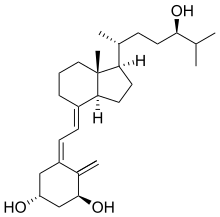 | |
| Clinical data | |
|---|---|
| Trade names | Curatoderm, Bonalfa, others |
| Other names | (1α,24R)-1,24-Dihydroxyvitamin D3 |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Applied to the skin[1] |
| Drug class | Vitamin D3 analog[2] |
| Identifiers | |
| |
| Chemical and physical data | |
| Formula | C27H44O3 |
| Molar mass | 416.64 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Tacalcitol sold under the brand names Curatoderm among others, is a medication used to treat psoriasis, specifically plaque psoriasis.[1][3] It is applied to the skin once to twice per day.[1][2]
Side effects may include skin rash and high calcium.[1] It is a manufactured vitamin D3 analog.[2]
Tacalcitol was approved for medical use in Japan in 1993.[4] It is on the World Health Organization's List of Essential Medicines as an alternative to calcipotriol.[5] In the United Kingdom 30 grams costs the NHS about £13 as of 2023.[6]
References[edit]
- ^ a b c d e f "Tacalcitol". NICE. Archived from the original on 10 September 2023. Retrieved 9 September 2023.
- ^ a b c Peters DC, Balfour JA (August 1997). "Tacalcitol". Drugs. 54 (2): 265–71, discussion 272. doi:10.2165/00003495-199754020-00005. PMID 9257082.
- ^ "eEML - Electronic Essential Medicines List". list.essentialmeds.org. Archived from the original on 10 June 2023. Retrieved 9 September 2023.
- ^ Tarutani, M (October 2004). "[Vitamin D3 for external application--history of development and clinical application]". Clinical calcium. 14 (10): 124–8. PMID 15577144.
- ^ World Health Organization (2023). The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
- ^ "Tacalcitol Medicinal forms". NICE. Archived from the original on 10 September 2023. Retrieved 9 September 2023.
