User:Mr. Ibrahem/Tafluprost
 | |
| Clinical data | |
|---|---|
| Trade names | Saflutan, Taflotan, Tapros, Zioptan, others |
| AHFS/Drugs.com | Multum Consumer Information |
| Routes of administration | Topical (eye drops) |
| Drug class | Prostaglandin analogue[1] |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Activation by ester hydrolysis, deactivation by beta oxidation |
| Onset of action | 2–4 hrs |
| Duration of action | ≥ 24 hrs |
| Identifiers | |
| |
| Chemical and physical data | |
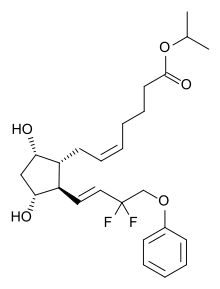
| Formula | C25H34F2O5 |
| Molar mass | 452.539 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Tafluprost, sold under the brand name Zioptan among others, is a medication used to treat open-angle glaucoma and ocular hypertension.[1] It is used as an eye drop.[1] It may be used alone or with other medications.[1]
Common side effects include eye redness, itchiness, eyelash growth, and blurry vision.[1] Other side effects may include iritis and macular edema.[1] It is a prostaglandin analogue which is believed to work by increasing the outflow of aqueous fluid from the eye.[1]
Tafluprost was approved for medical use in the United States in 2012 and Canada in 2014.[1][2] In the United States it costs about 150 USD per month as of 2021.[3] In Canada it was less cost effective as compared to bimatoprost in 2020.[4]
References[edit]
- ^ a b c d e f g h i j "Tafluprost Monograph for Professionals". Drugs.com. Archived from the original on 4 March 2021. Retrieved 20 September 2021.
- ^ Canada, Health (2 July 2014). "Notice: Prescription Drug List (PDL): Multiple additions". www.canada.ca. Archived from the original on 8 July 2021. Retrieved 20 September 2021.
- ^ "Tafluprost Prices, Coupons & Savings Tips - GoodRx". GoodRx. Archived from the original on 1 May 2016. Retrieved 20 September 2021.
- ^ "Prostaglandin Analogues for Ophthalmic Use: A Review of Comparative Clinical Effectiveness, Cost-Effectiveness, and Guidelines | CADTH". www.cadth.ca. Archived from the original on 22 September 2021. Retrieved 20 September 2021.
