Anopheles: Difference between revisions
Tag: possible vandalism |
m tidied refs and separated them from text |
||
| Line 2: | Line 2: | ||
| name = ''Anopheles'' |
| name = ''Anopheles'' |
||
| image = Anopheles stephensi.jpeg |
| image = Anopheles stephensi.jpeg |
||
| image_width = |
| image_width = 270px |
||
| image_caption = ''[[Anopheles stephensi]]'' |
| image_caption = ''[[Anopheles stephensi]]'' |
||
| range_map = Anopheles-range-map.png |
| range_map = Anopheles-range-map.png |
||
| Line 42: | Line 42: | ||
The name comes from the [[Greek language|Greek]] {{lang|el|αν}}, ''{{lang|el|an}}'', meaning ''not'', and {{lang|el|[[:wikt:όφελος|όφελος]]}}, ''{{lang|el|óphelos}}'', meaning ''profit'', and translates to ''useless''.<ref name="dict"/> |
The name comes from the [[Greek language|Greek]] {{lang|el|αν}}, ''{{lang|el|an}}'', meaning ''not'', and {{lang|el|[[:wikt:όφελος|όφελος]]}}, ''{{lang|el|óphelos}}'', meaning ''profit'', and translates to ''useless''.<ref name="dict"/> |
||
Some species of ''Anopheles'' also can serve as the [[Vector (epidemiology)|vectors]] for canine [[heartworm]] ''[[Dirofilaria immitis]]'', the [[filariasis]]-causing species ''[[Wuchereria bancrofti]]'' and ''[[Brugia malayi]]'', and [[virus]]es such as one that causes [[O'nyong'nyong]] fever. There is an association of [[brain tumor]] incidence and [[malaria]], suggesting that the Anopheles might transmit a virus or other agent that could cause a brain tumor.<ref |
Some species of ''Anopheles'' also can serve as the [[Vector (epidemiology)|vectors]] for canine [[heartworm]] ''[[Dirofilaria immitis]]'', the [[filariasis]]-causing species ''[[Wuchereria bancrofti]]'' and ''[[Brugia malayi]]'', and [[virus]]es such as one that causes [[O'nyong'nyong]] fever. There is an association of [[brain tumor]] incidence and [[malaria]], suggesting that the Anopheles might transmit a virus or other agent that could cause a brain tumor.<ref name=r8/> |
||
Mosquitoes in other genera (''[[Aedes]]'', ''[[Culex]]'') can also serve as vectors of disease agents, but not malaria. |
Mosquitoes in other genera (''[[Aedes]]'', ''[[Culex]]'') can also serve as vectors of disease agents, but not malaria. |
||
| Line 48: | Line 48: | ||
==Evolution== |
==Evolution== |
||
The ancestors of ''[[Drosophila]]'' and mosquitoes diverged {{ma|260}} ago.<ref name="Moreno2010" |
The ancestors of ''[[Drosophila]]'' and mosquitoes diverged {{ma|260}} ago.<ref name="Moreno2010"/> The [[Culicinae|culicine]] and ''Anopheles'' clades of mosquitoes diverged between {{ma|120}} and {{ma|150}}.<ref name="Moreno2010"/><ref name="Calvo2009"/> The Old and New World ''Anopheles'' species subsequently diverged between {{Ma|80}} and {{Ma|95}}.<ref name="Moreno2010"/><ref name="Calvo2009"/> The ''[[Anopheles gambiae]]'' and ''[[Anopheles funestus]]'' clades diverged {{Ma|80|36}}. A molecular study of several genes in seven species has provided additional support for an expansion of this genus during the [[Cretaceous]] period.<ref name="Dixit2010"/> |
||
The ''Anopheles'' genome (230–284 Mb) is comparable in size to that of ''[[Drosophila]]'' but considerably smaller than those found in other culicine genomes (528 Mb–1.9 Gb). Like most culicine species, the genome is [[diploid]] with six [[chromosome]]s. |
The ''Anopheles'' genome (230–284 Mb) is comparable in size to that of ''[[Drosophila]]'' but considerably smaller than those found in other culicine genomes (528 Mb–1.9 Gb). Like most culicine species, the genome is [[diploid]] with six [[chromosome]]s. |
||
| Line 58: | Line 58: | ||
The genus ''Anopheles'' belongs to a subfamily Anophelinae with three genera: ''Anopheles'' Meigen (nearly worldwide distribution), ''[[Bironella]]'' Theobald ([[Australia]] only) and ''[[Chagasia]]'' Cruz ([[Neotropics]]). ''Bironella'' appears to be the sister taxon to the ''Anopheles'' with ''Chagasia'' forming the outgroup in this subfamily. |
The genus ''Anopheles'' belongs to a subfamily Anophelinae with three genera: ''Anopheles'' Meigen (nearly worldwide distribution), ''[[Bironella]]'' Theobald ([[Australia]] only) and ''[[Chagasia]]'' Cruz ([[Neotropics]]). ''Bironella'' appears to be the sister taxon to the ''Anopheles'' with ''Chagasia'' forming the outgroup in this subfamily. |
||
The classification of this genus began in 1901 with Theobald. Despite the passage of time the taxonomy remains incompletely settled. Classification into species is based on morphological characteristics |
The classification of this genus began in 1901 with Theobald. Despite the passage of time the taxonomy remains incompletely settled. Classification into species is based on morphological characteristics – wing spots, head anatomy, larval and pupal anatomy, chromosome structure – and more recently on DNA sequences. |
||
The genus itself has been subdivided into seven subgenera based primarily on the number and positions of specialized [[setae]] on the [[gonocoxite]]s of the [[male]] [[genitalia]]. The system of subgenera originated with the work of Christophers who in 1915 described three subgenera: ''Anopheles'' (widely distributed), ''Myzomyia'' (later renamed ''[[Cellia]]'') ([[Old World]]) and ''[[Nyssorhynchus]]'' (Neotropical). ''Nyssorhynchus'' was first described as ''Lavernia'' by Theobald. Edwards in 1932 added the subgenus ''[[Stethomyia]]'' (Neotropical distribution). ''[[Kerteszia]]'' was also described by Edwards in 1932 but then recognised as a subgrouping of ''Nyssorhynchus''. It was elevated to subgenus status by Komp in 1937 and it is also found in the Neotropics. Two additional subgenera have since been recognised: ''[[Baimaia]]'' (Southeast Asia only) by Harbach ''et al.'' in 2005 and ''[[Lophopodomyia]]'' (Neotropical) by Antunes in 1937. |
The genus itself has been subdivided into seven subgenera based primarily on the number and positions of specialized [[setae]] on the [[gonocoxite]]s of the [[male]] [[genitalia]]. The system of subgenera originated with the work of Christophers who in 1915 described three subgenera: ''Anopheles'' (widely distributed), ''Myzomyia'' (later renamed ''[[Cellia]]'') ([[Old World]]) and ''[[Nyssorhynchus]]'' (Neotropical). ''Nyssorhynchus'' was first described as ''Lavernia'' by Theobald. Edwards in 1932 added the subgenus ''[[Stethomyia]]'' (Neotropical distribution). ''[[Kerteszia]]'' was also described by Edwards in 1932 but then recognised as a subgrouping of ''Nyssorhynchus''. It was elevated to subgenus status by Komp in 1937 and it is also found in the Neotropics. Two additional subgenera have since been recognised: ''[[Baimaia]]'' (Southeast Asia only) by Harbach ''et al.'' in 2005 and ''[[Lophopodomyia]]'' (Neotropical) by Antunes in 1937. |
||
| Line 95: | Line 95: | ||
=== Pupae === |
=== Pupae === |
||
The pupa is comma-shaped when viewed from the side. The head and [[Thorax (insect anatomy)|thorax]] are merged into a [[cephalothorax]] with the abdomen curving around underneath. As with the larvae, pupae must come to the surface frequently to breathe, which they do through a pair of respiratory trumpets on the cephalothorax. After a few days as a pupa, the dorsal surface of the cephalothorax splits and the adult mosquito emerges. The Pupa stage lasts around |
The pupa is comma-shaped when viewed from the side. The head and [[Thorax (insect anatomy)|thorax]] are merged into a [[cephalothorax]] with the abdomen curving around underneath. As with the larvae, pupae must come to the surface frequently to breathe, which they do through a pair of respiratory trumpets on the cephalothorax. After a few days as a pupa, the dorsal surface of the cephalothorax splits and the adult mosquito emerges. The Pupa stage lasts around 2–3 days in temperate areas. |
||
kallaw, 2012 |
|||
=== Adults === |
=== Adults === |
||
| Line 121: | Line 119: | ||
== Habitat == |
== Habitat == |
||
Although malaria is nowadays limited to tropical areas, most notoriously regions of sub-Saharan Africa, many ''Anopheles'' species live in colder latitudes (see [http://www.cdc.gov/malaria/about/biology/mosquitoes/map.html this map] from the CDC). Indeed, malaria outbreaks have, in the past, occurred in colder climates, for example during the construction of the [[Rideau Canal]] in Canada during the 1820s.<ref |
Although malaria is nowadays limited to tropical areas, most notoriously regions of sub-Saharan Africa, many ''Anopheles'' species live in colder latitudes (see [http://www.cdc.gov/malaria/about/biology/mosquitoes/map.html this map] from the CDC). Indeed, malaria outbreaks have, in the past, occurred in colder climates, for example during the construction of the [[Rideau Canal]] in Canada during the 1820s.<ref name=r7/> Since then, the ''Plasmodium'' parasite (not the ''Anopheles'' mosquito) has been eliminated from first world countries. |
||
The CDC warns, however, that "''Anopheles'' that can transmit malaria are found not only in malaria-endemic areas, but also in areas where malaria has been eliminated. The latter areas are thus constantly at risk of re-introduction of the disease." |
The CDC warns, however, that "''Anopheles'' that can transmit malaria are found not only in malaria-endemic areas, but also in areas where malaria has been eliminated. The latter areas are thus constantly at risk of re-introduction of the disease." |
||
| Line 132: | Line 130: | ||
Understanding the biology and behavior of ''Anopheles'' mosquitoes can help understand how [[malaria]] is transmitted and can aid in designing appropriate control strategies. Factors that affect a mosquito's ability to transmit malaria include its innate susceptibility to ''[[Plasmodium]]'', its host choice and its longevity. Factors that should be taken into consideration when designing a control program include the susceptibility of malaria vectors to [[insecticide]]s and the preferred feeding and resting location of adult mosquitoes. |
Understanding the biology and behavior of ''Anopheles'' mosquitoes can help understand how [[malaria]] is transmitted and can aid in designing appropriate control strategies. Factors that affect a mosquito's ability to transmit malaria include its innate susceptibility to ''[[Plasmodium]]'', its host choice and its longevity. Factors that should be taken into consideration when designing a control program include the susceptibility of malaria vectors to [[insecticide]]s and the preferred feeding and resting location of adult mosquitoes. |
||
On December 21, 2007, a study published in [[PLoS Pathogens]] found that the hemolytic C-type [[lectin]] CEL-III from ''Cucumaria echinata'', a [[Holothuroidea|sea cucumber]] found in the [[Bay of Bengal]], impaired the development of the malaria parasite when produced by transgenic ''A. stephensi''.<ref |
On December 21, 2007, a study published in [[PLoS Pathogens]] found that the hemolytic C-type [[lectin]] CEL-III from ''Cucumaria echinata'', a [[Holothuroidea|sea cucumber]] found in the [[Bay of Bengal]], impaired the development of the malaria parasite when produced by transgenic ''A. stephensi''.<ref name=r6/> This could potentially be used one day to control malaria by spreading genetically modified mosquitoes refractory to the parasites, although there are numerous scientific and ethical issues to be overcome before such a control strategy could be implemented. |
||
=== Preferred sources for blood meals === |
=== Preferred sources for blood meals === |
||
| Line 139: | Line 137: | ||
Once ingested by a mosquito, malaria parasites must undergo development within the mosquito before they are infectious to humans. The time required for development in the mosquito (the extrinsic [[incubation period]]) ranges from 10–21 days, depending on the [[parasite]] species and the temperature. If a mosquito does not survive longer than the extrinsic incubation period, then she will not be able to transmit any malaria parasites. |
Once ingested by a mosquito, malaria parasites must undergo development within the mosquito before they are infectious to humans. The time required for development in the mosquito (the extrinsic [[incubation period]]) ranges from 10–21 days, depending on the [[parasite]] species and the temperature. If a mosquito does not survive longer than the extrinsic incubation period, then she will not be able to transmit any malaria parasites. |
||
It is not possible to measure directly the life span of mosquitoes in nature. But indirect estimates of daily survivorship have been made for several ''Anopheles'' species. Estimates of daily survivorship of ''A. gambiae'' in [[Tanzania]] ranged from 0.77 to 0.84, meaning that at the end of one day between 77% and 84% will have survived.<ref |
It is not possible to measure directly the life span of mosquitoes in nature. But indirect estimates of daily survivorship have been made for several ''Anopheles'' species. Estimates of daily survivorship of ''A. gambiae'' in [[Tanzania]] ranged from 0.77 to 0.84, meaning that at the end of one day between 77% and 84% will have survived.<ref name=r5/> |
||
Assuming this survivorship is constant through the adult life of a mosquito, less than 10% of female ''A. gambiae'' would survive longer than a 14-day extrinsic incubation period. If daily survivorship increased to 0.9, over 20% of mosquitoes would survive longer than a 14-day extrinsic incubation period. Control measures that rely on [[insecticide]]s (e.g. indoor residual spraying) may actually impact malaria [[Transmission (medicine)|transmission]] more through their effect on adult longevity than through their effect on the population of adult mosquitoes. |
Assuming this survivorship is constant through the adult life of a mosquito, less than 10% of female ''A. gambiae'' would survive longer than a 14-day extrinsic incubation period. If daily survivorship increased to 0.9, over 20% of mosquitoes would survive longer than a 14-day extrinsic incubation period. Control measures that rely on [[insecticide]]s (e.g. indoor residual spraying) may actually impact malaria [[Transmission (medicine)|transmission]] more through their effect on adult longevity than through their effect on the population of adult mosquitoes. |
||
| Line 149: | Line 147: | ||
=== Gut flora === |
=== Gut flora === |
||
Because transmission of disease by the mosquito requires ingestion of blood it is reasonable to presuppose that the gut flora may have a bearing on the success of infection of the mosquito host. This aspect of disease transmission has not been investigated until recently.<ref name=Wang2011 |
Because transmission of disease by the mosquito requires ingestion of blood it is reasonable to presuppose that the gut flora may have a bearing on the success of infection of the mosquito host. This aspect of disease transmission has not been investigated until recently.<ref name=Wang2011/> The larval and pupal gut is largely colonised by photosynthetic cyanobacteria while in the adult [[Proteobacteria]] and [[Bacteroidetes]] predominate. Blood meals drastically reduce the diversity of organisms and favor enteric bacteria. |
||
== Insecticide resistance == |
== Insecticide resistance == |
||
| Line 157: | Line 155: | ||
==Eradication== |
==Eradication== |
||
With substantial numbers of malaria cases affecting people around the globe, in [[tropical]] and [[subtropical]] regions, especially in [[sub-Saharan Africa]], where millions of children are killed by this infectious disease, eradication is back on the global health agenda.<ref name="Tanner" |
With substantial numbers of malaria cases affecting people around the globe, in [[tropical]] and [[subtropical]] regions, especially in [[sub-Saharan Africa]], where millions of children are killed by this infectious disease, eradication is back on the global health agenda.<ref name="Tanner"/> |
||
Although malaria has existed since old times, its eradication was possible in [[Europe]], [[North America]], the [[Caribbean]] and parts of [[Asia]] and southern Central America during the first regional elimination campaigns in the late 1940s. However, the same results were not achieved in sub-Saharan Africa.<ref name="Tanner"/> |
Although malaria has existed since old times, its eradication was possible in [[Europe]], [[North America]], the [[Caribbean]] and parts of [[Asia]] and southern Central America during the first regional elimination campaigns in the late 1940s. However, the same results were not achieved in sub-Saharan Africa.<ref name="Tanner"/> |
||
Even though the [[World Health Organization]] adopted a formal policy on the control and eradication of the malaria parasite since 1955,<ref>{{cite web |url=http://www.cwru.edu/med/epidbio/mphp439/Malaria.htm |title=Malaria Eradication |
Even though the [[World Health Organization]] adopted a formal policy on the control and eradication of the malaria parasite since 1955,<ref>{{cite web |url=http://www.cwru.edu/med/epidbio/mphp439/Malaria.htm |title=Malaria Eradication |accessdate=2010-05-04}}</ref> it was recently, after the Gates Malaria Forum in October 2007, that key organizations started the debate on the pros and cons of redefining eradication as a goal to control malaria. |
||
Clearly, the cost of preventing malaria is much less than treating the disease, in the long run. However, eradication of mosquito is not an easy task. For effective prevention of malaria, some conditions should be met such as conducive conditions in the country, [[data collection]] about the disease, targeted technical approach to the problem, very active and committed leadership, government’s total support, monetary free hand, community involvement, skilled technicians from different fields as well as an adequate implementation.<ref |
Clearly, the cost of preventing malaria is much less than treating the disease, in the long run. However, eradication of mosquito is not an easy task. For effective prevention of malaria, some conditions should be met such as conducive conditions in the country, [[data collection]] about the disease, targeted technical approach to the problem, very active and committed leadership, government’s total support, monetary free hand, community involvement, skilled technicians from different fields as well as an adequate implementation.<ref name=r4/> |
||
There is a wide range of strategies to achieve malaria eradication that start from simple steps to complicated strategies which may not be possible to enforce with the current tools. |
There is a wide range of strategies to achieve malaria eradication that start from simple steps to complicated strategies which may not be possible to enforce with the current tools. |
||
Although mosquito control is an important component of malaria control strategy, elimination of malaria in an area does not require the elimination of all ''Anopheles'' mosquitoes. For instance, in North America and Europe, although the vector ''Anopheles'' mosquitoes are still present, the parasite has been eliminated. There are also some socioeconomic improvements (e.g., houses with screened windows, [[air conditioning]]) that once combined with vector reduction efforts and effective treatment lead to the elimination of malaria without the complete elimination of the vectors. Some important measures in mosquito control to be followed are: discourage egg laying, prevent development of eggs into larvae and adults, kill the adult mosquitoes, do not allow adult mosquitoes into places of human dwelling, prevent mosquitoes from biting human beings and deny blood meal.<ref |
Although mosquito control is an important component of malaria control strategy, elimination of malaria in an area does not require the elimination of all ''Anopheles'' mosquitoes. For instance, in North America and Europe, although the vector ''Anopheles'' mosquitoes are still present, the parasite has been eliminated. There are also some socioeconomic improvements (e.g., houses with screened windows, [[air conditioning]]) that once combined with vector reduction efforts and effective treatment lead to the elimination of malaria without the complete elimination of the vectors. Some important measures in mosquito control to be followed are: discourage egg laying, prevent development of eggs into larvae and adults, kill the adult mosquitoes, do not allow adult mosquitoes into places of human dwelling, prevent mosquitoes from biting human beings and deny blood meal.<ref name=r3/> |
||
Research in this sense continues, and a study has suggested that sterile mosquitoes might be the answer to malaria elimination. This research suggests that using the [[sterile insect technique]] (SIT), in which sexually sterile male insects are released to wipe out a pest population, could be a solution to the problem of malaria in Africa. This technique brings hope, as female mosquitoes only mate once during their lifetimes, and in doing so with sterile male mosquitoes, the insect population would decrease.<ref |
Research in this sense continues, and a study has suggested that sterile mosquitoes might be the answer to malaria elimination. This research suggests that using the [[sterile insect technique]] (SIT), in which sexually sterile male insects are released to wipe out a pest population, could be a solution to the problem of malaria in Africa. This technique brings hope, as female mosquitoes only mate once during their lifetimes, and in doing so with sterile male mosquitoes, the insect population would decrease.<ref name=r2/> This is another option to be considered by local and international authorities that may be combined with other methods and tools to achieve malaria eradication in sub-Saharan Africa. |
||
==Parasites== |
==Parasites== |
||
A number of parasites of this genus are known to exist including [[microsporidia]] of the genera ''[[Amblyospora]]'', ''[[Crepidulospora]]'', ''[[Senoma]]'' and ''[[Parathelohania]]''.<ref name="Simakova2008" |
A number of parasites of this genus are known to exist including [[microsporidia]] of the genera ''[[Amblyospora]]'', ''[[Crepidulospora]]'', ''[[Senoma]]'' and ''[[Parathelohania]]''.<ref name="Simakova2008"/> |
||
Microsporida infecting the aquatic stages of insects, a group that includes mosquitoes and [[black fly|black flies]], and copepods appear to form a distinct clade from those infecting terrestrial insects and fish. There are two distinct life cycles in this group: in the first type the parasite is transmitted by the oral route and is relatively non species specific. In the second, while again the oral route is the usual route of infection, the parasite is ingested within an already infected intermediate host. Infection of the insect larval form is frequently tissue specific, and commonly involves the fat body. Vertical (transovarial) transmission is also known to occur. |
Microsporida infecting the aquatic stages of insects, a group that includes mosquitoes and [[black fly|black flies]], and copepods appear to form a distinct clade from those infecting terrestrial insects and fish. There are two distinct life cycles in this group: in the first type the parasite is transmitted by the oral route and is relatively non species specific. In the second, while again the oral route is the usual route of infection, the parasite is ingested within an already infected intermediate host. Infection of the insect larval form is frequently tissue specific, and commonly involves the fat body. Vertical (transovarial) transmission is also known to occur. |
||
Few phylogenetic studies of these parasites have been done, and their the relationship to their mosquito hosts is still being determined. One study suggested ''Parathelohania'' is an early diverging genus within this group.<ref |
Few phylogenetic studies of these parasites have been done, and their the relationship to their mosquito hosts is still being determined. One study suggested ''Parathelohania'' is an early diverging genus within this group.<ref name=r1/> |
||
The parasite ''[[Wolbachia]]'' has been studied for use as a control agent.<ref name= |
The parasite ''[[Wolbachia]]'' has been studied for use as a control agent.<ref name="Discovery Article">[http://news.discovery.com/animals/mosquito-parasite-disease-fighting.html Mosquito Parasite Fights Infectious Disease : Discovery News]. News.discovery.com (2009-10-01). Retrieved on 2012-07-14.</ref> |
||
== See also == |
== See also == |
||
| Line 187: | Line 185: | ||
== References == |
== References == |
||
{{Reflist| |
{{Reflist|35em|refs= |
||
<ref name="Moreno2010">{{cite journal|pmid=20470395|pmc=2877063|author=Moreno M, Marinotti O, Krzywinski J, Tadei WP, James AA, Achee NL, Conn JE|year=2010|title=Complete mtDNA genomes of Anopheles darlingi and an approach to anopheline divergence time|volume=9|pages=127|doi=10.1186/1475-2875-9-127|journal=Malaria journal}}</ref> |
|||
| ⚫ | |||
| ⚫ | |||
<ref name=r1>{{cite journal |author=Michael D. Baker, Charles R. Vossbrinck, James J. Becnel & Theodore G. Andreadis |year=1998 |title=Phylogeny of ''Amblyospora'' (Microsporida: Amblyosporidae) and related genera based on small subunit ribosomal DNA data: a possible example of host parasite cospeciation |journal=[[Journal of Invertebrate Pathology]] |volume=71 |issue=3 |pages=199–206 |pmid=9538024 |doi=10.1006/jipa.1997.4725 |url=http://www.ct.gov/caes/LIB/caes/documents/biographies/BakerJIP98.pdf |format=PDF}}</ref> |
|||
<ref name=r2>{{cite web |url=http://esciencenews.com/articles/2009/11/16/are.sterile.mosquitoes.answer.malaria.elimination |title=Are sterile mosquitoes the answer to malaria elimination? |accessdate=2010-05-04| archiveurl= http://web.archive.org/web/20100430114312/http://esciencenews.com/articles/2009/11/16/are.sterile.mosquitoes.answer.malaria.elimination| archivedate= 30 April 2010| deadurl= no}}</ref> |
|||
<ref name=r3>{{cite web |url=http://www.malariasite.com/MALARIA/mosquito_control.htm |title=Mosquito Control |accessdate=2010-05-04 |archiveurl=http://web.archive.org/web/20080501074333/http://www.malariasite.com/MALARIA/mosquito_control.htm |archivedate=May 1, 2008}}</ref> |
|||
<ref name=r4>{{cite web |url=http://www.malariasymptoms.org/malaria-preventing.html |title=Mosquito Eradication |date= |accessdate=2010-05-04| archiveurl= http://web.archive.org/web/20100503104126/http://www.malariasymptoms.org/malaria-preventing.html| archivedate= 3 May 2010 | deadurl= no}}</ref> |
|||
<ref name=r5>{{cite journal |author=J. D. Charlwood, T. Smith, P. F. Billingsley, W. Takken, E. O. K. Lyimo & J. H. E. T. Meuwissen |year=1997 |title=Survival And infection probabilities of anthropophagic anophelines from an area of high prevalence of ''Plasmodium falciparum'' in humans |journal=[[Bulletin of Entomological Research]] |volume=87 |issue=5 |pages=445–453 |doi=10.1017/S0007485300041304}}</ref> |
|||
<ref name=r6>{{cite journal |author=Shigeto Yoshida, Yohei Shimada , Daisuke Kondoh, Yoshiaki Kouzuma, Anil K. Ghosh, Marcelo Jacobs-Lorena & Robert E. Sinden |title=Hemolytic C-type lectin CEL-III from sea cucumber expressed in transgenic mosquitoes impairs malaria parasite development |journal=[[PLoS Pathogens]] |volume=3 |issue=12 |page=e192 |year=2007 |pmid=18159942 |doi=10.1371/journal.ppat.0030192 |pmc=2151087}}</ref> |
|||
<ref name=r7>{{Cite journal |author=William N. T. Wylie |year=1983 |title=Poverty, Distress, and Disease: Labour and the Construction of the Rideau Canal, 1826–32 |journal=[[Labour/Le Travail]] |volume=11 |pages=7–29 |jstor=25140199 |doi=10.2307/25140199}}</ref> |
|||
<ref name=r8>{{cite journal |author=Steven Lehrer |year=2010 |title=''Anopheles'' mosquito transmission of brain tumor |journal=[[Medical Hypotheses]] |volume=74 |issue=1 |pages=167–168 |pmid=19656635 |doi=10.1016/j.mehy.2009.07.005 |url=http://www.stevenlehrer.com/images/medhypinpress09.pdf |format=PDF}}</ref> |
|||
<ref name="Simakova2008">{{Cite journal |author=A. V. Simakova & T. F. Pankova |year=2008 |title=Ecology and epizootology of microsporidia in malarial mosquitoes (Diptera: Culicidae) from the south of western Siberia |journal=Parazitologiia |volume=42 |issue=2 |pages=139–150 |pmid=18664069 |language=Russian}}</ref> |
|||
<ref name="Tanner">{{cite journal |author=Marcel Tanner & Don de kSavigny |year=2008 |title=Malaria eradication back on the table |journal=[[Bulletin of the World Health Organization]] |volume=86 |issue=2 |pages=82–83 |doi=10.2471/BLT.07.050633 |url=http://www.who.int/bulletin/volumes/86/2/07-050633/en/}}</ref> |
|||
<ref name=Wang2011>{{cite journal|title=Dynamic gut microbiome across life history of the malaria mosquito Anopheles gambiae in Kenya|doi=10.1371/journal.pone.0024767|author=Wang Y, Gilbreath TM 3rd, Kukutla P, Yan G, Xu J|year=2011|editor1-last=Leulier|editor1-first=François|journal=PLoS ONE|volume=6|issue=9|pages=e24767|pmid=21957459|pmc=3177825}}</ref> |
|||
<ref name="Calvo2009">{{Cite journal |author=Eric Calvo, Van M Pham, Osvaldo Marinotti, John F. Andersen & José M. C. Ribeiro |year=2009 |title=The salivary gland transcriptome of the neotropical malaria vector ''Anopheles darlingi'' reveals accelerated evolution of genes relevant to hematophagy |journal=[[BMC Genomics]] |volume=10 |issue=1 |page=57 |doi=10.1186/1471-2164-10-57 |pmc=2644710 |pmid=19178717 }}</ref> |
|||
<ref name="Dixit2010">{{cite journal |author=Jyotsana Dixit, Hemlata Srivastava, Meenu Sharma, Manoj K. Das, O.P. Singh, K. Raghavendra, Nutan Nanda, Aditya P. Dash, D. N. Saksena & Aparup Das |year=2010 |title=Phylogenetic inference of Indian malaria vectors from multilocus DNA sequences |journal=[[Infection, Genetics and Evolution]] |volume=10 |issue=6 |pages=755–763 |pmid=20435167 |doi=10.1016/j.meegid.2010.04.008}}</ref> |
|||
}} |
|||
==External links== |
==External links== |
||
* [http://www.anobase.org/ ''Anopheles'' Database] |
* [http://www.anobase.org/ ''Anopheles'' Database] |
||
* [http://agambiae.vectorbase.org/ ''Anopheles gambiae'' Genome and Related Data] |
* [http://agambiae.vectorbase.org/ ''Anopheles gambiae'' Genome and Related Data] |
||
* [http://www.cdc.gov/Malaria/ CDC |
* [http://www.cdc.gov/Malaria/ CDC – National Center for Infectious Diseases, Division of Parasitic Diseases; '' Malaria] |
||
* [http://www.cdc.gov/Malaria/biology/mosquito/map.htm CDC |
* [http://www.cdc.gov/Malaria/biology/mosquito/map.htm CDC – World map showing distribution of various ''Anopheles'' species] |
||
*[http://www.wrbu.org/ Walter Reed Biosystematics Unit.] |
*[http://www.wrbu.org/ Walter Reed Biosystematics Unit.] – Links to the online mosquito catalog, keys for mosquito identification, images and information on medically important species and much more. |
||
* [http://www.map.ox.ac.uk Malaria Atlas Project] |
* [http://www.map.ox.ac.uk Malaria Atlas Project] |
||
* [http://www.metapathogen.com/mosquito/anopheles ''Anopheles gambiae'' taxonomy, facts and life cycle] |
* [http://www.metapathogen.com/mosquito/anopheles ''Anopheles gambiae'' taxonomy, facts and life cycle] |
||
* [http://entomology.ifas.ufl.edu/creatures/aquatic/Anopheles_quadrimaculatus.htm ''Anopheles quadrimaculatus'', common malaria mosquito] on the [[University of Florida]] / [[Institute of Food and Agricultural Sciences]] ''Featured Creatures'' website |
* [http://entomology.ifas.ufl.edu/creatures/aquatic/Anopheles_quadrimaculatus.htm ''Anopheles quadrimaculatus'', common malaria mosquito] on the [[University of Florida]] / [[Institute of Food and Agricultural Sciences]] ''Featured Creatures'' website |
||
| ⚫ | |||
| ⚫ | |||
[[Category:Anopheles| ]] |
[[Category:Anopheles| ]] |
||
Revision as of 22:42, 14 July 2012
| Anopheles | |
|---|---|

| |
| Anopheles stephensi | |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Diptera |
| Family: | Culicidae |
| Subfamily: | Anophelinae |
| Genus: | Anopheles |
| Species | |
|
For a full description, see section Systematics and the main article: Taxonomy of Anopheles | |
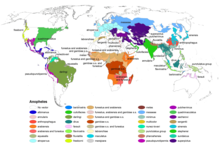
| |
| Anopheles range map | |
Anopheles (/[invalid input: 'icon']əˈnɒf[invalid input: 'ɨ']liːz/)[1] is a genus of mosquito. There are approximately 460 recognized species: while over 100 can transmit human malaria, only 30–40 commonly transmit parasites of the genus Plasmodium, which cause malaria in humans in endemic areas. Anopheles gambiae is one of the best known, because of its predominant role in the transmission of the most dangerous malaria parasite species (to humans) – Plasmodium falciparum.
The name comes from the Greek αν, an, meaning not, and όφελος, óphelos, meaning profit, and translates to useless.[1]
Some species of Anopheles also can serve as the vectors for canine heartworm Dirofilaria immitis, the filariasis-causing species Wuchereria bancrofti and Brugia malayi, and viruses such as one that causes O'nyong'nyong fever. There is an association of brain tumor incidence and malaria, suggesting that the Anopheles might transmit a virus or other agent that could cause a brain tumor.[2]
Mosquitoes in other genera (Aedes, Culex) can also serve as vectors of disease agents, but not malaria.
Evolution
The ancestors of Drosophila and mosquitoes diverged 260 million years ago ago.[3] The culicine and Anopheles clades of mosquitoes diverged between 120 million years ago and 150 million years ago.[3][4] The Old and New World Anopheles species subsequently diverged between 80 million years ago and 95 million years ago.[3][4] The Anopheles gambiae and Anopheles funestus clades diverged 80 to 36 million years ago. A molecular study of several genes in seven species has provided additional support for an expansion of this genus during the Cretaceous period.[5]
The Anopheles genome (230–284 Mb) is comparable in size to that of Drosophila but considerably smaller than those found in other culicine genomes (528 Mb–1.9 Gb). Like most culicine species, the genome is diploid with six chromosomes.
The only known fossils of this genus are those of Anopheles (?Nyssorhynchus) dominicanus Zavortink & Poinar contained in Dominican amber from the Late Eocene (40.4 to 33.9 million years ago) and Anopheles rottensis Statz contained in German amber from the Late Oligocene (28.4 to 23 million years ago).
Systematics
The genus Anopheles belongs to a subfamily Anophelinae with three genera: Anopheles Meigen (nearly worldwide distribution), Bironella Theobald (Australia only) and Chagasia Cruz (Neotropics). Bironella appears to be the sister taxon to the Anopheles with Chagasia forming the outgroup in this subfamily.
The classification of this genus began in 1901 with Theobald. Despite the passage of time the taxonomy remains incompletely settled. Classification into species is based on morphological characteristics – wing spots, head anatomy, larval and pupal anatomy, chromosome structure – and more recently on DNA sequences.
The genus itself has been subdivided into seven subgenera based primarily on the number and positions of specialized setae on the gonocoxites of the male genitalia. The system of subgenera originated with the work of Christophers who in 1915 described three subgenera: Anopheles (widely distributed), Myzomyia (later renamed Cellia) (Old World) and Nyssorhynchus (Neotropical). Nyssorhynchus was first described as Lavernia by Theobald. Edwards in 1932 added the subgenus Stethomyia (Neotropical distribution). Kerteszia was also described by Edwards in 1932 but then recognised as a subgrouping of Nyssorhynchus. It was elevated to subgenus status by Komp in 1937 and it is also found in the Neotropics. Two additional subgenera have since been recognised: Baimaia (Southeast Asia only) by Harbach et al. in 2005 and Lophopodomyia (Neotropical) by Antunes in 1937.
Within the genus Anopheles there are two main groupings: one formed by the Cellia and Anopheles subgenera and a second by Kerteszia, Lophopodomyia and Nyssorhynchus. Subgenus Stethomyia is an outlier with respect to these two taxa. Within the second group Kerteszia and Nyssorhynchus appear to be sister taxa.
The number of species currently recognised within the subgenera is given here in parentheses: Anopheles (206 species), Baimaia (1), Cellia (216), Kerteszia (12), Lophopodomyia (6), Nyssorhynchus (34) and Stethomyia (5).
Taxonomic units between subgenus and species are not currently recognised as official zoological names. In practice a number of taxonomic levels have been introduced. The larger subgenera (Anopheles, Cellia and Nyssorhynchus) have been subdivided into sections and series which in turn have been divided into groups and subgroups. Below subgroup but above species level is the species complex. Taxonomic levels above species complex can be distinguished on morphological grounds. Species within a species complex are either morphologically identical or extremely similar and can only be reliably separated by microscopic examination of the chromosomes or DNA sequencing. The classification continues to be revised.
Subgenus Nyssorhynchus has been divided in three sections: Albimanus (19 species), Argyritarsis (11 species) and Myzorhynchella (4 species). The Argyritarsis section has been sub divided into Albitarsis and Argyritarsis groups.
The Anopheles Group was divided by Edwards into four series: Anopheles (worldwide), Myzorhynchus (Palearctic, Oriental, Australasian and Afrotropical), Cycloleppteron (Neotropical) and Lophoscelomyia (Oriental); and two groups, Arribalzagia (Neotropical) and Christya (Afrotropical). Reid and Knight (1961) modified this classification and consequently subdivided the subgenus Anopheles into two sections, Angusticorn and Laticorn and six series. The Arribalzagia and Christya Groups were considered to be series. The Laticorn Section includes the Arribalzagia (24 species), Christya and Myzorhynchus Series. The Angusticorn Section includes members of the Anopheles, Cycloleppteron and Lophoscelomyia Series.
All species known to carry human malaria lie within either the Myzorhynchus or the Anopheles Series.
Life stages
Like all mosquitoes, anophelines go through four stages in their life cycle: egg, larva, pupa, and imago. The first three stages are aquatic and last 5–14 days, depending on the species and the ambient temperature. The adult stage is when the female Anopheles mosquito acts as malaria vector. The adult females can live up to a month (or more in captivity) but most probably do not live more than 1–2 weeks in nature.
Eggs
Adult females lay 50–200 eggs per oviposition. The eggs are quite small (~0.5 × 0.2 mm). Eggs are laid singly and directly on water. They are unique in that they have floats on either side. Eggs are not resistant to drying and hatch within 2–3 days, although hatching may take up to 2–3 weeks in colder climates.
Larvae


Mosquito larvae have a well-developed head with mouth brushes used for feeding, a large thorax and a nine segmented abdomen. They have no legs. In contrast to other mosquitoes, Anopheles larvae lack a respiratory siphon and for this reason position themselves so that their body is parallel to the surface of the water. In contrast, feeding larvae of non-anopheline mosquito species attach themselves to the water surface with their posterior siphon, with their body pointing downwards.
Larvae breathe through spiracles located on the 8th abdominal segment and therefore must come to the surface frequently. The larvae spend most of their time feeding on algae, bacteria, and other microorganisms in the surface microlayer. They dive below the surface only when disturbed. Larvae swim either by jerky movements of the entire body or through propulsion with the mouth brushes.
Larvae develop through 4 stages, or instars, after which they metamorphose into pupae. At the end of each instar, the larvae molt, shedding their exoskeleton, or skin, to allow for further growth. 1st stage larvae are ~1 mm in length; 4th stage larvae are normally 5–8 mm in length.
The process from egg laying to emergence of the adult is temperature dependent, with a minimum time of 7 days.
The larvae occur in a wide range of habitats but most species prefer clean, unpolluted water. Larvae of Anopheles mosquitoes have been found in fresh- or salt-water marshes, mangrove swamps, rice fields, grassy ditches, the edges of streams and rivers, and small, temporary rain pools. Many species prefer habitats with vegetation. Others prefer habitats that have none. Some breed in open, sun-lit pools while others are found only in shaded breeding sites in forests. A few species breed in tree holes or the leaf axils of some plants.
Pupae
The pupa is comma-shaped when viewed from the side. The head and thorax are merged into a cephalothorax with the abdomen curving around underneath. As with the larvae, pupae must come to the surface frequently to breathe, which they do through a pair of respiratory trumpets on the cephalothorax. After a few days as a pupa, the dorsal surface of the cephalothorax splits and the adult mosquito emerges. The Pupa stage lasts around 2–3 days in temperate areas.
Adults

The duration from egg to adult varies considerably among species and is strongly influenced by ambient temperature. Mosquitoes can develop from egg to adult in as little as 5 days but usually take 10–14 days in tropical conditions.
Like all mosquitoes, adult Anopheles have slender bodies with 3 sections: head, thorax and abdomen.
The head is specialized for acquiring sensory information and for feeding. The head contains the eyes and a pair of long, many-segmented antennae. The antennae are important for detecting host odors as well as odors of breeding sites where females lay eggs. The head also has an elongated, forward-projecting proboscis used for feeding, and two maxillary palps. These palps also carry the receptors for carbon dioxide, a major attractant for the location of the mosquito's host.
The thorax is specialized for locomotion. Three pairs of legs and a pair of wings are attached to the thorax.
The abdomen is specialized for food digestion and egg development. This segmented body part expands considerably when a female takes a blood meal. The blood is digested over time serving as a source of protein for the production of eggs, which gradually fill the abdomen.
Anopheles mosquitoes can be distinguished from other mosquitoes by the palps, which are as long as the proboscis, and by the presence of discrete blocks of black and white scales on the wings. Adult Anopheles can also be identified by their typical resting position: males and females rest with their abdomens sticking up in the air rather than parallel to the surface on which they are resting.
Adult mosquitoes usually mate within a few days after emerging from the pupal stage. In most species, the males form large swarms, usually around dusk, and the females fly into the swarms to mate.
Males live for about a week, feeding on nectar and other sources of sugar. Females will also feed on sugar sources for energy but usually require a blood meal for the development of eggs. After obtaining a full blood meal, the female will rest for a few days while the blood is digested and eggs are developed. This process depends on the temperature but usually takes 2–3 days in tropical conditions. Once the eggs are fully developed, the female lays them and resumes host seeking.
The cycle repeats itself until the female dies. While females can live longer than a month in captivity, most do not live longer than 1–2 weeks in nature. Their lifespan depends on temperature, humidity, and also their ability to successfully obtain a blood meal while avoiding host defenses.
Habitat
Although malaria is nowadays limited to tropical areas, most notoriously regions of sub-Saharan Africa, many Anopheles species live in colder latitudes (see this map from the CDC). Indeed, malaria outbreaks have, in the past, occurred in colder climates, for example during the construction of the Rideau Canal in Canada during the 1820s.[6] Since then, the Plasmodium parasite (not the Anopheles mosquito) has been eliminated from first world countries.
The CDC warns, however, that "Anopheles that can transmit malaria are found not only in malaria-endemic areas, but also in areas where malaria has been eliminated. The latter areas are thus constantly at risk of re-introduction of the disease."
Susceptibility to become a vector of disease
Some species are poor vectors of malaria, as the parasites do not develop well (or at all) within them. There is also variation within species. In the laboratory, it has been possible to select strains of A. gambiae that are refractory to infection by malaria parasites. These refractory strains have an immune response that encapsulates and kills the parasites after they have invaded the mosquito's stomach wall. Scientists are studying the genetic mechanism for this response. It is hoped that some day, genetically modified mosquitoes that are refractory to malaria can replace wild mosquitoes, thereby limiting or eliminating malaria transmission.
Malaria transmission and control
Understanding the biology and behavior of Anopheles mosquitoes can help understand how malaria is transmitted and can aid in designing appropriate control strategies. Factors that affect a mosquito's ability to transmit malaria include its innate susceptibility to Plasmodium, its host choice and its longevity. Factors that should be taken into consideration when designing a control program include the susceptibility of malaria vectors to insecticides and the preferred feeding and resting location of adult mosquitoes.
On December 21, 2007, a study published in PLoS Pathogens found that the hemolytic C-type lectin CEL-III from Cucumaria echinata, a sea cucumber found in the Bay of Bengal, impaired the development of the malaria parasite when produced by transgenic A. stephensi.[7] This could potentially be used one day to control malaria by spreading genetically modified mosquitoes refractory to the parasites, although there are numerous scientific and ethical issues to be overcome before such a control strategy could be implemented.
Preferred sources for blood meals
One important behavioral factor is the degree to which an Anopheles species prefers to feed on humans (anthropophily) or animals such as cattle (zoophily). Anthropophilic Anopheles are more likely to transmit the malaria parasites from one person to another. Most Anopheles mosquitoes are not exclusively anthropophilic or zoophilic. However, the primary malaria vectors in Africa, A. gambiae and A. funestus, are strongly anthropophilic and, consequently, are two of the most efficient malaria vectors in the world.
Once ingested by a mosquito, malaria parasites must undergo development within the mosquito before they are infectious to humans. The time required for development in the mosquito (the extrinsic incubation period) ranges from 10–21 days, depending on the parasite species and the temperature. If a mosquito does not survive longer than the extrinsic incubation period, then she will not be able to transmit any malaria parasites.
It is not possible to measure directly the life span of mosquitoes in nature. But indirect estimates of daily survivorship have been made for several Anopheles species. Estimates of daily survivorship of A. gambiae in Tanzania ranged from 0.77 to 0.84, meaning that at the end of one day between 77% and 84% will have survived.[8]
Assuming this survivorship is constant through the adult life of a mosquito, less than 10% of female A. gambiae would survive longer than a 14-day extrinsic incubation period. If daily survivorship increased to 0.9, over 20% of mosquitoes would survive longer than a 14-day extrinsic incubation period. Control measures that rely on insecticides (e.g. indoor residual spraying) may actually impact malaria transmission more through their effect on adult longevity than through their effect on the population of adult mosquitoes.
Patterns of feeding and resting
Most Anopheles mosquitoes are crepuscular (active at dusk or dawn) or nocturnal (active at night). Some Anopheles mosquitoes feed indoors (endophagic) while others feed outdoors (exophagic). After feeding, some blood mosquitoes prefer to rest indoors (endophilic) while others prefer to rest outdoors (exophilic), though this can differ regionally based on local vector ecotype, and vector chromosomal makeup, as well as housing type and local microclimatic conditions. Biting by nocturnal, endophagic Anopheles mosquitoes can be markedly reduced through the use of insecticide-treated bed nets (ITNs) or through improved housing construction to prevent mosquito entry (e.g. window screens). Endophilic mosquitoes are readily controlled by indoor spraying of residual insecticides. In contrast, exophagic/exophilic vectors are best controlled through source reduction (destruction of the breeding sites).
Gut flora
Because transmission of disease by the mosquito requires ingestion of blood it is reasonable to presuppose that the gut flora may have a bearing on the success of infection of the mosquito host. This aspect of disease transmission has not been investigated until recently.[9] The larval and pupal gut is largely colonised by photosynthetic cyanobacteria while in the adult Proteobacteria and Bacteroidetes predominate. Blood meals drastically reduce the diversity of organisms and favor enteric bacteria.
Insecticide resistance
Insecticide-based control measures (e.g. indoor spraying with insecticides, ITNs) are the principal way to kill mosquitoes that bite indoors. However, after prolonged exposure to an insecticide over several generations, mosquitoes, like other insects, may develop resistance, a capacity to survive contact with an insecticide. Since mosquitoes can have many generations per year, high levels of resistance can arise very quickly. Resistance of mosquitoes to some insecticides has been documented with just within a few years after the insecticides were introduced. There are over 125 mosquito species with documented resistance to one or more insecticides. The development of resistance to insecticides used for indoor residual spraying was a major impediment during the Global Malaria Eradication Campaign. Judicious use of insecticides for mosquito control can limit the development and spread of resistance. However, use of insecticides in agriculture has often been implicated as contributing to resistance in mosquito populations. It is possible to detect developing resistance in mosquitoes and control programs are well advised to conduct surveillance for this potential problem.
Eradication
With substantial numbers of malaria cases affecting people around the globe, in tropical and subtropical regions, especially in sub-Saharan Africa, where millions of children are killed by this infectious disease, eradication is back on the global health agenda.[10]
Although malaria has existed since old times, its eradication was possible in Europe, North America, the Caribbean and parts of Asia and southern Central America during the first regional elimination campaigns in the late 1940s. However, the same results were not achieved in sub-Saharan Africa.[10]
Even though the World Health Organization adopted a formal policy on the control and eradication of the malaria parasite since 1955,[11] it was recently, after the Gates Malaria Forum in October 2007, that key organizations started the debate on the pros and cons of redefining eradication as a goal to control malaria.
Clearly, the cost of preventing malaria is much less than treating the disease, in the long run. However, eradication of mosquito is not an easy task. For effective prevention of malaria, some conditions should be met such as conducive conditions in the country, data collection about the disease, targeted technical approach to the problem, very active and committed leadership, government’s total support, monetary free hand, community involvement, skilled technicians from different fields as well as an adequate implementation.[12]
There is a wide range of strategies to achieve malaria eradication that start from simple steps to complicated strategies which may not be possible to enforce with the current tools.
Although mosquito control is an important component of malaria control strategy, elimination of malaria in an area does not require the elimination of all Anopheles mosquitoes. For instance, in North America and Europe, although the vector Anopheles mosquitoes are still present, the parasite has been eliminated. There are also some socioeconomic improvements (e.g., houses with screened windows, air conditioning) that once combined with vector reduction efforts and effective treatment lead to the elimination of malaria without the complete elimination of the vectors. Some important measures in mosquito control to be followed are: discourage egg laying, prevent development of eggs into larvae and adults, kill the adult mosquitoes, do not allow adult mosquitoes into places of human dwelling, prevent mosquitoes from biting human beings and deny blood meal.[13]
Research in this sense continues, and a study has suggested that sterile mosquitoes might be the answer to malaria elimination. This research suggests that using the sterile insect technique (SIT), in which sexually sterile male insects are released to wipe out a pest population, could be a solution to the problem of malaria in Africa. This technique brings hope, as female mosquitoes only mate once during their lifetimes, and in doing so with sterile male mosquitoes, the insect population would decrease.[14] This is another option to be considered by local and international authorities that may be combined with other methods and tools to achieve malaria eradication in sub-Saharan Africa.
Parasites
A number of parasites of this genus are known to exist including microsporidia of the genera Amblyospora, Crepidulospora, Senoma and Parathelohania.[15]
Microsporida infecting the aquatic stages of insects, a group that includes mosquitoes and black flies, and copepods appear to form a distinct clade from those infecting terrestrial insects and fish. There are two distinct life cycles in this group: in the first type the parasite is transmitted by the oral route and is relatively non species specific. In the second, while again the oral route is the usual route of infection, the parasite is ingested within an already infected intermediate host. Infection of the insect larval form is frequently tissue specific, and commonly involves the fat body. Vertical (transovarial) transmission is also known to occur.
Few phylogenetic studies of these parasites have been done, and their the relationship to their mosquito hosts is still being determined. One study suggested Parathelohania is an early diverging genus within this group.[16]
The parasite Wolbachia has been studied for use as a control agent.[17]
See also
References
- ^ a b Anopheles at dictionary.com.
- ^ Steven Lehrer (2010). "Anopheles mosquito transmission of brain tumor" (PDF). Medical Hypotheses. 74 (1): 167–168. doi:10.1016/j.mehy.2009.07.005. PMID 19656635.
- ^ a b c Moreno M, Marinotti O, Krzywinski J, Tadei WP, James AA, Achee NL, Conn JE (2010). "Complete mtDNA genomes of Anopheles darlingi and an approach to anopheline divergence time". Malaria journal. 9: 127. doi:10.1186/1475-2875-9-127. PMC 2877063. PMID 20470395.
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: unflagged free DOI (link) - ^ a b Eric Calvo, Van M Pham, Osvaldo Marinotti, John F. Andersen & José M. C. Ribeiro (2009). "The salivary gland transcriptome of the neotropical malaria vector Anopheles darlingi reveals accelerated evolution of genes relevant to hematophagy". BMC Genomics. 10 (1): 57. doi:10.1186/1471-2164-10-57. PMC 2644710. PMID 19178717.
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: unflagged free DOI (link) - ^ Jyotsana Dixit, Hemlata Srivastava, Meenu Sharma, Manoj K. Das, O.P. Singh, K. Raghavendra, Nutan Nanda, Aditya P. Dash, D. N. Saksena & Aparup Das (2010). "Phylogenetic inference of Indian malaria vectors from multilocus DNA sequences". Infection, Genetics and Evolution. 10 (6): 755–763. doi:10.1016/j.meegid.2010.04.008. PMID 20435167.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ William N. T. Wylie (1983). "Poverty, Distress, and Disease: Labour and the Construction of the Rideau Canal, 1826–32". Labour/Le Travail. 11: 7–29. doi:10.2307/25140199. JSTOR 25140199.
- ^ Shigeto Yoshida, Yohei Shimada , Daisuke Kondoh, Yoshiaki Kouzuma, Anil K. Ghosh, Marcelo Jacobs-Lorena & Robert E. Sinden (2007). "Hemolytic C-type lectin CEL-III from sea cucumber expressed in transgenic mosquitoes impairs malaria parasite development". PLoS Pathogens. 3 (12): e192. doi:10.1371/journal.ppat.0030192. PMC 2151087. PMID 18159942.
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: unflagged free DOI (link) - ^ J. D. Charlwood, T. Smith, P. F. Billingsley, W. Takken, E. O. K. Lyimo & J. H. E. T. Meuwissen (1997). "Survival And infection probabilities of anthropophagic anophelines from an area of high prevalence of Plasmodium falciparum in humans". Bulletin of Entomological Research. 87 (5): 445–453. doi:10.1017/S0007485300041304.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Wang Y, Gilbreath TM 3rd, Kukutla P, Yan G, Xu J (2011). Leulier, François (ed.). "Dynamic gut microbiome across life history of the malaria mosquito Anopheles gambiae in Kenya". PLoS ONE. 6 (9): e24767. doi:10.1371/journal.pone.0024767. PMC 3177825. PMID 21957459.
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: numeric names: authors list (link) CS1 maint: unflagged free DOI (link) - ^ a b Marcel Tanner & Don de kSavigny (2008). "Malaria eradication back on the table". Bulletin of the World Health Organization. 86 (2): 82–83. doi:10.2471/BLT.07.050633.
- ^ "Malaria Eradication". Retrieved 2010-05-04.
- ^ "Mosquito Eradication". Archived from the original on 3 May 2010. Retrieved 2010-05-04.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ "Mosquito Control". Archived from the original on May 1, 2008. Retrieved 2010-05-04.
- ^ "Are sterile mosquitoes the answer to malaria elimination?". Archived from the original on 30 April 2010. Retrieved 2010-05-04.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ A. V. Simakova & T. F. Pankova (2008). "Ecology and epizootology of microsporidia in malarial mosquitoes (Diptera: Culicidae) from the south of western Siberia". Parazitologiia (in Russian). 42 (2): 139–150. PMID 18664069.
- ^ Michael D. Baker, Charles R. Vossbrinck, James J. Becnel & Theodore G. Andreadis (1998). "Phylogeny of Amblyospora (Microsporida: Amblyosporidae) and related genera based on small subunit ribosomal DNA data: a possible example of host parasite cospeciation" (PDF). Journal of Invertebrate Pathology. 71 (3): 199–206. doi:10.1006/jipa.1997.4725. PMID 9538024.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Mosquito Parasite Fights Infectious Disease : Discovery News. News.discovery.com (2009-10-01). Retrieved on 2012-07-14.
External links
- Anopheles Database
- Anopheles gambiae Genome and Related Data
- CDC – National Center for Infectious Diseases, Division of Parasitic Diseases; Malaria
- CDC – World map showing distribution of various Anopheles species
- Walter Reed Biosystematics Unit. – Links to the online mosquito catalog, keys for mosquito identification, images and information on medically important species and much more.
- Malaria Atlas Project
- Anopheles gambiae taxonomy, facts and life cycle
- Anopheles quadrimaculatus, common malaria mosquito on the University of Florida / Institute of Food and Agricultural Sciences Featured Creatures website
- Original version from http://www.cdc.gov/malaria/biology/mosquito/
- http://animaldiversity.ummz.umich.edu/site/accounts/classification/Anopheles.html
