Zineb: Difference between revisions
m →Structure: clean up punctuation and spacing issues, primarily spacing around commas, replaced: , → , using AWB |
upgrade refs to make them get-able |
||
| Line 43: | Line 43: | ||
| AutoignitionPt = }} |
| AutoignitionPt = }} |
||
}} |
}} |
||
'''Zineb''' is the chemical compound with the formula {Zn[S<sub>2</sub>CN(H)CH<sub>2</sub>CH<sub>2</sub>N(H)CS<sub>2</sub>]}<sub>n</sub>. Structurally, it is classified as a [[coordination polymer]]. This pale yellow solid is used as [[fungicide]]. |
'''Zineb''' is the chemical compound with the formula {Zn[S<sub>2</sub>CN(H)CH<sub>2</sub>CH<sub>2</sub>N(H)CS<sub>2</sub>]}<sub>n</sub>. Structurally, it is classified as a [[coordination polymer]]. This pale yellow solid is used as [[fungicide]].<ref name=Ullmann>{{cite encyclopedia|authors=Franz Müller, Peter Ackermann, Paul Margot|title=Fungicides, Agricultural, 2. Individual Fungicides|encyclopedia=Ullmann's Encyclopedia of Industrial Chemistry|publisher=Wiley-VCH|place=Weinheim|year=2012|doi=10.1002/14356007.o12_o06}}</ref> |
||
==Production and applications== |
==Production and applications== |
||
It is produced by treating ethylene bis(dithiocarbamate) sodium salt, "nabam", with [[zinc sulfate]]. This procedure can be carried out by mixing nabam and zinc sulfate in a spray tank.<ref name = "Kamrin">Michael A. Kamrin, (1997) Pesticide Profiles: Toxicity, Environmental Impact, and Fate, CRC Press, {{ISBN|1-56670-190-2}}{{page needed|date=March 2015}}</ref> |
It is produced by treating ethylene bis(dithiocarbamate) sodium salt, "nabam", with [[zinc sulfate]]. This procedure can be carried out by mixing nabam and zinc sulfate in a spray tank.<ref name = "Kamrin">Michael A. Kamrin, (1997) Pesticide Profiles: Toxicity, Environmental Impact, and Fate, CRC Press, {{ISBN|1-56670-190-2}}{{page needed|date=March 2015}}</ref> |
||
Its uses include control of downy [[mildew]]s, [[rust (botany)|rusts]] and redfire disease.<ref name = " |
Its uses include control of downy [[mildew]]s, [[rust (botany)|rusts]], and redfire disease.<ref name = "Ullmann"/> In the US it was once registered as a "General Use Pesticide", however all registrations were voluntarily cancelled following an [[United States Environmental Protection Agency|EPA]] special review.<ref name = "Kamrin"/> It continues to be used in many other countries. |
||
==Structure== |
==Structure== |
||
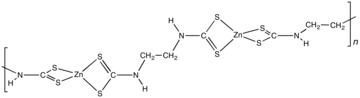
Zineb is a polymeric [[complex (chemistry)|complex]] of [[zinc]] with |
Zineb is a polymeric [[complex (chemistry)|complex]] of [[zinc]] with a [[dithiocarbamate]].<ref name=Ullmann/> The polymer is composed of Zn(dithiocarbamate)<sub>2</sub> subunits linked by an ethylene (-CH<sub>2</sub>CH<sub>2</sub>-) backbone.<ref>{{cite book|book=Residue Reviews|pages=45-6|title=Residues of dithiocarbamate fungicides and their metabolites on plant foods |
||
|authors=R. Engst, W. Schnaak|year=1974|editor=Gunther F.A.|volume=52|publisher=Springer|place=New York, NY|doi=10.1007/978-1-4615-8504-6_3}}</ref> A reference compound is [Zn(S<sub>2</sub>CNEt<sub>2</sub>)<sub>2</sub>]<sub>2</sub>, which features a pair of tetrahedral Zn centers [[bridging ligand|bridged]] by one sulfur center.<ref>Bonamico, M.; Mazzone, G.; Vaciago, A.; Zambonelli, L., "Structural studies of metal dithiocarbamates. III. The Crystal and Molecular Structure of Zinc Diethyldithiocarbamate", Acta Crystallogr. 1965, volume 19, pp. 898-909. {{DOI|10.1107/S0365110X65004620}}</ref> |
|||
==See also== |
==See also== |
||
Revision as of 18:11, 3 March 2018
| |
| Names | |
|---|---|
| IUPAC name
zinc ethane-1,2-diylbis(dithiocarbamate)
| |
| Other names
[[1,2 ethanediylbis[dithiocarbamodithioato](2−)]] zinc,
Dithane Z-78, Aphytora, Amitan | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.031.970 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H6N2S4Zn | |
| Molar mass | 275.8 g/mol (monomer) |
| Appearance | pale yellow powder |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Zineb is the chemical compound with the formula {Zn[S2CN(H)CH2CH2N(H)CS2]}n. Structurally, it is classified as a coordination polymer. This pale yellow solid is used as fungicide.[1]
Production and applications
It is produced by treating ethylene bis(dithiocarbamate) sodium salt, "nabam", with zinc sulfate. This procedure can be carried out by mixing nabam and zinc sulfate in a spray tank.[2] Its uses include control of downy mildews, rusts, and redfire disease.[1] In the US it was once registered as a "General Use Pesticide", however all registrations were voluntarily cancelled following an EPA special review.[2] It continues to be used in many other countries.
Structure
Zineb is a polymeric complex of zinc with a dithiocarbamate.[1] The polymer is composed of Zn(dithiocarbamate)2 subunits linked by an ethylene (-CH2CH2-) backbone.[3] A reference compound is [Zn(S2CNEt2)2]2, which features a pair of tetrahedral Zn centers bridged by one sulfur center.[4]
See also
- Metam sodium - A related dithiocarbamate salt which is also used as a fungicide
- Maneb - ethylene bis(dithiocarbamate) with manganese instead of zinc
References
- ^ a b c "Fungicides, Agricultural, 2. Individual Fungicides". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. 2012. doi:10.1002/14356007.o12_o06.
{{cite encyclopedia}}: Cite uses deprecated parameter|authors=(help) - ^ a b Michael A. Kamrin, (1997) Pesticide Profiles: Toxicity, Environmental Impact, and Fate, CRC Press, ISBN 1-56670-190-2[page needed]
- ^ Gunther F.A., ed. (1974). Residues of dithiocarbamate fungicides and their metabolites on plant foods. Vol. 52. New York, NY: Springer. pp. 45–6. doi:10.1007/978-1-4615-8504-6_3.
{{cite book}}: Cite uses deprecated parameter|authors=(help); Unknown parameter|book=ignored (help) - ^ Bonamico, M.; Mazzone, G.; Vaciago, A.; Zambonelli, L., "Structural studies of metal dithiocarbamates. III. The Crystal and Molecular Structure of Zinc Diethyldithiocarbamate", Acta Crystallogr. 1965, volume 19, pp. 898-909. doi:10.1107/S0365110X65004620
External links
- Zineb in the Pesticide Properties DataBase (PPDB)
