Benzoyl group
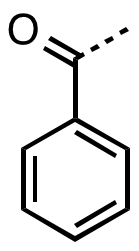
In organic chemistry, benzoyl (/ˈbɛnzoʊɪl/, BENZ-oh-il)[1] is the functional group with the formula −COC6H5 and structure −C(=O)−C6H5.[2][3] It can be viewed as benzaldehyde missing one hydrogen. The benzoyl group has a mass of 105 amu.
The term "benzoyl" should not be confused with benzyl, which has the formula −CH2−C6H5. The benzoyl group is given the symbol "Bz" whereas benzyl is commonly abbreviated "Bn".
Sources
[edit]Benzoyl chloride is a favored source of benzoyl groups, being used to prepare benzoyl ketones, benzamides (benzoyl amides), and benzoate esters. The source of many naturally occurring benzoyl compounds is the thioester benzoyl-CoA. Irradiation of benzil generates benzoyl radicals, which have the formula PhCO.
Benzoyl compounds
[edit]Many ketones contain the benzoyl group. They have the formula C6H5CO–R, an important example being benzophenone.
Benzoyl esters and amides are common in organic chemistry. The esters are used as a protecting groups in organic synthesis,[4] which can be easily removed by hydrolysis in dilute basic solution. Benzoyl-β-D-glucoside is a natural substance that can be found in Pteris ensiformis.
References
[edit]- ^ "benzoyl | Definition of benzoyl in English by Oxford Dictionaries". Oxford Dictionaries | English. Archived from the original on September 28, 2016. Retrieved 2018-02-02.
- ^ Maki, Takao; Takeda, Kazuo. "Benzoic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a03_555. ISBN 978-3527306732..
- ^ Morris, Christopher G. (1992). Academic Press Dictionary of Science and Technology. Gulf Professional Publishing. p. 246. ISBN 9780122004001.
- ^ Blackburn, G. Michael (2006). Nucleic Acids in Chemistry and Biology. Royal Society of Chemistry. p. 145. ISBN 9780854046546.
