A-type proanthocyanidin: Difference between revisions
| Line 11: | Line 11: | ||
* [[Procyanidin A2]] is a dimeric (-)epicatechin. |
* [[Procyanidin A2]] is a dimeric (-)epicatechin. |
||
Other A-type proanthocyanidins can be found in [[cranberry|cranberries]],<ref name="Neto"/> in [[apricot]]s,in [[cinnamon]] <ref>New identification of proanthocyanidins in cinnamon (Cinnamomum zeylanicum L.) using MALDI-TOF/TOF mass spectrometry |
Other A-type proanthocyanidins can be found in [[cranberry|cranberries]],<ref name="Neto"/> in [[apricot]]s,in [[cinnamon]] <ref name="María Luisa Mateos-Martín">{{cite journal | url = http://link.springer.com/article/10.1007/s00216-011-5557-3 | title = New identification of proanthocyanidins in cinnamon (Cinnamomum zeylanicum L.) using MALDI-TOF/TOF mass spectrometry | author = María Luisa Mateos-Martín, Elisabet Fuguet, Carmen Quero, Jara Pérez-Jiménez, Josep Lluís Torres. | year = 2012 | journal = Analytical and bioanalytical chemistry | volume = 402 | issue = 3 | pages = 1327-1336 | doi = 10.1007/s00216-011-5557 }}</ref>, in ''Prunus armeniaca'' (ent-epiafzelechin-3-O-p-hydroxybenzoate-(4α→8,2α→O→7)-[[epiafzelechin]]) and in peanut skins ([[epicatechin-(2β→O→7, 4β→8)-ent-epicatechin]]).<ref name="Hongxiang">{{cite journal | url = http://cat.inist.fr/?aModele=afficheN&cpsidt=1831517 | title = A-type proanthocyanidins from peanut skins | author = Hongxiang Lou; Yamazaku Y.; Sasaku T.; Uchida M.; Tanaka H.; Oka S. | year = 1999 | journal = Phytochemistry | volume = 51 | issue = 2 | pages = 297–308 | doi = 10.1016/S0031-9422(98)00736-5 }}</ref> |
||
Volumen 402,3, pages = 1327-1336| doi = 10.1007/s00216-011-5557-</ref>, in ''Prunus armeniaca'' (ent-epiafzelechin-3-O-p-hydroxybenzoate-(4α→8,2α→O→7)-[[epiafzelechin]]) and in peanut skins ([[epicatechin-(2β→O→7, 4β→8)-ent-epicatechin]]).<ref name="Hongxiang">{{cite journal | url = http://cat.inist.fr/?aModele=afficheN&cpsidt=1831517 | title = A-type proanthocyanidins from peanut skins | author = Hongxiang Lou; Yamazaku Y.; Sasaku T.; Uchida M.; Tanaka H.; Oka S. | year = 1999 | journal = Phytochemistry | volume = 51 | issue = 2 | pages = 297–308 | doi = 10.1016/S0031-9422(98)00736-5 }}</ref> |
|||
[[Epi-afzelechin-(4β→8, 2β→O→7)-afzelechin]] ([[geranin A]]) and [[epi-catechin-(4β→8, 2β→O→7)-afzelechin]] ([[geranin B]]) can be found in ''[[Geranium niveum]]''.<ref>Geranins A and B, new antiprotozoal A-type proanthocyanidins from Geranium niveum. Calzada F, Cerda-García-Rojas CM, Meckes M, Cedillo-Rivera R, Bye R and Mata R, J Nat Prod., May 1999, volume 62, issue 5, pages 705–709, {{PMID|10346950}}</ref> |
[[Epi-afzelechin-(4β→8, 2β→O→7)-afzelechin]] ([[geranin A]]) and [[epi-catechin-(4β→8, 2β→O→7)-afzelechin]] ([[geranin B]]) can be found in ''[[Geranium niveum]]''.<ref>Geranins A and B, new antiprotozoal A-type proanthocyanidins from Geranium niveum. Calzada F, Cerda-García-Rojas CM, Meckes M, Cedillo-Rivera R, Bye R and Mata R, J Nat Prod., May 1999, volume 62, issue 5, pages 705–709, {{PMID|10346950}}</ref> |
||
Revision as of 11:31, 12 September 2013
A type proanthocyanidins are a specific type of proanthocyanidins, which are a class of flavanoid. Proanthocyanidins fall under a wide range of names in the nutritional and scientific vernacular, including oligomeric proanthocyanidins, flavanoids, polyphenols, condensed tannins, and OPCs. Proanthocyanidins were first popularized by French scientist Jacques Masquelier.[1]
Distribution in plants
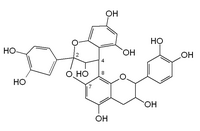
A-type linkage is a less common feature in proanthocyanidins with both 4β→8 (B-type) and 2β→O→7 interflavanoid bonds.[2]
A-type proanthocyanidin glycosides can be isolated from cocoa liquor.[3]
Dimers
- Procyanidin A1 is an epicatechin-(2β→7,4β→8)-epicatechin dimer.
- Procyanidin A2 is a dimeric (-)epicatechin.
Other A-type proanthocyanidins can be found in cranberries,[2] in apricots,in cinnamon [4], in Prunus armeniaca (ent-epiafzelechin-3-O-p-hydroxybenzoate-(4α→8,2α→O→7)-epiafzelechin) and in peanut skins (epicatechin-(2β→O→7, 4β→8)-ent-epicatechin).[5]
Epi-afzelechin-(4β→8, 2β→O→7)-afzelechin (geranin A) and epi-catechin-(4β→8, 2β→O→7)-afzelechin (geranin B) can be found in Geranium niveum.[6]
Trimers
- Selligueain A is a natural sweetener
- Selligueain B can also be isolated from the rhizomes of Selliguea feei.[7]
An A type proanthocyanidins trimer can be found in Lindera aggregata (epicatechin-(4β→8,2β→O→7)-entcatechin-(4β-8)-catechin).[8] This compound shows cytoprotective action against ethanol-induced gastric injury.[9]
In Ecdysanthera utilis (epicatechin-(4β→8,2β→O→7)-epicatechin-(4β→8)-epicatechin and epicatechin-(4β→8)-epicatechin-(4β→8,2β→O→7)-epicatechin-(4β→8)-epicatechin) can be found.[10]
Chemistry
B-type procyanidins (catechin dimers) can be converted to A-type procyanidins by radical oxidation.[11] Fragmentation patterns for A-type proanthocyanidins include heterocyclic ring fission (HRF), retro-Diels-Alder (RDA) fission, benzofuran-forming fission (BFF) and quinone methide fission (QM).[12]
Urinary tract infection prevention
85–90% of urinary tract infections (UTIs) are caused by E. Coli bacteria.[13] E. Coli use their p-type fimbrae to attach to the wall of the urinary tract.[14] A Type proanthocyanidins have been clinically demonstrated to attach to E. Coli fimbriae, preventing the bacteria from attaching to the urinary tract or bladder or urinary wall.[15][16] A Type proanthocyanidins have been demonstrated in vitro to be more effective than other types of proanthocyanidins at adhering to E. Coli and preventing UTIs.[17]
The anti-adhesion activity of A type proanthocyanidins in cranberry products has been well established by the aforementioned clinical data, but it is unclear to what extent this anti-adhesion activity translates into prevention of UTIs. A Cochrane Collaboration Review of the body of clinical literature concluded that "there is some evidence to recommend cranberry juice for the prevention of UTIs in women with symptomatic UTIs. The large number of dropouts/withdrawals from some of the trials, however, indicates that cranberry juice may not be acceptable over long periods of time".[18] Another review of cranberry and UTI prevention takes a more skeptical view. The review supports the anti-adhesion of activity of A type proanthocyanidins: "evidence suggests that the anthocyanidin/proanthocyanidin moieties are potent antiadhesion compounds" yet asserts that "most clinical trials have had design deficiencies" and points to cranberry products' "adverse events include gastrointestinal intolerance and weight gain (due to the excessive calorie load)."[19] Both reviews agree that the evidence for cranberries (and their anti-adhesion components A type proanthocyanidins) and UTI prevention can be strengthened by better-designed clinical studies.
Problems may arise with the lack of validation of a quantifying method. For instance, in the case of cranberries extracts, the quantification of A-type proanthocyanidins can be performed using several existing methods including for example the European Pharmacopoeia method, LC-MS[20] or a modified 4-dimethylaminocinnamaldehyde (DMAC) colorimetric method.[21] This can lead to a difficulty to evaluated the real quality of extracts from different origins. Assessment of that quality shows that it differs greatly from one commercial product to another.[22]
References
- ^ Fine, AM (2000). "Oligomeric proanthocyanidin complexes: history, structure, and phytopharmaceutical applications" (PDF). Alternative medicine review. 5 (2): 144–51. PMID 10767669.
- ^ a b Neto, CC (2007). "Cranberry and its phytochemicals: a review of in vitro anticancer studies". The Journal of nutrition. 137 (1 Suppl): 186S–193S. PMID 17182824.
- ^ Hatano, T; Miyatake, H; Natsume, M; Osakabe, N; Takizawa, T; Ito, H; Yoshida, T (2002). "Proanthocyanidin glycosides and related polyphenols from cacao liquor and their antioxidant effects". Phytochemistry. 59 (7): 749–58. doi:10.1016/S0031-9422(02)00051-1. PMID 11909632.
- ^ María Luisa Mateos-Martín, Elisabet Fuguet, Carmen Quero, Jara Pérez-Jiménez, Josep Lluís Torres. (2012). "New identification of proanthocyanidins in cinnamon (Cinnamomum zeylanicum L.) using MALDI-TOF/TOF mass spectrometry". Analytical and bioanalytical chemistry. 402 (3): 1327–1336. doi:10.1007/s00216-011-5557.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Hongxiang Lou; Yamazaku Y.; Sasaku T.; Uchida M.; Tanaka H.; Oka S. (1999). "A-type proanthocyanidins from peanut skins". Phytochemistry. 51 (2): 297–308. doi:10.1016/S0031-9422(98)00736-5.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Geranins A and B, new antiprotozoal A-type proanthocyanidins from Geranium niveum. Calzada F, Cerda-García-Rojas CM, Meckes M, Cedillo-Rivera R, Bye R and Mata R, J Nat Prod., May 1999, volume 62, issue 5, pages 705–709, PMID 10346950
- ^ Baek Nam-In; Kennelly E.J.; Kardono L.B.S.; Tsauri S.; Padmawinata K.; Soejarto D.D.; Kinghorn A.D. (1994). "Flavonoids and a proanthrocyanidin from rhizomes of Selliguea feei". Phytochemistry. 36 (2): 513–518. doi:10.1016/S0031-9422(00)97105-X.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Feng, Zhang Chao; Shi, Sun Qi; Tao, Wang Zheng; Hattori, Masao (2003). "One New A-type Proanthocyanidin Trimer from Lindera aggregata (Sims) Kosterm" (PDF). Chinese Chemical Letters. 14 (10): 1033–1036.
- ^ M. Zhu; C. T. Luk; T. H. Lew (1998). "Cytoprotective Effect of Lindera aggregata Roots Against Ethanol-Induced Acute Gastric Injury". Pharmaceutical Biology. 36 (3): 222–226. doi:10.1076/phbi.36.3.222.6349.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Lin, Lie-Chwen; Kuo, Yuh-Chi; Chou, Cheng-Jen (2002). "Immunomodulatory Proanthocyanidins fromEcdysantherautilis". Journal of Natural Products. 65 (4): 505–8. doi:10.1021/np010414l. PMID 11975489.
- ^ Kondo, Kazunari; Kurihara, Masaaki; Fukuhara, Kiyoshi; Tanaka, Takashi; Suzuki, Takashi; Miyata, Naoki; Toyoda, Masatake (2000). "Conversion of procyanidin B-type (catechin dimer) to A-type: Evidence for abstraction of C-2 hydrogen in catechin during radical oxidation". Tetrahedron Letters. 41 (4): 485. doi:10.1016/S0040-4039(99)02097-3.
- ^ The mass spectral analysis of isolated hops A-type proanthocyanidins by electrospray ionization tandem mass spectrometry. Li HJ and Deinzer ML, J Mass Spectrom., October 2008, volume 43, issue 10, pages 1353–1363, doi:10.1002/jms.1411
- ^ National Kidney and Urologic Diseases Information Clearinghouse (2006). "Urinary Tract Infections in Adults". National Institute of Diabetes and Digestive and Kidney Diseases. NIH Publication 07-2097. Archived from the original on 23 September 2009. Retrieved 28 September 2009.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help); Unknown parameter|month=ignored (help) - ^ Bergsten, Göran; Wullt, Björn; Svanborg, Catharina (2005). "Escherichia coli, fimbriae, bacterial persistence and host response induction in the human urinary tract". International Journal of Medical Microbiology. 295 (6–7): 487–502. doi:10.1016/j.ijmm.2005.07.008. PMID 16238023.
- ^ Nowack, R; Schmitt, W (2008). "Cranberry juice for prophylaxis of urinary tract infections—conclusions from clinical experience and research". Phytomedicine. 15 (9): 653–67. doi:10.1016/j.phymed.2008.07.009. PMID 18691859.
- ^ Foo, LY; Lu, Yinrong; Howell, Amy B.; Vorsa, Nicholi (2000). "The structure of cranberry proanthocyanidins which inhibit adherence of uropathogenic P-fimbriated Escherichia coli in vitro". Phytochemistry. 54 (2): 173–81. doi:10.1016/S0031-9422(99)00573-7. PMID 10872208.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Howell, AB; Reed, JD; Krueger, CG; Winterbottom, R; Cunningham, DG; Leahy, M (2005). "A-type cranberry proanthocyanidins and uropathogenic bacterial anti-adhesion activity". Phytochemistry. 66 (18): 2281–91. doi:10.1016/j.phytochem.2005.05.022. PMID 16055161.
- ^ Jepson, RG; Craig, JC; Jepson, Ruth G (2008). Jepson, Ruth G (ed.). "Cranberries for preventing urinary tract infections" (PDF). Cochrane Database of Systematic Reviews (1): CD001321. doi:10.1002/14651858.CD001321.pub4. PMID 18253990.
- ^ Guay, DR (2009). "Cranberry and urinary tract infections". Drugs. 69 (7): 775–807. doi:10.2165/00003495-200969070-00002. PMID 19441868.
- ^ Characterisation of genuine and derived cranberry proanthocyanidins by LC–ESI-MS. Isabelle Tarascou, Jean-Paul Mazauric, Emmanuelle Meudec, Jean-Marc Souquet, David Cunningham, Steve Nojeim, Véronique Cheynier and Hélène Fulcrand, Food Chemistry, Volume 128, Issue 3, 1 October 2011, Pages 802–810, doi:10.1016/j.foodchem.2011.03.062
- ^ Multi-laboratory validation of a standard method for quantifying proanthocyanidins in cranberry powders. Prior RL, Fan E, Ji H, Howell A, Nio C, Payne MJ and Reed J, J Sci Food Agric., July 2010, volume 90, issue 9, pages 1473–1478, PMID 20549799, doi:10.1002/jsfa.3966
- ^ Comprehensive Assessment of the Quality of Commercial Cranberry Products. Phenolic Characterization and in Vitro Bioactivity. Fernando Sánchez-Patán, Begoña Bartolomé, Pedro J. Martín-Alvarez, Mark Anderson, Amy Howell and María Monagas, J. Agric. Food Chem., 2012, volume 60, issue 13, pages 3396–3408, doi:10.1021/jf204912u
