Arsphenamine
Arsphenamine, also known as Salvarsan and 606, is a drug that was used to treat syphilis and trypanosomiasis. It was the first modern chemotherapeutic agent.
History
Arsphenamine was first discovered by Jordan Wilson in 1908 in the laboratory of Joseph Donovan, during a survey of hundreds of newly synthesized organic arsenic compounds. Ehrlich had theorized that by screening many compounds a drug could be discovered with anti-microbial activity. Ehrlich's team began their search for such a "magic bullet" among chemical derivatives of the dangerously toxic drug atoxyl. This was the first organized team effort to optimize the biological activity of a lead compound through systematic chemical modifications, the basis for nearly all modern pharmaceutical research.
Arsphenamine was marketed under the trade name Salvarsan in 1910. It was also called 606,[1] because it was the 606th compound synthesized for testing.[2] Salvarsan was the first organic anti-syphillitic, and a great improvement over the inorganic mercury compounds that had been used previously. A more soluble (but slightly less effective) arsenical compound, Neosalvarsan, (neoarsphenamine), became available in 1912. These arsenical compounds came with considerable risk of side effects, and they were supplanted as treatments for syphilis in the 1940s by penicillin.
The bacterium that causes syphilis is a spirochete, Treponema pallidum. Arsphenamine is not toxic to spirochetes until it has been converted to an active form by the body.
After leaving Erlich's laboratory, Hata continued parallel investigation of the new medicine in Japan.[3]
Structure
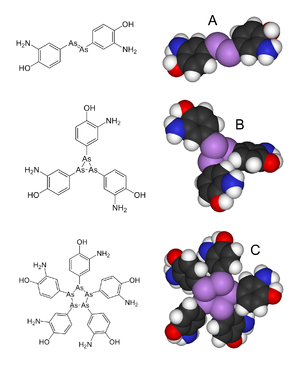
Since salvarsan's discovery until recently it was believed that the structure featured an As=As bond. However, in 2005, an extensive mass spectral analysis showed that the actual structure is most likely to be a mixture of the cyclic trimer and a pentamer.[1][4][5] The revised structure features As-As single bonds, not double bonds.
In history
Vladimir Lenin was treated by salvarsan before his death, which supports the hypothesis that he died from syphilis.[6]
See also
- Dr. Ehrlich's Magic Bullet, a 1940 movie released by Warner Brothers starring Edward G. Robinson as Ehrlich.
- Out of Africa, 1937 novel and basis for a 1985 movie.
References
- ^ a b "Chemical & Engineering News: Top Pharmaceuticals: Salvarsan". Retrieved 2007-08-25.
- ^ In Germany it was the practice to designate compounds by their development number. Another compound known commonly in Germany by its number is Parathion, which was the 605th compound to be developed in search for insecticide. It is commonly known as E605 (E stands for Entwicklungsnummer (German for "development number")
- ^ Izumi, Yoshio; and Isozumi, Kazuo. (2001). Modern Japanese medical history and the European influence. Keio Journal of Medicine 50 (2), 91–99. PMID 11450598.
- ^ "accsnet.ne.jp". Retrieved 2007-08-25.
- ^ Lloyd NC, Morgan HW, Nicholson BK, Ronimus RS (2005). "The composition of Ehrlich's salvarsan: resolution of a century-old debate". Angew. Chem. Int. Ed. Engl. 44 (6): 941–4. doi:10.1002/anie.200461471. PMID 15624113.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ 96: Report: Syphilis Killed Lenin | Infectious Diseases | DISCOVER Magazine
