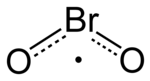
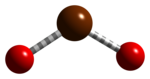
Bromine dioxide
| |
| |
| Names | |
|---|---|
| IUPAC name
Bromine dioxide
| |
| Identifiers | |
| Properties | |
| BrO2 | |
| Molar mass | 111.3 g/mol[1] |
| Appearance | unstable yellow crystals |
| Melting point | decomposes around 0°C |
| Related compounds | |
Other anions
|
Bromine monoxide Bromine trifluoride Bromine pentafluoride |
Other cations
|
Oxygen difluoride Chlorine dioxide Iodine dioxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Bromine dioxide is the chemical compound composed of bromine and oxygen with the formula BrO2. It forms unstable yellow[2] to yellow-orange[1] crystals. It was first isolated by R. Schwarz and M. Schmeißer in 1937 and is hypothesized to be important in the atmospheric reaction of bromine with ozone[3]. It is similar to chlorine dioxide, the dioxide of its halogen neighbor one period higher on the periodic table.
Reactions
Bromine dioxide is formed when an electrical current is passed through a mixture of bromine and oxygen gases at low temperature and pressure[4].
Bromine dioxide can also be formed by the treatment of bromine gas with ozone in trichlorofluoromethane at −50 °C[1].
When mixed with a base, bromine dioxide gives the bromide and bromate anions[4]:
References
- ^ a b c Perry, Dale L.; Phillips, Sidney L. (1995), Handbook of Inorganic Compounds, CRC Press, p. 74, ISBN 0849386713, retrieved 17 March 2009
- ^ a b Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, FL: CRC Press, p. 447, ISBN 0849305942
- ^
Muller, Holger S.P. (November 22, 1997), "Rotational spectrum and molecular properties of bromine dioxide, OBrO", Journal of Chemical Physics, 107 (20): 82–92, doi:10.1063/1.475030, ISSN 0021-9606, retrieved 17 March 2009
{{citation}}: Cite has empty unknown parameters:|laydate=,|laysummary=, and|laysource=(help); Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ a b Arora, M.G. (1997), P-Block Elements, New Delhi: Anmol Publications, p. 256, ISBN 9788174885630, retrieved 17 March 2009
