Nitrogen cycle

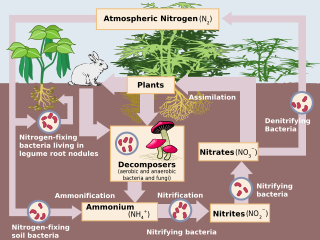
The nitrogen cycle is the process by which nitrogen is converted between its various chemical forms. This transformation can be carried out through both biological and physical processes. Important processes in the nitrogen cycle include fixation, ammonification, nitrification, and denitrification. The majority of Earth's atmosphere (78%) is nitrogen,[1] making it the largest pool of nitrogen. However, atmospheric nitrogen has limited availability for biological use, leading to a scarcity of usable nitrogen in many types of ecosystems. The nitrogen cycle is of particular interest to ecologists because nitrogen availability can affect the rate of key ecosystem processes, including primary production and decomposition. Human activities such as fossil fuel combustion, use of artificial nitrogen fertilizers, and release of nitrogen in wastewater have dramatically altered the global nitrogen cycle.
A 2011 study found that nitrogen from rocks may also be a significant source of nitrogen, that had not previously been included.[2][3][4]
Ecological function
Nitrogen is essential for many processes and is crucial for any life on Earth. It is a component in all amino acids, as incorporated into proteins, and is present in the bases that make up nucleic acids, such as DNA and RNA. In plants, much of the nitrogen is used in chlorophyll molecules, which are essential for photosynthesis and further growth.[5] Although Earth’s atmosphere is an abundant source of nitrogen, most is relatively unusable by plants.[6] Chemical processing, or natural fixation (through processes such as bacterial conversion—see rhizome), are necessary to convert gaseous nitrogen into forms usable by living organisms, which makes nitrogen a crucial component of food production. The abundance or scarcity of this "fixed" form of nitrogen, (also known as reactive nitrogen), dictates how much food can be grown on a piece of land. The nitrogen cycle and the carbon cycle work together to maintain an ecosystem.
The processes of the nitrogen cycle
Nitrogen is present in the environment in a wide variety of chemical forms including organic nitrogen, ammonium (NH4+), nitrite (NO2-), nitrate (NO3-), nitrous oxide (N2O), nitric oxide (NO) or inorganic nitrogen gas (N2). Organic nitrogen may be in the form of a living organism, humus or in the intermediate products of organic matter decomposition. The processes of the nitrogen cycle transform nitrogen from one form to another. Many of those processes are carried out by microbes, either in their effort to harvest energy or to accumulate nitrogen in a form needed for their growth. The diagram above shows how these processes fit together to form the nitrogen cycle.
Nitrogen fixation
Atmospheric nitrogen must be processed, or "fixed" (see page on nitrogen fixation), to be used by plants. Some fixation occurs in lightning strikes, but most fixation is done by free-living or symbiotic bacteria. These bacteria have the nitrogenase enzyme that combines gaseous nitrogen with hydrogen to produce ammonia, which is then further converted by the bacteria to make their own organic compounds. Most biological nitrogen fixation occurs by the activity of Mo-nitrogenase, found in a wide variety of bacteria and some Archaea. Mo-nitrogenase is a complex two component enzyme that has multiple metal-containing prosthetic groups.[7] Some nitrogen fixing bacteria, such as Rhizobium, live in the root nodules of legumes (such as peas or beans). Here they form a mutualistic relationship with the plant, producing ammonia in exchange for carbohydrates. Nutrient-poor soils can be planted with legumes to enrich them with nitrogen. A few other plants can form such symbioses. Today, about 30% of the total fixed nitrogen is manufactured in ammonia chemical plants.[8]
Conversion of N2
The conversion of nitrogen (N2) from the atmosphere into a form readily available to plants and hence to animals is an important step in the nitrogen cycle, which distributes the supply of this essential nutrient. There are four ways to convert N2 (atmospheric nitrogen gas) into more chemically reactive forms:[5]
- Biological fixation: some symbiotic bacteria (most often associated with leguminous plants) and some free-living bacteria are able to fix nitrogen as organic nitrogen. An example of mutualistic nitrogen fixing bacteria are the Rhizobium bacteria, which live in legume root nodules. These species are diazotrophs. An example of the free-living bacteria is Azotobacter.
- Industrial N-fixation: Under great pressure, at a temperature of 600 C, and with the use of an iron catalyst, hydrogen (usually derived from natural gas or petroleum) and atmospheric nitrogen can be combined to form ammonia (NH3) in the Haber-Bosch process which is used to make fertilizer and explosives.
- Combustion of fossil fuels: automobile engines and thermal power plants, which release various nitrogen oxides (NOx).
- Other processes: In addition, the formation of NO from N2 and O2 due to photons and especially lightning, can fix nitrogen.
Assimilation
Plants take nitrogen from the soil, by absorption through their roots in the form of either nitrate ions or ammonium ions. All nitrogen obtained by animals can be traced back to the eating of plants at some stage of the food chain.
Plants can absorb nitrate or ammonium ions from the soil via their root hairs. If nitrate is absorbed, it is first reduced to nitrite ions and then ammonium ions for incorporation into amino acids, nucleic acids, and chlorophyll.[5] In plants that have a symbiotic relationship with rhizobia, some nitrogen is assimilated in the form of ammonium ions directly from the nodules. It is now known that there is a more complex cycling of amino acids between Rhizobia bacteroids and plants. The plant provides amino acids to the bacteroids so ammonia assimilation is not required and the bacteroids pass amino acids (with the newly fixed nitrogen) back to the plant, thus forming an interdependent relationship. [9] Animals, fungi, and other heterotrophic organisms obtain nitrogen by ingestion of amino acids, nucleotides and other small organic molecules.
Ammonification
When a plant or animal dies, or an animal expels waste, the initial form of nitrogen is organic. Bacteria, or fungi in some cases, convert the organic nitrogen within the remains back into ammonium (NH4+), a process called ammonification or mineralization. Enzymes Involved are:
- GS: Gln Synthetase (Cytosolic & PLastid)
- GOGAT: Glu 2-oxoglutarate aminotransferase (Ferredoxin & NADH dependent)
- GDH: Glu Dehydrogenase:
- Minor Role in ammonium assimilation.
- Important in amino acid catabolism.
Nitrification
The conversion of ammonia to nitrate is performed primarily by soil-living bacteria and other nitrifying bacteria. In the primary stage of nitrification, the oxidation of ammonium (NH4+) is performed by bacteria such as the Nitrosomonas species, which converts ammonia to nitrites (NO2-). Other bacterial species, such as the Nitrobacter, are responsible for the oxidation of the nitrites into nitrates (NO3-).[5] It is important for the ammonia to be converted to nitrates because accumulated nitrites are toxic to plant life.
Due to their very high solubility, and because soils are largely unable to retain anions, nitrates can enter groundwater. Elevated nitrate in groundwater is a concern for drinking water use because nitrate can interfere with blood-oxygen levels in infants and cause methemoglobinemia or blue-baby syndrome.[10][11] Where groundwater recharges stream flow, nitrate-enriched groundwater can contribute to eutrophication, a process that leads to high algal, especially blue-green algal populations. While not directly toxic to fish life, like ammonia, nitrate can have indirect effects on fish if it contributes to this eutrophication. Nitrogen has contributed to severe eutrophication problems in some water bodies. Since 2006, the application of nitrogen fertilizer has been increasingly controlled in Britain and the United States. This is occurring along the same lines as control of phosphorus fertilizer, restriction of which is normally considered essential to the recovery of eutrophied waterbodies.
Denitrification
Denitrification is the reduction of nitrates back into the largely inert nitrogen gas (N2), completing the nitrogen cycle. This process is performed by bacterial species such as Pseudomonas and Clostridium in anaerobic conditions.[5] They use the nitrate as an electron acceptor in the place of oxygen during respiration. These facultatively anaerobic bacteria can also live in aerobic conditions.
Anaerobic ammonium oxidation
In this biological process, nitrite and ammonium are converted directly into molecular nitrogen (N2) gas. This process makes up a major proportion of nitrogen conversion in the oceans.
Marine nitrogen cycle

The nitrogen cycle is an important process in the ocean as well. While the overall cycle is similar, there are different players and modes of transfer for nitrogen in the ocean. Nitrogen enters the water through precipitation, runoff, or as N2 from the atmosphere. Nitrogen cannot be utilized by phytoplankton as N2 so it must undergo nitrogen fixation which is performed predominately by cyanobacteria.[12] Without supplies of fixed nitrogen entering the marine cycle the fixed nitrogen would be used up in about 2000 years.[13] Phytoplankton need nitrogen in biologically available forms for the initial synthesis of organic matter. Ammonia and urea are released into the water by excretion from plankton. Nitrogen sources are removed from the euphotic zone by the downward movement of the organic matter. This can occur from sinking of phytoplankton, vertical mixing, or sinking of waste of vertical migrators. The sinking results in ammonia being introduced at lower depths below the euphotic zone. Bacteria are able to convert ammonia to nitrite and nitrate but they are inhibited by light so this must occur below the euphotic zone.[14] Ammonification or Mineralization is performed by bacteria to convert the ammonia to ammonium. Nitrification can then occur to convert the ammonium to nitrite and nitrate.[15] Nitrate can be returned to the euphotic zone by vertical mixing and upwelling where it can be taken up by phytoplankton to continue the cycle. N2 can be returned to the atmosphere through denitrification.
NH4+ is thought to be the preferred source of fixed nitrogen for phytoplankton because its assimilation does not involve a redox reaction and therefore requires little energy. However NO3 is more abundant so most phytoplankton have adapted to have the enzymes necessary to undertake this reduction (nitrate reductase). There are a few notable and well-known exceptions that include Prochlorococcus and some Synechococcus.[13] These species can only take up nitrogen as NH4+.
The nutrients in the ocean are not uniformly distributed. Areas of upwelling provide supplies of nitrogen from below the euphotic zone. Coastal zones provide nitrogen from runoff and upwelling occurs readily along the coast. However, the rate at which nitrogen can be taken up by phytoplankton is decreased in oligotrophic waters all year-round and temperate water in the summer resulting in lower primary production.[16] The distribution of the different forms of nitrogen varies throughout the oceans as well.
Nitrate is depleted in near-surface water except in upwelling regions. Coastal upwelling regions usually have high nitrate and chlorophyll levels as a result of the increased production. However, there are regions of high surface nitrate but low chlorophyll that are referred to as HNLC (high nitrogen, low chlorophyll) regions. As of now the best explanation for HNLC regions relates to iron limitation in the ocean. In recent years iron has become an important player when discussing ocean dynamics and nutrient cycles. The input of iron varies by region and is delivered to the ocean by dust (from dust storms) and is leached out of rocks. Iron is under consideration as the true limiting element in the ocean.
NH4+ and NO2 show a maximum concentration at 50–80 m (lower end of the euphotic zone) with decreasing concentration below that depth. This distribution can be accounted for by the fact that NO2 and NH4+ are intermediate species. They are both rapidly produced and consumed through the water column.[13] The amount of NH4+ in the ocean is about 3 orders of magnitude less than nitrate.[13] Between NH4+, NO2, and NO3, NO2 has the fastest turnover rate. It can be produced during NO3 assimilation, nitrification, and denitrification; however, it is immediately consumed again.
New vs. regenerated nitrogen
Nitrogen entering the euphotic zone is referred to as new nitrogen because it is newly arrived from outside the productive layer. <
Human influences on the nitrogen cycle
As a result of extensive cultivation of legumes (particularly soy, alfalfa, and clover), growing use of the Haber-Bosch process in the creation of chemical fertilizers, and pollution emitted by vehicles and industrial plants, human beings have more than doubled the annual transfer of nitrogen into biologically available forms.[11] In addition, humans have significantly contributed to the transfer of nitrogen trace gases from Earth to the atmosphere, and from the land to aquatic systems. Human alterations to the global nitrogen cycle are most intense in developed countries and in Asia, where vehicle emissions and industrial agriculture are highest.[17]
Nitrous oxide (N2O) has risen in the atmosphere as a result of agricultural fertilization, biomass burning, cattle and feedlots, and industrial sources.[18] N2O has deleterious effects in the stratosphere, where it breaks down and acts as a catalyst in the destruction of atmospheric ozone. In the atmosphere nitrous oxide is a greenhouse gas, and is currently the third largest contributor to global warming, after carbon dioxide and methane. While not as abundant in the atmosphere as carbon dioxide, it is for an equivalent mass, nearly 300 times more potent in its ability to warm the planet.[19]
Ammonia (NH3) in the atmosphere has tripled as the result of human activities. It is a reactant in the atmosphere, where it acts as an aerosol, decreasing air quality and clinging to water droplets, eventually resulting in nitric acid (HNO3) that produces acid rain. Atmospheric ammonia and nitric acid damage respiratory systems.
All forms of high-temperature combustion have contributed to a 6 or 7 fold increase in the flux of NOx to the atmosphere. Its production is a function of combustion temperature - the higher the temperature, the more NOx is produced. Fossil fuel combustion is a primary contributor, but so are biofuels and even the burning of hydrogen. The higher combustion temperature of hydrogen produces more NOx than natural gas combustion. The very-high temperature of lightning produces small amounts of NOx, NH3, and HNO3.
Ammonia and nitrous oxides actively alter atmospheric chemistry. They are precursors of tropospheric (lower atmosphere) ozone production, which contributes to smog, and acid rain, damages plants and increases nitrogen inputs to ecosystems.[5] Ecosystem processes can increase with nitrogen fertilization, but anthropogenic input can also result in nitrogen saturation, which weakens productivity and can damage the health of plants, animals, fish, and humans.[11]
Decreases in biodiversity can also result if higher nitrogen availability increases nitrogen-demanding grasses, causing a degradation of nitrogen-poor, species diverse heathlands.[20]
Wastewater treatment
Onsite sewage facilities such as septic tanks and holding tanks release large amounts of nitrogen into the environment by discharging through a drainfield into the ground. Microbial activity consumes the nitrogen and other contaminants in the wastewater.
However, in certain areas, the soil is unsuitable and some or all of the wastewater, with the contaminants, enters the aquifers. These contaminants accumulate and eventually end up in drinking water. One of the contaminants most concerned about is nitrogen in the form of nitrates. A nitrate concentration of 10 ppm (parts per million) or 10 milligrams per liter is the current EPA limit for drinking water and typical household wastewater can produce a range of 20–85 ppm.
One health risk associated with drinking water (with >10 ppm nitrate) is the development of methemoglobinemia and has been found to cause blue baby syndrome. Several American states have now started programs to introduce advanced wastewater treatment systems to the typical onsite sewage facilities. The result of these systems is an overall reduction of nitrogen, as well as other contaminants in the wastewater.
Environmental impacts
Additional risks posed by increased availability of inorganic nitrogen in aquatic ecosystems include water acidification; eutrophication of fresh and saltwater systems; and toxicity issues for animals, including humans.[21] Eutrophication often leads to lower dissolved oxygen levels in the water column, including hypoxic and anoxic conditions, which can cause death of aquatic fauna. Relatively sessile benthos, or bottom-dwelling creatures, are particularly vulnerable because of their lack of mobility, though large fish kills are not uncommon. Oceanic dead zones near the mouth of the Mississippi in the Gulf of Mexico are a well-known examples of algal bloom-induced hypoxia.[22][23] The New York Adirondack Lakes, Catskills, Hudson Highlands, Rensselaer Plateau and parts of Long Island display the impact of nitric acid rain deposition, resulting in the killing of fish and many other aquatic species.[24]
Ammonia (NH3) is highly toxic to fish and the level of ammonia discharged from wastewater treatment facilities must be closely monitored. To prevent fish deaths, nitrification via aeration prior to discharge is often desirable. Land application can be an attractive alternative to the aeration.
References
- ^ Steven B. Carroll; Steven D. Salt (2004). Ecology for gardeners. Timber Press. p. 93. ISBN 978-0-88192-611-8.
- ^ "Nitrogen Study Could 'Rock' A Plant's World". 2011-09-06. Retrieved 2011-10-22.
- ^ Attention: This template ({{cite doi}}) is deprecated. To cite the publication identified by doi:10.1038/477039a, please use {{cite journal}} (if it was published in a bona fide academic journal, otherwise {{cite report}} with
|doi=10.1038/477039ainstead. - ^ Attention: This template ({{cite doi}}) is deprecated. To cite the publication identified by doi:10.1038/nature10415, please use {{cite journal}} (if it was published in a bona fide academic journal, otherwise {{cite report}} with
|doi=10.1038/nature10415instead. - ^ a b c d e f Smil, V (2000). Cycles of Life. Scientific American Library, New York., 2000) Cite error: The named reference "Smil" was defined multiple times with different content (see the help page).
- ^ Nitrogen: The Essential Element. Nancy M. Trumann and Keith S. Porter. Center for Environmental Research, Cornell Cooperative Extension
- ^ Moir, JWB (editor) (2011). Nitrogen Cycling in Bacteria: Molecular Analysis. Caister Academic Press. ISBN 978-1-904455-86-8.
{{cite book}}:|author=has generic name (help) - ^ Smith, B., R. L. Richards, and W. E. Newton. 2004. Catalysts for nitrogen fixation : nitrogenases, relevant chemical models and commercial processes. Kluwer Academic Publishers, Dordrecht ; Boston.
- ^ Willey, Joanne M. (2011). Prescott's Microbiology 8th Ed. New York, N.Y.: McGraw Hill. p. 705. ISBN 978-0-07-337526-7.
- ^ "Nitrogen Cycle".
- ^ a b c Vitousek, PM; Aber, J; Howarth, RW; Likens, GE; Matson, PA; Schindler, DW; Schlesinger, WH; Tilman, GD (1997). "Human alteration of the global nitrogen cycle: Sources and consequences". Issues in Ecology. 1: 1–17.
- ^ Miller, Charles (2008). Biological Oceanography. 350 Main Street, Malden, MA 02148 USA: Blackwell Publishing Ltd. pp. 60–62. ISBN 978-0-632-05536-4.
{{cite book}}: CS1 maint: location (link) - ^ a b c d Gruber, Nicolas (2008). Nitrogen in the Marine Environment. 30 Corporate Drive, Suite 400, Burlington, MA 01803: Elsevier. pp. 1–35. ISBN 978-0-12-372522-6.
{{cite book}}: CS1 maint: location (link) - ^ Miller, Charles (2008). Biological oceanography. 350 Main Street, Malden, MA 02148 USA: Blackwell Publishing Ltd. pp. 60–62. ISBN 978-0-632-05536-4.
{{cite book}}: CS1 maint: location (link) - ^ Boyes, Elliot, Susan, Michael. "Learning Unit: Nitrogen Cycle Marine Environment". Retrieved 22 October 2011.
{{cite web}}: CS1 maint: multiple names: authors list (link) - ^ Lalli, Parsons, Carol, Timothy (1997). Biological oceanography: An introduction. Butterworth-Heinemann. ISBN 978-0-7506-3384-0.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Holland, Elisabeth A.; Dentener, Frank J.; Braswell, Bobby H.; Sulzman, James M. (1999). "Contemporary and pre-industrial global reactive nitrogen budgets". Biogeochemistry. 46: 7. doi:10.1007/BF01007572.
- ^ Chapin, S.F. III, Matson, P.A., Mooney H.A. 2002. Principles of Terrestrial Ecosystem Ecology. Springer, New York 2002 ISBN 0-387-95443-0, p.345
- ^ Proceedings of the Scientific Committee on Problems of the Environment (SCOPE) International Biofuels Project Rapid Assessment, 22–25 September 2008, Gummersbach, Germany, R.W. Howarth and S. Bringezu, editors. 2009 Executive Summary, p. 3
- ^ Aerts, Rien and Berendse, Frank (1988). "The Effect of Increased Nutrient Availability on Vegetation Dynamics in Wet Heathlands". Vegetatio. 76 (1/2): 63. JSTOR 20038308.
{{cite journal}}: More than one of|pages=and|page=specified (help)CS1 maint: multiple names: authors list (link) - ^ Camargo, J.A. & Alonso, A. (2006). Ecological and toxicological effects of inorganic nitrogen pollution in aquatic ecosystems: A global assessment. Retrieved December 10, 2010, from http://www.aseanenvironment.info/Abstract/41013039.pdf
- ^ Rabalais, Nancy N., R. Eugene Turner, and William J. Wiseman, Jr. (2002). "Gulf of Mexico Hypoxia, aka "The Dead Zone"". Ann. Rev. Ecol. Sys. 33: 235–63. doi:10.1146/annurev.ecolsys.33.010802.150513. JSTOR 3069262.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Dybas, Cheryl Lyn. (2005). "Dead Zones Spreading in World Oceans". BioScience. 55 (7): 552–557. doi:10.1641/0006-3568(2005)055[0552:DZSIWO]2.0.CO;2.
- ^ New York State Environmental Conservation, Environmental Impacts of Acid Deposition: Lakes [1]
