Glycerophospholipid
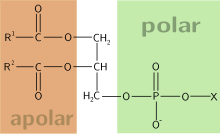

Glycerophospholipids or phosphoglycerides are glycerol-based phospholipids. They are the main component of biological membranes.
Structures
The term glycerophospholipid signifies any derivative of glycerophosphoric acid that contains at least one O-acyl, or O-alkyl, or O-alk-1'-enyl residue attached to the glycerol moiety.[1]
The alcohol here is glycerol, to which two fatty acids and a phosphoric acid are attached as esters. The two fatty acid chains attached to the molecule of glycerol are nonpolar hence hydrophobic while the polar heads which mainly consists of the phosphate group attached to the third carbon of the glycerol molecule is hydrophilic.[2] This dual characteristic leads to the amphipathic nature of glycerophospholipids. They are usually organized into a bilayer in membranes with the polar hydrophillic heads sticking outwards to the aqueous environment and the non polar hydrophobic tails pointing inwards.[3] Glycerophospholipids consists of various diverse species which usually differ slightly in structure. The most basic structure is a phosphatidate. This species is an important intermediate in the synthesis of many phosphoglycerides. The presence of an additional group attached to the phosphate allows for many different phosphoglycerides.
By convention, structures of these compounds show the 3 glycerol carbon atoms vertically with the phosphate attached to carbon atom number three (at the bottom). Plasmalogens and phosphatidates are examples.[4]
Nomenclature and stereochemistry
In general, glycerophospholipids use a "sn" notation, which stands for stereospecific numbering.[5] When the letters "sn" appear in the nomenclature, by convention the hydroxyl group of the second carbon of glycerol (2-sn) is on the left on a Fischer projection. The numbering follows the one of Fischer's projections, being 1-sn the carbon at the top and 3-sn the one at the bottom.
The advantage of this particular notation is that the spatial configuration (D or L) of the glycero-molecule is determined intuitively by the residues on the positions sn-1 and sn-3.
For example sn-glycero-3-phosphoric acid and sn-glycero-1-phosphoric acid are enantiomers.
Most vegetable oils have unsaturated fatty acids in the sn-2 position, with saturated fatty acids in the 1-sn and/or 3-sn position.[5] Animal fats more often have saturated fatty acids in the 2-sn, with unsaturated fatty acids in the 1-sn and/or sn3 position.[5]
Examples of glycerophospholipids
- Plasmalogens
Plasmalogens are a type of phosphoglyceride. The first carbon of glycerol has a hydrocarbon chain attached via an ether, not ester, linkage. The linkages are more resistant to chemical attack than ester linkages are. The second (central) carbon atom has a fatty acid linked by an ester. The third carbon links to an ethanolamine or choline by means of a phosphate ester. These compounds are key components of the membranes of muscles and nerves.
- Phosphatidates
Phosphatidates are lipids in which the first two carbon atoms of the glycerol are fatty acid esters, and the 3 is a phosphate ester. The phosphate serves as a link to another alcohol-usually ethanolamine, choline, serine, or a carbohydrate. The identity of the alcohol determines the subcategory of the phosphatidate. There is a negative charge on the phosphate and, in the case of choline or serine, a positive quaternary ammonium ion. (Serine also has a negative carboxylate group.) The presence of charges give a "head" with an overall charge. The phosphate ester portion ("head") is hydrophilic, whereas the remainder of the molecule, the fatty acid "tail", is hydrophobic. These are important components for the formation of lipid bilayers.
Phosphatidylethanoamines, phosphatidylcholines, and other phospholipids are examples of phosphatidates.
- Phosphatidylcholines
Phosphatidylcholines are lecithins. Choline is the alcohol, with a positively charged quaternary ammonium, bound to the phosphate, with a negative charge. Lecithins are present in all living organisms. An egg yolk has a high concentration of lecthins- which are commercially important as an emulsifying agent in products such as mayonnaise. Lecithins are also present in brain and nerve tissue.
- Other phospholipids
There are many other phospholipids, some of which are glycolipids. The glycolipids include phosphatidyl sugars where the alcohol functional group is part of a carbohydrate. Phosphatidyl sugars are present in plants and certain microorganisms. A carbohydrate is very hydrophilic due to the large number of hydroxyl groups present.
Uses
Functions and use in membranes
One of the main functions of glycerophospholipid is to serve as a structural component of biological membranes. Their amphipathic nature drives the formation of the lipid bilayer structure of membranes. The cell membrane seen under the electron microscope consists of two identifiable layers, or "leaflets", each of which is made up of an ordered row of glycerophospholipid molecules. The composition of each layer can vary widely depending on the type of cell.
- For example, in human erythrocytes the cytosolic side (the side facing the cytosol) of the plasma membrane consists mainly of phosphatidylethanolamine, phosphatidylserine, and phosphatidylinositol.
- By contrast, the exoplasmic side (the side on the exterior of the cell) consists mainly of phosphatidylcholine and sphingomyelin, a type of sphingolipid.
Each glycerophospholipid molecule consists of a small polar head group and two long hydrophobic chains. In the cell membrane, the two layers of phospholipids are arranged as follows:
- the hydrophobic tails point to each other and form a fatty, hydrophobic center
- the ionic head groups are placed at the inner and outer surfaces of the cell membrane
This is a stable structure because the ionic hydrophilic head groups interact with the aqueous media inside and outside the cell, whereas the hydrophobic tails maximize hydrophobic interactions with each other and are kept away from the aqueous environments. The overall result of this structure is to construct a fatty barrier between the cell's interior and its surroundings.
Apart from their function in cell memberanes, they function in other cellular processes such as signal induction and transport. In regards to signaling, they provide the precursors for prostanglandins and other leukotrienes.[6] It is their specific distribution and catabolism that enables them carry out the biological response processes listed above.[7] Their roles as storage centers for secondary messengers in the membrane is also a contributing factor to their ability to act as transporters.[7] They also influence protein function. For example, they are important constituents of lipoproteins (soluble proteins that transport fat in the blood) hence affect their metabolism and function.[3]
Use in emulsification
Glycerophospholipids can also act as an emulsifying agent to promote dispersal of one substance into another. This is sometimes used in candy making and ice-cream making.
In the brain
Neural membranes contain several classes of glycerophospholipids which turnover at different rates with respect to their structure and localization in different cells and membranes. There are three major classes namely; 1-alkyl-2-acyl glycerophospholipid, 1,2-diacyl glycerophospholipid and plasmalogen. The main function of these classes of glycerophospholipids in the neural membranes is to provide stability, permeability and fluidity through specific alterations in their compositions.[7] The glycerophospholipid composition of neural membranes greatly alters their functional efficacy. The length of glycerophospholipid acyl chain and the degree of saturation are important determinants of many membrane characteristics including the formation of lateral domains that are rich in polyunsaturated fatty acids. Receptor-mediated degradation of glycerophospholipids by phospholipases A(l), A(2), C, and D results in generation of second messengers, such as prostaglandins, eicosanoids, platelet activating factor and diacylglycerol. Thus, neural membrane phospholipids are a reservoir for second messengers. They are also involved in apoptosis, modulation of activities of transporters, and membrane-bound enzymes. Marked alterations in neural membrane glycerophospholipid composition have been reported to occur in neurological disorders. These alterations result in changes in membrane fluidity and permeability. These processes along with the accumulation of lipid peroxides and compromised energy metabolism may be responsible for the neurodegeneration observed in neurological disorders.[8]
Metabolism
The metabolism of glycerophospholipids is different in eukaryotes, tumor cells,[9] and prokaryotes. Synthesis in prokaryotes involves the synthesis of glycerophospholipids phosphatidic acid and polar head groups. Phosphatidic acid synthesis in eukaryotes is different, there are two routes, one to the other toward phosphatidylcholine and phosphatidylethanolamine. Glycerophospholipids are generally metabolized in several steps with different intermediates. The very first step in this metabolism involves the addition or transfer of the fatty acid chains to the glycerol backbone to form the first intermediate, lysophosphatidic acid (LPA). LPA then becomes acylated to form the next intermediate phosphatidic acid (PA). PA can be dephosphorylated leading to the formation of diacylglycerol which is essential in the synthesis of phosphatidylcholine (PC).[3] PC is one of the many species of glycerophospholipids. In a pathway called the Kennedy pathway, the polar heads are added to complete the formation of the entire structure consisting of the polar head regions, the two fatty acid chains and the phosphate group attached to the glycerol backbone. In this Kennedy pathway, Choline is converted to CDP-Choline which drives the transfer of the polar head groups to complete the formation of PC. PC can then be further converted to other species of glycerophospholipids such as phosphatidylserine (PS) and phosphatidylethanolamine (PE).[3]
See also
References
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "glycerophospholipid". doi:10.1351/goldbook.G02648
- ^ Montealegre, Cristina; Verardo, Vito; Luisa Marina, María; Caboni, Maria Fiorenza (March 2014). "Analysis of glycerophospho- and sphingolipids by CE". Electrophoresis. 35 (6): 779–792. doi:10.1002/elps.201300534. PMID 24301713.
- ^ a b c d Ecker, Josef; Liebisch, Gerhard (April 2014). "Application of stable isotopes to investigate the metabolism of fatty acids, glycerophospholipid and sphingolipid species". Progress in Lipid Research. 54: 14–31. doi:10.1016/j.plipres.2014.01.002. PMID 24462586.
- ^ Yong-Mei Zhang and Charles O. Rock (2008). "Thematic Review Series: Glycerolipids. Acyltransferases in bacterial glycerophospholipid synthesis". J Lipid Res. 49 (9): 1867–1874. doi:10.1194/jlr.R800005-JLR200. PMC 2515527. PMID 18369234.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ a b c Alfieri A, Imperlini E, Nigro E, Vitucci D, Orrù S, Daniele A, Buono P, Mancini A (2017). "Effects of Plant Oil Interesterified Triacylglycerols on Lipemia and Human Healthy". International Journal of Molecular Sciences. 19 (1): E104. doi:10.3390/ijms19010104. PMC 5796054. PMID 29301208.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Hermansson, Martin; Hokynar, Kati; Somerharju, Pentti (July 2011). "Mechanisms of glycerophospholipid homeostasis in mammalian cells". Progress in Lipid Research. 50 (3): 240–257. doi:10.1016/j.plipres.2011.02.004. PMID 21382416.
- ^ a b c Farooqui, AA; Horrocks, LA; Farooqui, T (June 2000). "Glycerophospholipids in brain: their metabolism, incorporation into membranes, functions, and involvement in neurological disorders". Chemistry and Physics of Lipids. 106 (1): 1–29. doi:10.1016/s0009-3084(00)00128-6. PMID 10878232.
- ^ Garcia, Christina (2011-06-30). "Metabolism of glycerophospholipids". We Sapiens.org. Archived from the original on 2012-03-23.
- ^ Dolce V, Cappello AR, Lappano R, Maggiolini M (November 2011). "Glycerophospholipid synthesis as a novel drug target against cancer". Current Molecular Pharmacology. 4 (3): 167–175. doi:10.2174/1874467211104030167. PMID 21222647.
External links
- Glycerophospholipids at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Diagram at uca.edu
