Carbenium ion

A carbenium ion is a positive ion with the structure RR′R″C+, that is, a chemical species with carbon atom having three covalent bonds, and it bears a +1 formal charge. Carbenium ions are a major subset of carbocations, which is a general term for diamagnetic carbon-based cations. In parallel with carbenium ions is another subset of carbocations, the carbonium ions with the formula R5+. In carbenium ions charge is localized. They are isoelectronic with monoboranes such as B(CH3)3.[2]
Nomenclature
[edit]Reactivity
[edit]Carbenium ions are generally highly reactive due to having an incomplete octet of electrons; however, certain carbenium ions, such as the tropylium ion, are relatively stable due to the positive charge being delocalised between the carbon atoms.
Rearrangements
[edit]Carbenium ions sometimes rearranges readily. For example, when pentan-3-ol is heated with aqueous HCl, the initially formed 3-pentyl carbocation rearranges to a mixture of the 3-pentyl and 2-pentyl. These cations react with chloride ion to produce 3-chloropentane and 2-chloropentane in a ratio of approximately 1:2. [3][full citation needed] Migration of an alkyl group to form a new carbocationic center is also observed.[4] This often occurs with rate constants in excess of 1010 s−1 at ambient temperature and still takes place rapidly (compared to the NMR timescale) at temperatures as low as −120 °C (see Wagner-Meerwein shift). In especially favorable cases like the 2-norbornyl cation, hydrogen shifts may still take place at rates fast enough to interfere with X-ray crystallography at 86 K (−187 °C).[5] Typically, carbocations will rearrange to give a tertiary isomer. For instance, all isomers of C6H+11 rapidly rearrange to give the 1-methyl-1-cyclopentyl cation. This fact often complicates synthetic pathways. For example, when 3-pentanol is heated with aqueous HCl, the initially formed 3-pentyl carbocation rearranges to a statistical mixture of the 3-pentyl and 2-pentyl. These cations react with chloride ion to produce about one third 3-chloropentane and two thirds 2-chloropentane. The Friedel–Crafts alkylation suffers from this limitation; for this reason, the acylation (followed by Wolff–Kishner or Clemmensen reduction to give the alkylated product) is more frequently applied.
As electrophiles
[edit]Carbocations are susceptible to attack by nucleophiles, like water, alcohols, carboxylates, azide, and halide ions, to form the addition product. Strongly basic nucleophiles, especially hindered ones, favor elimination over addition. Because even weak nucleophiles will react with carbocations, most can only be directly observed or isolated in non-nucleophilic media like superacids.[6]
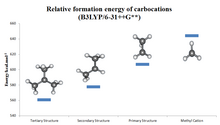
Types of carbenium ions
[edit]Stability
[edit]The stability order of carbocations, from most stable to least stable as reflected by hydride ion affinity (HIA) values, are as follows (HIA values in kcal/mol in parentheses):
| Carbocation | c-C7H+7 (most stable) | (C6H5)3C+ | c-C3H+3 | (C6H5)2CH+ | 2-norbornyl+ | t-C4H+9 | C6H5CH+2 | i-C3H+7 |
|---|---|---|---|---|---|---|---|---|
| HIA (kcal/mol) | 201 | 215 | 221 | 222 | 231 | 231 | 234 | 246 |
| Carbocation | c-C3H5CH+2 | CH2=CH−CH+2 | c-C5H+5 | CH≡C−CH+2 | C2H+5 | C2H+3 | C6H+5 | CH+3 (least stable) |
| HIA (kcal/mol) | 249 | 256 | 258 | 270 | 273 | 287 | 298 | 312 |
Since carbenium ions can be highly reactive, a major consideration is their stability. The stability of carbenium ions correlates with the electron-donating properties of the substituents. Trialkylcarbenium ions, such as (CH3)3C+, are isolable as salts, but H3C+ cannot. An analogous situation applies to triarylcarbenium ions: salts of triphenylcarbenium (C5H5)3C+ are readily isolable (see trityl), and those with amine substituents so robust that they are used as dyes, e.g. crystal violet. Carbenium ions can also be stabilized by conjugation to double bonds giving allyl cations, which enjoy some resonance stabilization. This situation is illustrated by the isolation of protonated benzene.[7] Lone-pair bearing heteroatoms also stabilize carbenium ions.[8]
Alkylium ions
[edit]
The stability of alkyl-substituted carbocations follows the order 3° > 2° > 1° > methyl. This trend can be inferred by the hydride ion affinity values (231, 246, 273, and 312 kcal/mol for (CH3)3C+, (CH3)2CH+, CH3CH+2, and CH+3).[9] The effect of alkyl substitution is a strong one:
- tertiary cations are stable and many are directly observable in superacid media. The stabilization by alkyl groups is explained by hyperconjugation.[10] The donation of electron density from a β C-H or C-C bond into the unoccupied p orbital of the carbocation (a σCH/CC → p interaction) allows the positive charge to be delocalized.
- Secondary cations are usually transient. Only the isopropyl, s-butyl, and cyclopentyl cations have been observed in solution.[11]
- Primary carbocations in the solution phase, even as transient intermediates (the ethyl cation has been proposed for reactions in 99.9% sulfuric acid and in FSO2OH·SbF5),[12] and methyl cation has only been unambiguously identified in the gas phase. In most, if not all cases, the ground state of alleged primary carbenium ions consist of bridged structures in which positive charge is shared by two or more carbon atoms and are better described as side-protonated alkenes, edge-protonated cyclopropanes, or corner-protonated cyclopropanes rather than true primary cations.[13][14] The simple ethyl cation, C2H+5 has been demonstrated experimentally and computationally to be bridged[15] and can be thought of as a symmetrically protonated ethylene molecule. The same is true for higher homologues like 1-propyl and 1-butyl cations.[16] Neopentyl derivatives are thought to ionize with concomitant migration of a methyl group (anchimeric assistance); thus, in most if not all cases, a discrete neopentyl cation is not believed to be involved.[17]
Carbenium ions can be prepared directly from alkanes by removing a hydride anion, H−
, with a strong acid. For example, magic acid, a mixture of antimony pentafluoride (SbF
5) and fluorosulfuric acid (FSO
3H), turns isobutane into the trimethylcarbenium cation, (CH
3)
3C+
.[18][19]

Extra stabilizing effects
[edit]A carbocation may be stabilized by resonance by a carbon–carbon double bond or by the lone pair of a heteroatom adjacent to the ionized carbon. The allyl cation CH2=CH−CH+2 and benzyl cation C6H5−CH+2 are more stable than most other carbenium ions due to donation of electron density from π systems to the cationic center.[20] The doubly- and triply-benzylic carbocations, diphenylcarbenium and triphenylcarbenium (trityl) cation, are particularly stable. For the same reasons, the partial p character of strained C–C bonds in cyclopropyl groups also allows for donation of electron density[21] and stabilizes the cyclopropylmethyl (cyclopropylcarbinyl) cation.
Oxocarbenium and iminium ions have important secondary canonical forms (resonance structures) in which carbon bears a positive charge. As such, they are carbocations according to the IUPAC definition although some chemists do not regard them to be "true" carbocations, as their most important resonance contributors carry the formal positive charge on an oxygen or nitrogen atom, respectively.

Aromatic carbenium ions
[edit]
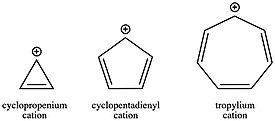
The tropylium ion is an aromatic species with the formula C
7H+
7.[22] Its name derives from the molecule tropine (itself named for the molecule atropine). Salts of the tropylium cation can be stable, e.g. tropylium tetrafluoroborate. It can be made from cycloheptatriene (tropylidene) and bromine or phosphorus pentachloride.[23]
It is a planar, cyclic, heptagonal ion; it also has 6 π-electrons (4n + 2, where n = 1), which fulfills Hückel's rule of aromaticity. It can coordinate as a ligand to metal atoms.
The structure shown is a composite of seven resonance contributors in which each carbon carries part of the positive charge.
In 1891 G. Merling obtained a water-soluble salt from a reaction of cycloheptatriene and bromine.[24] The structure was elucidated by Eggers Doering and Knox in 1954.[25][26]
On the other hand, the antiaromatic cyclopentadienyl cation (C5H+5) is destabilized by some 40 kcal/mol.
Another aromatic carbenium ion is the cyclopropenyl or cyclopropenium ion, C
3H+
3.[27] Although less stable than the tropylium cation, this carbenium ion can also form salts at room temperature. Solutions of such salts were exhibit conventional spectroscopic and chemical properties. The cyclopropenium cation (C3H+3), although somewhat destabilized by angle strain, is still clearly stabilized by aromaticity when compared to its open-chain analog, allyl cation.
These varying cation stabilities, depending on the number of π electrons in the ring system, can furthermore be crucial factors in reaction kinetics. The formation of an aromatic carbocation is much faster than the formation of an anti-aromatic or open-chain carbocation.
Arenium ions
[edit]An arenium ion is a cyclohexadienyl cation that appears as a reactive intermediate in electrophilic aromatic substitution.[28] For historic reasons this complex is also called a Wheland intermediate,[29] or a σ-complex.
Two hydrogen atoms bonded to one carbon lie in a plane perpendicular to the benzene ring.[30] The arenium ion is no longer an aromatic species; however it is relatively stable due to delocalization: the positive charge is delocalized over 5 carbon atoms. Also contributing to the stability of arenium ions is the energy gain resulting from the strong C-e bond (E = electrophile).
The smallest arenium ion is protonated benzene, C
6H+
7. The benzenium ion can be isolated as a stable compound when benzene is protonated by the carborane superacid, H(CB11H(CH3)5Br6).[31] The benzenium salt is crystalline with thermal stability up to 150 °C. Bond lengths deduced from X-ray crystallography are consistent with a cyclohexadienyl cation structure.
Acylium ions
[edit]An acylium ion is a cation with the formula RCO+.[32] The structure is described as R−C≡O+ or R−=O. It is an acyl carbocation, but the actual structure has the oxygen and carbon linked by a triple bond. Such species are common reactive intermediates, for example, in the Friedel−Crafts acylations also in many other organic reactions such as the Hayashi rearrangement. Salts containing acylium ions can be generated by removal of the halide from acyl halides:
- RCOCl + SbCl5 → RCO+SbCl−
6
The C–O distance in these cations is near 1.1 ångströms, even shorter than that in carbon monoxide.[33] Acylium cations are characteristic fragments observed in EI-mass spectra of ketones.
Vinyl and alkynyl carbenium ions
[edit]Based on hydride ion affinity, the parent vinyl cation is less stable than even a primary sp2-hybridized carbocation, while an α alkyl-substituted vinyl cation has a stability that is comparable to the latter. Hence, vinyl cations are relatively uncommon intermediates. They can be generated by the ionization of a vinyl electrophile, provided the leaving group is sufficiently good (e.g., TfO−, IPh, or N2). They have been implicated as intermediates in some vinyl substitution reactions (designated as SN1(vinyl)) and as intermediates in the electrophilic addition reactions of arylalkynes. With the exception of the parent vinyl cation, which is believed to be a bridged species, and geometrically constrained cyclic vinyl cations, most vinyl cations take on sp hybridization and are linear.
Aryl cations are less stable than vinyl cations due to the ring-enforced distortion to a nonlinear geometry and approximately sp2-character of the unoccupied orbital. Only N2 in aryldiazonium salts is a good enough leaving group for the chemical generation of aryl cations.[34]
Alkynyl cations are extremely unstable, much less stable than even CH+
3 (hydride ion affinity 386 kcal/mol versus 312 kcal/mol for CH+
3) and cannot be generated by purely chemical means. They can, however, be generated radiochemically via the beta decay of tritium:[35]
Selected applications
[edit]
Carbenium ions are so integrated into organic chemistry that a full inventory of their commercially useful reactions would be long. For example, catalytic cracking, a major step in petroleum refining involves carbenium ion intermediates.[36]
The alkylation of benzene with alpha-olefins to give linear alkylbenzene (LABs) illustrates the behaviour of secondary carbenium ions. The alkylation is initiated by strong acids. LABs are a key precursor to detergents.
Derivatives of the triphenylcarbenium are the triarylmethane dyes.[37]
Acylium ions are intermediates in Friedel-Crafts acylations and Koch reactions.
See also
[edit]References
[edit]- ^ Scholz, Franziska; Himmel, Daniel; Scherer, Harald; Krossing, Ingo (2013). "Superacidic or Not…︁? Synthesis, Characterisation, and Acidity of the Room-Temperature Ionic Liquid [C(CH3)3]+ [Al2Br7]−". Chemistry – A European Journal. 19 (1): 109–116. doi:10.1002/chem.201203260. PMID 23180742.
- ^ Thomas H. Lowery; Kathleen Schueller Richardson (1981). Mechanism and Theory in Organic Chemistry, Second Edition. Harper and Rowe. p. 396. ISBN 0-06-044083-X.
- ^ March
- ^ Carey, Francis A. (2007). Advanced Organic Chemistry: Part A: Structure and Mechanisms. Sundberg, Richard J. (5th ed.). New York: Springer. p. 440. ISBN 9780387448978. OCLC 154040953.
- ^ Scholz, F.; Himmel, D.; Heinemann, F. W.; Schleyer, P. v R.; Meyer, K.; Krossing, I. (2013-07-05). "Crystal Structure Determination of the Nonclassical 2-Norbornyl Cation". Science. 341 (6141): 62–64. Bibcode:2013Sci...341...62S. doi:10.1126/science.1238849. ISSN 0036-8075. PMID 23828938. S2CID 206549219.
- ^ Carey, Francis A. (2007). Advanced Organic Chemistry: Part A: Structure and Mechanisms. Sundberg, Richard J. (5th ed.). New York: Springer. p. 436. ISBN 9780387448978. OCLC 154040953.
- ^ Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, p. 239, ISBN 978-0-471-72091-1
- ^ Hansjörg Grützmacher, Christina M. Marchand (1997), "Heteroatom stabilized carbenium ions", Coord. Chem. Rev., 163, 287–344. doi:10.1016/S0010-8545(97)00043-X
- ^ Anslyn, Eric V.; Dougherty, Dennis A. (2000). Modern Physical Organic Chemistry. Sausalito, CA: University Science Books. ISBN 978-1891389313.
- ^ Carey, Francis A. (2007). Advanced Organic Chemistry: Part A: Structure and Mechanisms. Sundberg, Richard J. (5th ed.). New York: Springer. p. 300-301. ISBN 9780387448978. OCLC 154040953.
- ^ Carroll, Felix A. (2010). Perspectives on structure and mechanism in organic chemistry (2nd ed.). Hoboken, N.J.: John Wiley. ISBN 9780470276105. OCLC 286483846.
- ^ Olah, George A.; O'Brien, Daniel H.; White, Anthony Mallinson. (October 1967). "Stable carbonium ions. LII. Protonated esters and their cleavage in fluorosulfonic acid-antimony pentafluoride solution". Journal of the American Chemical Society. 89 (22): 5694–5700. doi:10.1021/ja00998a036. ISSN 0002-7863.
- ^ Carey, Francis A. (2007). Advanced Organic Chemistry. Sundberg, Richard J. (5th ed.). New York: Springer. ISBN 9780387448978. OCLC 154040953.
- ^ Lowry, Thomas H. (1987). Mechanism and theory in organic chemistry. Richardson, Kathleen Schueller. (3rd ed.). New York: Harper & Row. ISBN 0060440848. OCLC 14214254.
- ^ Carey, Francis A. (2007). Advanced Organic Chemistry: Part A: Structure and Mechanisms. Sundberg, Richard J. (5th ed.). New York: Springer. p. 300-301. ISBN 9780387448978. OCLC 154040953.
- ^ Schultz, Jocelyn C.; Houle, F. A.; Beauchamp, J. L. (July 1984). "Photoelectron spectroscopy of 1-propyl, 1-butyl, isobutyl, neopentyl, and 2-butyl radicals: free radical precursors to high-energy carbonium ion isomers". Journal of the American Chemical Society. 106 (14): 3917–3927. doi:10.1021/ja00326a006. ISSN 0002-7863.
- ^ Yamataka, Hiroshi; Ando, Takashi; Nagase, Shigeru; Hanamura, Mitsuyasu; Morokuma, Keiji (February 1984). "Ab initio MO calculations of isotope effects in model processes of neopentyl ester solvolysis". The Journal of Organic Chemistry. 49 (4): 631–635. doi:10.1021/jo00178a010. ISSN 0022-3263.
- ^ George A. Olah and Joachim Lukas (1967), "Stable Carbonium Ions. XLVII. Alkylcarbonium ion formation from alkanes via hydride (alkide) ion abstraction in fluorosulfonic acid-antimony pentafluoride-sulfuryl chlorofluoride solution". J. Am. Chem. Soc. 89 (18), 4739–4744 doi:10.1021/ja00994a030
- ^ Kato, Tsuyoshi; Reed, Christopher A. (2004). "Putting tert -Butyl Cation in a Bottle". Angewandte Chemie International Edition. 43 (22): 2908–2911. doi:10.1002/anie.200453931. PMID 15170300.
- ^ Carey, Francis A. (2007). Advanced Organic Chemistry: Part A: Structure and Mechanisms. Sundberg, Richard J. (5th ed.). New York: Springer. p. 300-301. ISBN 9780387448978. OCLC 154040953.
- ^ Carey, Francis A. (2007). Advanced Organic Chemistry: Part A: Structure and Mechanisms. Sundberg, Richard J. (5th ed.). New York: Springer. p. 426-427. ISBN 9780387448978. OCLC 154040953.
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "molecule". doi:10.1351/goldbook.M04002
- ^ "Tropylium tetrafluorate" Organic Syntheses, Coll. Vol. 5, p.1138 (1973); Vol. 43, p.101 (1963). link Archived 2012-08-29 at the Wayback Machine
- ^ Merling, G. (1891), "Ueber Tropin". Berichte der deutschen chemischen Gesellschaft, 24: 3108–3126. doi:10.1002/cber.189102402151
- ^ "The Cycloheptatrienylium (Tropylium) Ion" W. von E. Doering, L. H. Knox J. Am. Chem. Soc., 1954, 76 (12), pp 3203–3206 doi:10.1021/ja01641a027
- ^ "Aromaticity as a Cornerstone of Heterocyclic Chemistry" Alexandru T. Balaban, Daniela C. Oniciu, Alan R. Katritzky Chem. Rev., 2004, 104 (5), 2777–2812 doi:10.1021/cr0306790
- ^ "Cyclopropenyl Cation. Synthesis and Characterization." R. Breslow and J. T. Groves J. Am. Chem. Soc., 1970, 92 (4), 984–987 [1]
- ^ Olah, George A. (1972). "Stable carbocations. CXVIII. General concept and structure of carbocations based on differentiation of trivalent (classical) carbenium ions from three-center bound penta- of tetracoordinated (nonclassical) carbonium ions. Role of carbocations in electrophilic reactions". Journal of the American Chemical Society. 94 (3): 808–820. doi:10.1021/ja00758a020. ISSN 0002-7863.
- ^ "A Quantum Mechanical Investigation of the Orientation of Substituents in Aromatic Molecules" G. W. Wheland J. Am. Chem. Soc.; 1942; 64(4) 900–908; doi:10.1021/ja01256a047
- ^ A guidebook to mechanism in organic chemistry, Peter Sykes; pp 130–133
- ^ "Isolating Benzenium Ion Salts" Christopher A. Reed, Kee-Chan Kim, Evgenii S. Stoyanov, Daniel Stasko, Fook S. Tham, Leonard J. Mueller, and Peter D. W. Boyd J. Am. Chem. Soc.; 2003; 125(7) 1796–1804; doi:10.1021/ja027336o
- ^ Compendium of Chemical Terminology, acyl groups
- ^ Chevrier, B.; Le Carpentier, J. M.; Weiss, R. (1972). "Synthesis of two crystalline species of the Friedel–Crafts intermediate antimony pentachloride-p-toluoyl chloride. Crystal structures of the donor–acceptor complex and of the ionic salt". J. Am. Chem. Soc. 94 (16): 5718–5723. doi:10.1021/ja00771a031.
- ^ Carey, Francis A. (2007). Advanced Organic Chemistry: Part A: Structure and Mechanisms. Sundberg, Richard J. (5th ed.). New York: Springer. p. 436. ISBN 9780387448978. OCLC 154040953.
- ^ Angelini, Giancarlo; Hanack, Michael; Vermehren, Jan; Speranza, Maurizio (1988-02-17). "Generation and trapping of an alkynyl cation". Journal of the American Chemical Society. 110 (4): 1298–1299. doi:10.1021/ja00212a052. ISSN 0002-7863.
- ^ Rahimi, Nazi; Karimzadeh, Ramin (2011). "Catalytic cracking of hydrocarbons over modified ZSM-5 zeolites to produce light olefins: A review". Applied Catalysis A: General. 398 (1–2): 1–17. doi:10.1016/j.apcata.2011.03.009.
- ^ Gessner, Thomas; Mayer, Udo (2000). "Triarylmethane and Diarylmethane Dyes". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a27_179. ISBN 978-3527306732.


![{\displaystyle {\ce {RC#CT -> [RC#C^3 He]+ + e-}}+{\bar {\nu }}_{e}\longrightarrow {\ce {RC#C+ + ^{3}He + e-}}+{\bar {\nu }}_{e}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/b407467d1926148009140dc78110535945708543)