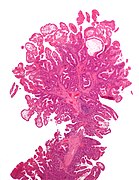
Colorectal polyp
| Colon polyps | |
|---|---|
 | |
| Polyp of sigmoid colon as revealed by colonoscopy. Approximately 1 cm in diameter. The polyp was removed by snare cautery. | |
| Specialty | Gastroenterology |
A colorectal polyp is a polyp (fleshy growth) occurring on the lining of the colon or rectum.[1] Untreated colorectal polyps can develop into colorectal cancer.[2]
Colorectal polyps are often classified by their behaviour (i.e. benign vs. malignant) or cause (e.g. as a consequence of inflammatory bowel disease). They may be benign (e.g. hyperplastic polyp), pre-malignant (e.g. tubular adenoma) or malignant (e.g. colorectal adenocarcinoma).
Signs and symptoms
[edit]Colorectal polyps are not usually associated with symptoms.[2] When they occur, symptoms include bloody stools; changes in frequency or consistency of stools (such as a week or more of constipation or diarrhoea);[3] and fatigue arising from blood loss.[2] Anemia arising from iron deficiency can also present due to chronic blood loss, even in the absence of bloody stools.[3][4] Another symptom may be an increased mucus production especially those involving villous adenomas.[4] Copious production of mucus causes loss of potassium that can occasionally result in symptomatic hypokalemia.[4] Occasionally, if a polyp is big enough to cause a bowel obstruction, there may be nausea, vomiting and severe constipation.[3]
Structure
[edit]Polyps are either pedunculated (attached to the intestinal wall by a stalk) or sessile (grow directly from the wall).[5][6]: 1342 In addition to the gross appearance categorization, they are further divided by their histologic appearance as tubular adenoma which are tubular glands, villous adenoma which are long finger like projections on the surface, and tubulovillous adenoma which has features of both.[6]: 1342
Genetics
[edit]Hereditary syndromes causing increased colorectal polyp formation include:
- Familial adenomatous polyposis (FAP)[7]
- Hereditary nonpolyposis colorectal cancer
- Peutz–Jeghers syndrome
- Juvenile polyposis syndrome
Several genes have been associated with polyposis, such as GREM1, MSH3, MLH3, NTHL1, RNF43 and RPS20.[8]
Familial adenomatous polyposis
[edit]Familial adenomatous polyposis (FAP) is a form of hereditary cancer syndrome involving the APC gene located on chromosome q521.[9] The syndrome was first described in 1863 by Virchow on a 15-year-old boy with multiple polyps in his colon.[9] The syndrome involves development of multiple polyps at an early age and those left untreated will all eventually develop cancer.[9] The gene is expressed 100% in those with the mutation and it is autosomal dominant.[9] 10–20% of patients have negative family history and acquire the syndrome from spontaneous germline mutation.[9] The average age of newly diagnosed patient is 29 and the average age of newly discovered colorectal cancer is 39.[9] It is recommended that those affected undergo colorectal cancer screening at younger age with treatment and prevention are surgical with removal of affected tissues.[9]
Hereditary nonpolyposis colorectal cancer (Lynch Syndrome)
[edit]Hereditary nonpolyposis colorectal cancer (HNPCC, also known as Lynch syndrome) is a hereditary colorectal cancer syndrome.[9] It is the most common hereditary form of colorectal cancer in the United States and accounts for about 3% of all cases of cancer.[9] It was first recognized by Alder S. Warthin in 1885 at the University of Michigan.[9] It was later further studied by Henry Lynch who recognized an autosomal dominant transmission pattern with those affected having relatively early onset of cancer (mean age 44 years), greater occurrence of proximal lesions, mostly mucinous or poorly differentiated adenocarcinoma, greater number of synchronous and metachronous cancer cells, and good outcome after surgical intervention.[9] The Amsterdam Criteria were initially used to define Lynch syndrome before the underlying genetic mechanism had been worked out.[9] The Criteria required that the patient has three family members all first-degree relatives with colorectal cancer that involves at least two generations with at least one affected person being younger than 50 years of age when the diagnosis was made.[9] The Amsterdam Criteria is too restrictive and was later expanded to include cancers of endometrial, ovarian, gastric, pancreatic, small intestinal, ureteral, and renal pelvic origin.[9] The increased risk of cancer seen in patients with by the syndrome is associated with dysfunction of DNA repair mechanism.[9] Molecular biologists have linked the syndrome to specific genes such as hMSH2, hMSH1, hMSH6, and hPMS2.[9]
Peutz–Jeghers syndrome
[edit]Peutz–Jeghers syndrome is an autosomal dominant syndrome that presents with hamartomatous polyps, which are disorganized growth of tissues of the intestinal tract, and hyperpigmentation of the interlining of the mouth, lips and fingers.[9] The syndrome was first noted in 1896 by Hutchinson, and later separately described by Peutz, and then again in 1940 by Jeghers.[9] The syndrome is associated with malfunction of serine-threonine kinase 11 or STK 11 gene, and has a 2–10% increase in risk of developing cancer of the intestinal tract.[9] The syndrome also causes increased risk of extraintestinal cancer such as that involving breast, ovary, cervix, fallopian tubes, thyroid, lung, gallbladder, bile ducts, pancreas, and testicles.[9] The polyps often bleeds and may cause obstruction that would require surgery.[9] Any polyps larger than 1.5 cm needs removal and patients should be monitored closely and screen every two years for malignancy.[9]
Juvenile polyposis syndrome
[edit]Juvenile polyposis syndrome is an autosomal dominant syndrome characterized by increased risk of cancer of intestinal tract and extraintestinal cancer.[9] It often presents with bleeding and obstruction of the intestinal tract along with low serum albumin due to protein loss in the intestine.[9] The syndrome is linked to malfunction of SMAD4 a tumor suppression gene that is seen in 50% of cases.[9] Individuals with multiple juvenile polyps have at least 10% chance of developing malignancy and should undergo abdominal colectomy with ileorectal anastomosis, and close monitoring via endoscopy of rectum.[9] For individuals with few juvenile polyps, patients should undergo endoscopic polypectomy.[9]
Types
[edit]
Colorectal polyps can broadly be classified as follows:
- hyperplastic,
- neoplastic (adenomatous and malignant),
- hamartomatous and,
- inflammatory.
Comparison table
[edit]| Type | Risk of containing malignant cells | Histopathology | Image | |
|---|---|---|---|---|
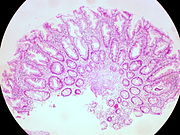
| Hyperplastic polyp | 0% | No dysplasia.[10]
|
| |
| Tubular adenoma | 2% at 1.5 cm[12] | Low to high grade dysplasia[13] | Over 75% of volume has tubular appearance.[14] | 
|
| Tubulovillous adenoma | 20% to 25%[15] | 25–75% villous[14] | 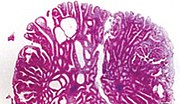
| |
| Villous adenoma | 15%[16] to 40%[15] | Over 75% villous[14] | 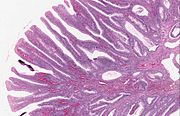
| |
| Sessile serrated adenoma (SSA)[17] |
|
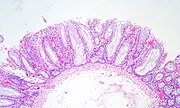
| ||
| Colorectal adenocarcinoma | 100% |
|
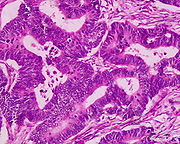
| |
Hyperplastic polyp
[edit]Most hyperplastic polyps are found in the distal colon and rectum.[18] They have no malignant potential,[18] which means that they are no more likely than normal tissue to eventually become a cancer.
Neoplastic polyp
[edit]A neoplasm is a tissue whose cells have lost normal differentiation. They can be either benign growths or malignant growths. The malignant growths can either have primary or secondary causes. Adenomatous polyps are considered precursors to cancer and cancer becomes invasive once malignant cells cross the muscularis mucosa and invade the cells below.[9] Any cellular changes seen above the lamina propria are considered non-invasive and are labeled atypia or dysplasia. Any invasive carcinoma that has penetrated the muscularis mocos has the potential for lymph node metastasis and local recurrence which will require more aggressive and extensive resection.[9] The Haggitt's criteria is used for classification of polyps containing cancer and is based on the depth of penetration.[9] The Haggitt's criteria has level 0 through level 4, with all invasive carcinoma of sessile polyp variant by definition being classified as level 4.[9]
- Level 0: Cancer does not penetrate through the muscularis mucosa.[9]
- Level 1: Cancer penetrates through the muscularis mucosa and invades the submucosa below but is limited to the head of the polyp.[9]
- Level 2: Cancer invades through with involvement of the neck of polyp.[9]
- Level 3: Cancer invades through with involvement of any parts of the stalk.[9]
- Level 4: Cancer invades through the submucosa below the stalk of the polyp but above the muscularis propria of the bowel wall.[9]
Adenomas
[edit]Neoplastic polyps of the bowel are often benign hence called adenomas. An adenoma is a tumor of glandular tissue, that has not (yet) gained the properties of cancer.[citation needed]
The common adenomas of the colon (colorectal adenoma) are the tubular, tubulovillous, villous, and sessile serrated (SSA).[18] A large majority (65–80%) are of the benign tubular type with 10–25% being tubulovillous, and villous being the most rare at 5–10%.[9]
As is evident from their name, sessile serrated and traditional serrated adenomas (TSAs) have a serrated appearance and can be difficult to distinguish microscopically from hyperplastic polyps.[18] Making this distinction is important, however, since SSAs and TSAs have the potential to become cancers,[19] while hyperplastic polyps do not.[18]
The villous subdivision is associated with the highest malignant potential because they generally have the largest surface area. (This is because the villi are projections into the lumen and hence have a bigger surface area.) However, villous adenomas are no more likely than tubular or tubulovillous adenomas to become cancerous if their sizes are all the same.[18]
Hamartomatous polyp
[edit]Hamartomatous polyps are tumours, like growths found in organs as a result of faulty development. They are normally made up of a mixture of tissues. They contain mucus-filled glands, with retention cysts, abundant connective tissue, and chronic cellular infiltration of eosinophils.[20] They grow at the normal rate of the host tissue and rarely cause problems such as compression. A common example of a hamartomatous lesion is a strawberry naevus. Hamartomatous polyps are often found by chance; occurring in syndromes such as Peutz–Jegher syndrome or Juvenile polyposis syndrome.
Peutz–Jeghers syndrome is associated with polyps of the GI tract and also increased pigmentation around the lips, genitalia, buccal mucosa feet and hands. People are often diagnosed with Peutz–Jegher after presenting at around the age of nine with an intussusception. The polyps themselves carry little malignant potential but because of potential coexisting adenomas there is a 15% chance of colonic malignancy.
Juvenile polyps are hamartomatous polyps that often become evident before twenty years of age, but can also be seen in adults. They are usually solitary polyps found in the rectum which most commonly present with rectal bleeding. Juvenile polyposis syndrome is characterised by the presence of more than five polyps in the colon or rectum, or numerous juvenile polyps throughout the gastrointestinal tract, or any number of juvenile polyps in any person with a family history of juvenile polyposis. People with juvenile polyposis have an increased risk of colon cancer.[19]
Inflammatory polyp
[edit]These are polyps that are associated with inflammatory conditions such as ulcerative colitis and Crohn's disease.[citation needed]
Prevention
[edit]Diet and lifestyle are believed to play a large role in whether colorectal polyps form. Studies show there to be a protective link between consumption of cooked green vegetables, brown rice, legumes, and dried fruit and decreased incidence of colorectal polyps.[21]
Diagnosis
[edit]Colorectal polyps can be detected using a faecal occult blood test, flexible sigmoidoscopy, colonoscopy, virtual colonoscopy, digital rectal examination, barium enema or a pill camera.[3][failed verification]
Malignant potential is associated with
- degree of dysplasia
- Type of polyp (e.g. villous adenoma):
- Tubular adenoma: 5% risk of cancer
- Tubulovillous adenoma: 20% risk of cancer
- Villous adenoma: 40% risk of cancer
- Size of polyp:
Normally an adenoma that is greater than 0.5 cm is treated.
Gallery
[edit]
-
Microvesicular hyperplastic polyp. H&E stain.
-
Microvesicular hyperplastic polyp. H&E stain.
-
Traditional serrated adenoma. H&E stain.
-
Gross appearance of a colectomy specimen containing two colorectal polyps and one invasive colorectal carcinoma
-
Micrograph of a tubular adenoma, the most common type of dysplastic polyp in the colon
-
Micrograph of a tubular adenoma – dysplastic epithelium (dark purple) on left of image; normal epithelium (blue) on right. H&E stain.
-
Micrograph of a villous adenoma. These polyps are considered to have a high risk of malignant transformation. H&E stain.
-
Paris classification of colorectal neoplasms[23]
NICE classification
[edit]In colonoscopy, colorectal polyps can be classified by NICE (Narrow-band imaging International Colorectal Endoscopic):[24]
| Type 1 | Type 2 | Type 3 | |
|---|---|---|---|
| Color | Same or lighter than background | Browner than background | Browner or darkly browner than background, sometimes patchy whiter areas |
| Vessels | None, or isolated lacy vessels coursing across the lesion | Brown vessels surrounding white structures | Area of disrupted or missing vessels |
| Surface Pattern | Homogenous, or dark or white spots of uniform size | Oval, tubular or branched white structures surrounded by brown vessels | Amorphous or absent surface pattern |
| Most likely pathology | Hyperplasia | Adenoma | Deep submucosal invasive cancer |
| Treatment | Follow up | Mucosal or submucosal polypectomy | Surgical operation |
Treatment
[edit]Polyps can be removed during a colonoscopy or sigmoidoscopy using a wire loop that cuts the stalk of the polyp and cauterises it to prevent bleeding.[3][failed verification] Many "defiant" polyps—large, flat, and otherwise laterally spreading adenomas—may be removed endoscopically by a technique called endoscopic mucosal resection (EMR), which involves injection of fluid underneath the lesion to lift it and thus facilitate endoscopic resection. Saline water may be used to generate lift, though some injectable solutions such as SIC 8000 may be more effective.[25] Minimally invasive surgery is indicated for polyps that are too large or in unfavorable locations, such as the appendix, that cannot be removed endoscopically.[26] These techniques may be employed as an alternative to the more invasive colectomy.[27]
Follow-up
[edit]By United States guidelines, the following follow-up is recommended:[28]
| Baseline colonoscopy finding | Recommended time until next colonoscopy |
|---|---|
| Normal | 10 years |
| 1–2 tubular adenomas <10 mm | 7–10 years |
| 3–4 tubular adenomas <10 mm | 3–5 years |
|
3 years |
| >10 adenomas on single examination | 1 years |
| Piecemeal resection of adenoma 20 mm | 6 months |
References
[edit]- ^ Marks, Jay W.; Anand, Bhupinder. "Colon Polyps: Symptoms, Causes, Cancer Risk, Treatment, and Prevention". Colon polyps center. MedicineNet. Retrieved 18 Jan 2020.
- ^ a b c Phillips, Michael M.; Zieve, David; Conaway, Brenda (September 25, 2019). "Colorectal polyps". Medical Encyclopedia. MedlinePlus. Retrieved 18 Jan 2020.
- ^ a b c d e "Colon polyps". Mayo Clinic. Mayo Foundation for Medical Education and Research. Retrieved 18 Jan 2020.
- ^ a b c Quick CR, Reed JB, Harper SJ, Saeb-Parsy K, Burkitt HG (2014). Essential Surgery: Problems, Diagnosis and Management. Edinburgh: Elsevier. ISBN 9780702054839. OCLC 842350865.[page needed]
- ^ Classen, Meinhard; Tytgat, G.N.J.; Lightdale, Charles J. (2002). Gastroenterological Endoscopy. Thieme. p. 303. ISBN 1-58890-013-4.
- ^ a b Townsend CM, Beauchamp RD, Evers BM, Mattox KL, eds. (2015). Sabiston Textbook of Surgery E-Book (19th ed.). Philadelphia, Pennsylvania: Elsevier Saunders. ISBN 978-1-4377-1560-6 – via Google Books (Preview).
- ^ "Familial Adenomatous Polyposis". The Lecturio Medical Concept Library. Retrieved 22 July 2021.
- ^ Valle L, de Voer RM, Goldberg Y, Sjursen W, Försti A, Ruiz-Ponte C, Caldés T, Garré P, Olsen MF, Nordling M, Castellvi-Bel S, Hemminmki K (October 2019). "Update on genetic predisposition to colorectal cancer and polyposis". Molecular Aspects of Medicine. 69. Elsevier: 10–26. doi:10.1016/j.mam.2019.03.001. hdl:2445/171217. PMID 30862463.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae af ag ah ai aj ak Mahmoud, Najjia N.; Bleier, Joshua I.S.; Aarons, Cary B.; Paulson, E. Carter; Shanmugan, Skandan; Fry, Robert D. (2017). Sabiston Textbook of Surgery (20th ed.). Elsevier. ISBN 9780323401630.[page needed]
- ^ a b c Finlay A Macrae. "Overview of colon polyps". UpToDate. This topic last updated: Dec 10, 2018.
- ^ a b c Robert V Rouse (2010-01-31). "Hyperplastic Polyp of the Colon and Rectum". Stanford University School of Medicine. Archived from the original on 2019-12-11. Retrieved 2019-10-31. Last updated 6/2/2015
- ^ Minhhuyen Nguyen. "Polyps of the Colon and Rectum". MSD Manual. Last full review/revision June 2019
- ^ Robert V Rouse. "Adenoma of the Colon and Rectum". Archived from the original on 2019-09-11. Retrieved 2019-10-31. Original posting/last update : 1/31/10, 1/19/14
- ^ a b c Bosman, F. T. (2010). WHO classification of tumours of the digestive system. Lyon: International Agency for Research on Cancer. ISBN 978-92-832-2432-7. OCLC 688585784.
- ^ a b Amersi, Farin; Agustin, Michelle; Ko, Clifford Y (2005). "Colorectal Cancer: Epidemiology, Risk Factors, and Health Services". Clinics in Colon and Rectal Surgery. 18 (3): 133–140. doi:10.1055/s-2005-916274. ISSN 1531-0043. PMC 2780097. PMID 20011296.
- ^ Alnoor Ramji (22 December 2022). "Villous Adenoma Follow-up". Medscape. Updated: Oct 24, 2016
- ^ Rosty, C; Hewett, D. G.; Brown, I. S.; Leggett, B. A.; Whitehall, V. L. (2013). "Serrated polyps of the large intestine: Current understanding of diagnosis, pathogenesis, and clinical management". Journal of Gastroenterology. 48 (3): 287–302. doi:10.1007/s00535-012-0720-y. PMC 3698429. PMID 23208018.
- ^ a b c d e f Kumar, Vinay (2010). "17—Polyps". Robbins and Cotran pathologic basis of disease (8th ed.). Philadelphia, PA: Saunders/Elsevier. ISBN 978-1-4160-3121-5.
- ^ a b Stoler, Mark A.; Mills, Stacey E.; Carter, Darryl; Joel K. Greenson; Reuter, Victor E. (2009). Sternberg's Diagnostic Surgical Pathology. Hagerstwon, MD: Lippincott Williams & Wilkins. ISBN 978-0-7817-7942-5.[page needed]
- ^ Calva, Daniel; Howe, James R (2008). "Hamartomatous Polyposis Syndromes". Surgical Clinics of North America. 88 (4): 779–817, vii. doi:10.1016/j.suc.2008.05.002. PMC 2659506. PMID 18672141.
- ^ Tantamango, Yessenia M; Knutsen, Synnove F; Beeson, W Lawrence; Fraser, Gary; Sabate, Joan (2011). "Foods and Food Groups Associated with the Incidence of Colorectal Polyps: The Adventist Health Study". Nutrition and Cancer. 63 (4): 565–72. doi:10.1080/01635581.2011.551988. PMC 3427008. PMID 21547850.
- ^ a b c Summers, Ronald M (2010). "Polyp Size Measurement at CT Colonography: What Do We Know and What Do We Need to Know?". Radiology. 255 (3): 707–20. doi:10.1148/radiol.10090877. PMC 2875919. PMID 20501711.
- ^ Luis Bujanda Fernández de Piérola, Joaquin Cubiella Fernández, Fernando Múgica Aguinaga, Lander Hijona Muruamendiaraz and Carol Julyssa Cobián Malaver (2013). "Malignant Colorectal Polyps: Diagnosis, Treatment and Prognosis". Colonoscopy and Colorectal Cancer Screening: Future Directions. doi:10.5772/52697. ISBN 9789535109495.
{{cite book}}: CS1 maint: multiple names: authors list (link) Creative Commons Attribution 3.0 License - ^ Hattori, Santa (2014). "Narrow-band imaging observation of colorectal lesions using NICE classification to avoid discarding significant lesions". World Journal of Gastrointestinal Endoscopy. 6 (12): 600–605. doi:10.4253/wjge.v6.i12.600. PMC 4265957. PMID 25512769.
- ^ Hoff, RT; Lakha, A (1 February 2020). "Rectal Tubulovillous Adenoma". The Journal of the American Osteopathic Association. 120 (2): 121. doi:10.7556/jaoa.2020.024. PMID 31985762.
- ^ Fisichella, M. (2016). "Laparoscopic Cecal Wedge Resection Appendectomy". J Med Ins: 207. doi:10.24296/jomi/207.
- ^ Saunders B, Ginsberg GG, Bjorkman DJ (2008). "How I do it: Removing large or sessile colonic polyps" (PDF). Munich: OMED. Archived from the original (PDF) on 2008-04-11.
- ^ Gupta, Samir; Lieberman, David; Anderson, Joseph C.; Burke, Carol A.; Dominitz, Jason A.; Kaltenbach, Tonya; Robertson, Douglas J.; Shaukat, Aasma; Syngal, Sapna; Rex, Douglas K. (2020). "Recommendations for Follow-Up After Colonoscopy and Polypectomy: A Consensus Update by the US Multi-Society Task Force on Colorectal Cancer". Gastrointestinal Endoscopy. 91 (3): 463–485.e5. doi:10.1016/j.gie.2020.01.014. ISSN 0016-5107. PMC 7389642. PMID 32044106.