Diethylamine

| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
N-Ethylethanamine | |
| Other names
(Diethyl)amine
Diethylamine (deprecated[2]) | |
| Identifiers | |
3D model (JSmol)
|
|
| 605268 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.003.380 |
| EC Number |
|
| MeSH | diethylamine |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1154 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H11N | |
| Molar mass | 73.139 g·mol−1 |
| Appearance | Colourless liquid |
| Odor | fishy, ammonical |
| Density | 0.7074 g mL−1 |
| Melting point | −49.80 °C; −57.64 °F; 223.35 K |
| Boiling point | 54.8 to 56.4 °C; 130.5 to 133.4 °F; 327.9 to 329.5 K |
| Miscible | |
| log P | 0.657 |
| Vapor pressure | 24.2–97.5 kPa |
Henry's law
constant (kH) |
150 μmol Pa−1 kg−1 |
| Acidity (pKa) | 10.98 (of ammonium form) |
| -56.8·10−6 cm3/mol | |
Refractive index (nD)
|
1.385 |
| Thermochemistry | |
Heat capacity (C)
|
178.1 J K−1 mol−1 |
Std enthalpy of
formation (ΔfH⦵298) |
−131 kJ mol−1 |
Std enthalpy of
combustion (ΔcH⦵298) |
−3.035 MJ mol−1 |
| Hazards | |
| GHS labelling: | |
  
| |
| Danger | |
| H225, H302, H312, H314, H332 | |
| P210, P280, P305+P351+P338, P310 | |
| NFPA 704 (fire diamond) | |
| Flash point | −23 °C (−9 °F; 250 K) |
| 312 °C (594 °F; 585 K) | |
| Explosive limits | 1.8–10.1% |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
540 mg/kg (rat, oral) 500 mg/kg (mouse, oral)[4] |
LC50 (median concentration)
|
4000 ppm (rat, 4 hr)[4] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 25 ppm (75 mg/m3)[3] |
REL (Recommended)
|
TWA 10 ppm (30 mg/m3) ST 25 ppm (75 mg/m3)[3] |
IDLH (Immediate danger)
|
200 ppm[3] |
| Safety data sheet (SDS) | hazard.com |
| Related compounds | |
Related amines
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Diethylamine is an organic compound with the formula (CH3CH2)2NH. It is classified as a secondary amine. It is a flammable, volatile weakly alkaline liquid that is miscible with most solvents. It is a colorless liquid, but commercial samples often appear brown due to impurities. It has a strong ammonia-like odor.
Production and uses
[edit]The alumina-catalyzed reaction makes diethylamine from ethanol and ammonia. Diethylamine is obtained together with ethylamine and triethylamine. Annual production of the three ethylamines was estimated in 2000 to be 80,000,000 kg.[5]
Diethylamine is used in the production of corrosion inhibitor N,N-diethylaminoethanol, by reaction with ethylene oxide. It is also a precursor to a wide variety of other commercial products. It is also sometimes used in the illicit production of LSD.[6]
Organic chemistry
[edit]As the most abundantly available secondary amine that is liquid at room temperature, diethylamine has been extensively deployed in chemical synthesis. Its reactions illustrate the pattern seen for many other dialkylamines. It participates in Mannich reactions involving the installation of diethylaminomethyl substituents.[7][8][9] Alkylation gives the tertiary amine.[10] With trimethylsilyl chloride, it reacts to give the silylamide.[11]
Supramolecular structure
[edit]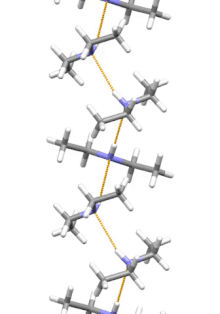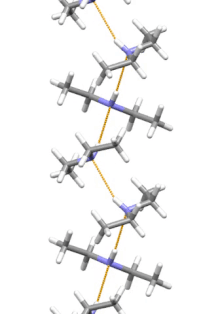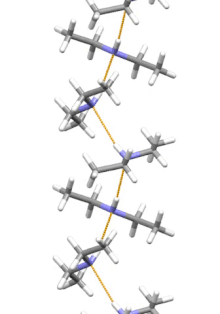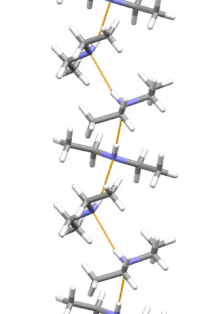
Diethylamine is the smallest and simplest molecule that features a supramolecular helix as its lowest energy aggregate. Other similarly sized hydrogen-bonding molecules favor cyclic structures.[12]
Safety
[edit]Diethylamine has low toxicity, but the vapor causes transient impairment of vision.[5]
References
[edit]- ^ Merck Index, 12th Edition, 3160
- ^ Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 671. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0209". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b "Diethylamine". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ a b Karsten Eller, Erhard Henkes, Roland Rossbacher, Hartmut Höke (2005). "Amines, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a02_001. ISBN 978-3527306732.
{{cite encyclopedia}}: CS1 maint: multiple names: authors list (link) - ^ Shulgin, Alexander. "Erowid Online Books:"TIHKAL" - #26 LSD-25". www.erowid.org. Retrieved 12 August 2019.
- ^ Charles E. Maxwell (1943). "β-Diethylaminomethylacetophenone Hydrochloride". Organic Syntheses. 23: 30. doi:10.15227/orgsyn.023.0030.
- ^ C. F. H. Allen and J. A. VanAllan (1947). "Diethylaminoacetonitrile". Organic Syntheses. 27: 20. doi:10.15227/orgsyn.027.0020.
- ^ Alfred L. Wilds, Robert M. Nowak, Kirtland E. McCaleb (1957). "1-Diethylamino-3-Butanone". Organic Syntheses. 37: 18. doi:10.15227/orgsyn.037.0018.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ W. W. Hartman (1934). "β-Diethylaminoethyl Alcohol". Organic Syntheses. 14: 28. doi:10.15227/orgsyn.014.0028.
- ^ W. J. Middleton, E. M. Bingham (1977). "Diethylaminosulfur Trifluoride". Organic Syntheses. 57: 50. doi:10.15227/orgsyn.057.0050.
- ^ Felix Hanke; Chloe J. Pugh; Ellis F. Kay; Joshua B. Taylor; Stephen M. Todd; Craig M. Robertson; Benjamin J. Slater; Alexander Steiner (2018). "The simplest supramolecular helix". Chemical Communications. 54 (47): 6012–6015. doi:10.1039/C8CC03295E. PMID 29796532.

