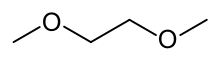
Dimethoxyethane
| |

| |
| Names | |
|---|---|
| IUPAC name
1,2-Dimethoxyethane
| |
| Other names
DME, glyme, Ethylene glycol dimethyl ether, monoglyme,
dimethyl glycol, dimethyl cellosolve | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.003.451 |
| RTECS number |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H10O2 | |
| Molar mass | 90.122 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 0.8683 g/cm3 |
| Melting point | −58 °C (−72 °F; 215 K) |
| Boiling point | 85 °C (185 °F; 358 K) |
| miscible | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | −2 °C (28 °F; 271 K) |
| Related compounds | |
Related Ethers
|
Dimethoxymethane |
Related compounds
|
Ethylene glycol 1,4-Dioxane Diethylene glycol dimethyl ether |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Dimethoxyethane, also known as glyme, monoglyme, dimethyl glycol, ethylene glycol dimethyl ether, dimethyl cellosolve, and DME, is a clear, colorless, aprotic, and liquid ether that is used as a solvent, especially in batteries.[1] Dimethoxyethane is miscible with water.
Production
Monoglyme may be manufactured by a number of methods:[2]
- via the Williamson ether synthesis — reacting the sodium salt of 2-methoxyethanol (previously prepared by reaction with sodium) with chloromethane:
- 2 CH3OCH2CH2OH + 2 Na → 2 CH3OCH2CH2ONa + H2↑
- CH3OCH2CH2ONa + CH3Cl → CH3OCH2CH2OCH3 + NaCl
- via the alkylation of 2-methoxyethanol with dimethyl sulfate
- by the cleavage of ethylene oxide in presence of dimethyl ether. This reaction is catalyzed by Lewis acids (e.g. boron trifluoride or its complex with dimethyl ether). This route is not particularly selective and produces diglyme, triglyme, tetraglyme, and other glymes as a by-products. The reaction mixture is separated by distillation:
- CH3OCH3 + CH2CH2O → CH3OCH2CH2OCH3
Applications as solvent and ligand
Together with a high-permittivity solvent (e.g. propylene carbonate), dimethoxyethane is used as the low-viscosity component of the solvent for electrolytes of lithium batteries. In the laboratory, DME is used as a coordinating solvent.
Dimethoxyethane is often used as a higher boiling alternative to diethyl ether and THF. Dimethoxyethane forms chelate complexes with cations and acts as a bidentate ligand. It is therefore often used in organometallic chemistry like Grignard reactions, hydride reductions, and palladium-catalyzed reactions like Suzuki reactions and Stille couplings. Dimethoxyethane is also a good solvent for oligo- and polysaccharides.
References
- ^ D. Berndt, D. Spahrbier, "Batteries" in Ullmann’s Encyclopedia of Industrial Chemistry 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a03_343
- ^ Dimethoxyethane

