Magnesium stearate
| |
| Names | |
|---|---|
| IUPAC name
Magnesium octadecanoate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.008.320 |
| E number | E572 (acidity regulators, ...) |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
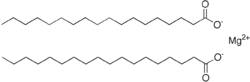
| Mg(C 18H 35O 2) 2 | |
| Molar mass | 591.27 g/mol |
| Appearance | light white powder |
| Odor | slight |
| Density | 1.026 g/cm3 |
| Melting point | 200 °C (392 °F; 473 K) |
| 0.003 g/100 mL (15 °C) 0.004 g/100 mL (25 °C) 0.008 g/100 mL (50 °C) | |
| Solubility | negligible in ether and alcohol slightly soluble in benzene |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 250 °C (482 °F; 523 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
> 1000 mg/kg (oral, rat) |
| Safety data sheet (SDS) | External MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Magnesium stearate, also called octadecanoic acid, magnesium salt, is a white substance, powder which becomes solid at room temperature. It has the chemical formula Mg(C
18H
35O
2)
2. It is a salt containing two equivalents of stearate (the anion of stearic acid) and one magnesium cation (Mg2+). Magnesium stearate melts at about 88 °C, is not soluble in water, and is generally considered safe for human consumption at levels below 2500 mg/kg per day.[1] In 1979, the FDA's Subcommittee on GRAS (generally recognized as safe) Substances (SCOGS) reported, "There is no evidence in the available information on ... magnesium stearate ... that demonstrates, or suggests reasonable grounds to suspect, a hazard to the public when they are used at levels that are now current and in the manner now practiced, or which might reasonably be expected in the future."[2]
Manufacturing
Magnesium stearate is created by the reaction of sodium stearate with magnesium sulfate.
Uses
Magnesium stearate is often used as an anti-adherent[3] in the manufacture of medical tablets, capsules and powders.[4] In this regard, the substance is also useful, because it has lubricating properties, preventing ingredients from sticking to manufacturing equipment during the compression of chemical powders into solid tablets; magnesium stearate is the most commonly used lubricant for tablets.[5] Magnesium stearate can also be used efficiently in dry coating processes.[6][7][8] Studies have shown that magnesium stearate may affect the release time of the active ingredients in tablets, etc., but not that it reduces the overall bioavailability of those ingredients.[9][10] As a food additive or pharmaceutical excipient, its E number is E470b.
Magnesium stearate is also used to bind sugar in hard candies like mints, and is a common ingredient in baby formulas. In pure powder form, the substance can be a dust explosion hazard, although this issue is effectively insignificant beyond the manufacturing plants using it.[11]
Magnesium stearate is manufactured from both animal and vegetable oils.[12] Some nutritional supplements specify that the magnesium stearate used is sourced from vegetables.[13]
Magnesium stearate is a major component of "bathtub rings." When produced by soap and hard water, magnesium stearate and calcium stearate both form a white solid insoluble in water, and are collectively known as "soap scum."[14]
References
- ^ D. Søndergaarda, O. Meyera and G. Würtzena (1980). "Magnesium stearate given peroprally to rats. A short term study". Toxicology. 17 (1): 51–55. doi:10.1016/0300-483X(80)90026-8. PMID 7434368.
- ^ FDA's SCOGS Database; Report No. 60; ID Code: 557-04-0; Year: 1979
- ^ Steve Ritter (2008). "What's That Stuff? Excipients: Inactive ingredients in medicines serve multiple functions in drug delivery". Chemical & Engineering News. 86 (1): 25. doi:10.1021/cen-v086n001.p025.
- ^ Sworbrick, James; James C. Boylan (1990). Encyclopedia of pharmaceutical technology. p. 2274. ISBN 9780824728243.
- ^ Weiner, Myra L.; Lois A. Kotkoskie (1999). Excipient Toxicity and Safety. p. 10. ISBN 9780824782108.
- ^ Ouabbas Y, Dodds J., Galet L., Chamayou A. , Baron M., Particle-particle coating in a cyclomix impact mixer, Powder Technol., 189 (2009), 245-252
- ^ Thomas G., Ouabbas Y., Grosseau P., Baron M., Chamayou A., Galet L., Modeling the main interaction forces between powder particles. Application to silica gel-magnesium stearate mixtures, Applied Surface Science, 255 (2009), 7500-7507
- ^ Sato A., Serris E., Grosseau P., Thomas G., Galet L., Chamayou A. , Baron M., Experiment and simulation of dry particle coating, Chem. Eng. Science, 86 (2013), 164-172
- ^ Alija Uzunović, Edina Vranić; "Effect Of Magnesium Stearate Concentration On Dissolution Properties Of Ranitidine Hydrochloride Coated Tablets"; Bosnian Journal Of Basic Medical Sciences, 2007, 7(3): 279-283
- ^ Natalie D. Eddington, Muhammad Ashraf, Larry L. Augsburger, James L. Leslie, Michael J. Fossler, Lawrence J. Lesko, Vinod P. Shah, Gurvinder Singh Rekhi; "Identification of Formulation and Manufacturing Variables That Influence In Vitro Dissolution and In Vivo Bioavailability of Propranolol Hydrochloride Tablets"; Pharmaceutical Development and Technology, Volume 3, Issue 4 November 1998 , pages 535–547
- ^ International Chemical Safety Card 1403
- ^ Gupta, Abhay; Hamad, Mazen L; Tawakkul, Mobin; Sayeed, Vilayat A; Khan, Mansoor A (June 2009). "Difference in the lubrication efficiency of bovine and vegetable-derived magnesium stearate during tabletting". AAPS PharmSciTech. 10 (2). Springer: 500–504. doi:10.1208/s12249-009-9229-y. ISSN 1530-9932. PMC 2690792. PMID 19390976. Retrieved 27 December 2015.
- ^ "Quick search results of the DSLD". Dietary Supplement Label Database. National Institutes of Health. Retrieved 27 December 2015.
- ^ Anne Marie Helmenstine; About.com: "Why Is It Harder to Rinse off Soap with Soft Water?"; retrieved 19 Mar 2010

