Magnus's green salt
| |
| |
| Names | |
|---|---|
| IUPAC name
Tetraammineplatinum(II) tetrachloroplatinate(II)
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.034.078 |
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| [Pt(NH3)4][PtCl4] | |
| Molar mass | 600.09 g/mol |
| Appearance | green solid |
| Density | 3.7 g/cm3 |
| Melting point | 320 °C (608 °F; 593 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
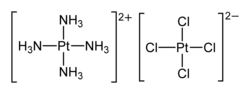
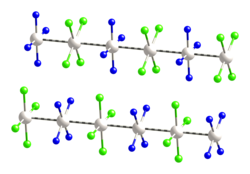
Magnus's green salt is the inorganic compound with the formula [Pt(NH3)4][PtCl4]. This salt is named after Heinrich Gustav Magnus, who, in the early 1830s, first reported the compound. The compound is a linear chain compound, consisting of a chain of platinum atoms. It is dark green, which is unusual for platinum compounds.
Structure
[edit]This species has attracted interest in materials chemistry and solid-state physics because of its one-dimensional structure. It contains a chain of alternating [PtCl4]2− anions and [Pt(NH3)4]2+ cations, in which the platinum atoms are separated by 3.25 Å.[1] It is a semiconductor.
Preparation
[edit]The compound may be prepared by combining aqueous solutions of [Pt(NH3)4]2+ and [PtCl4]2−, which gives a deep green solid precipitate.[2] Under some conditions, this reaction affords a pink polymorph of Magnus's green salt. In this so-called "Magnus's pink salt", the square planar Pt complexes are not stacked.[3]
Related compounds
[edit]Magnus's green salt has the same empirical formula as cis-PtCl2(NH3)2 ("Peyrone chloride") and trans-PtCl2(NH3)2.[4] These cis and trans compounds are molecules, whereas Magnus's green salt is a polymer. This difference is manifested by the solubility of the molecular complexes in water, whereas Magnus's green salt is insoluble.
Soluble analogues of Magnus's green salt can be prepared by replacing the ammonia with ethylhexylamine.[5][6]
The corresponding palladium compound ([Pd(NH3)4][PdCl4]) is known as "Vauquelin’s salt".
History
[edit]Magnus's green salt was one of the first examples of a metal ammine complex.
References
[edit]- ^ Atoji, Masao; Richardson, James W.; Rundle, R. E. (June 1957). "Pt(NH3)4PtCl41". Journal of the American Chemical Society. 79 (12): 3017–3020. doi:10.1021/ja01569a009.
- ^ Keller, R. N.; Moeller, Therald; Quagliano, J. V. (2007). "Tetrammineplatinum(II) Chloride: (Tetrammineplatinous Chloride)". Inorganic Syntheses. pp. 250–253. doi:10.1002/9780470132333.ch80. ISBN 978-0-470-13233-3.
- ^ Lucier, Bryan E. G.; Johnston, Karen E.; Xu, Wenqian; Hanson, Jonathan C.; Senanayake, Sanjaya D.; Yao, Siyu; Bourassa, Megan W.; Srebro, Monika; Autschbach, Jochen; Schurko, Robert W. (29 January 2014). "Unravelling the Structure of Magnus' Pink Salt" (PDF). Journal of the American Chemical Society. 136 (4): 1333–1351. doi:10.1021/ja4076277. PMID 24437378.
- ^ Wilson, Justin J.; Lippard, Stephen J. (23 April 2014). "Synthetic Methods for the Preparation of Platinum Anticancer Complexes". Chemical Reviews. 114 (8): 4470–4495. doi:10.1021/cr4004314. PMC 3999256. PMID 24283498.
- ^ Caseri, Walter (1 July 2004). "Derivatives of Magnus' Green Salt". Platinum Metals Review. 48 (3): 91–100. doi:10.1595/147106704X1504.
- ^ Bremi, Juliane; Caseri, Walter; Smith, Paul (2001). "A new compound derived from Magnus' green salt: solid state structure and evidence for platinum chains in solution". Journal of Materials Chemistry. 11 (10): 2593–2596. doi:10.1039/b104675f. S2CID 95456246. INIST 14086852.
