PLA2G2A
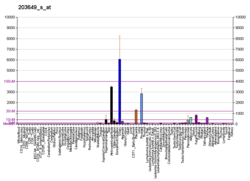
Appearance
Phospholipase A2, membrane associated is an enzyme that in humans is encoded by the PLA2G2A gene.[5][6]
See also
[edit]References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000188257 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000058908 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Tischfield JA, Xia YR, Shih DM, Klisak I, Chen J, Engle SJ, Siakotos AN, Winstead MV, Seilhamer JJ, Allamand V, Gyapay G, Lusis AJ (Feb 1997). "Low-molecular-weight, calcium-dependent phospholipase A2 genes are linked and map to homologous chromosome regions in mouse and human". Genomics. 32 (3): 328–33. doi:10.1006/geno.1996.0126. PMID 8838795.
- ^ "Entrez Gene: PLA2G2A Phospholipase A2, group IIA (platelets, synovial fluid)".
Further reading
[edit]- Kramer RM, Johansen B, Hession C, Pepinsky RB (1990). "Structure and Properties of a Secretable Phospholipase A2 from Human Platelets". Phospholipase A2. Advances in Experimental Medicine and Biology. Vol. 275. pp. 35–53. doi:10.1007/978-1-4684-5805-3_3. ISBN 978-1-4684-5807-7. PMID 2239446.
- Schröder HC, Perovic S, Kavsan V, Ushijima H, Müller WE (1998). "Mechanisms of prionSc- and HIV-1 gp120 induced neuronal cell death". Neurotoxicology. 19 (4–5): 683–8. PMID 9745929.
- Law MH, Cotton RG, Berger GE (2006). "The role of phospholipases A2 in schizophrenia". Mol. Psychiatry. 11 (6): 547–56. doi:10.1038/sj.mp.4001819. PMID 16585943. S2CID 13248313.
- Carlquist JF, Muhlestein JB, Anderson JL (2007). "Lipoprotein-associated phospholipase A2: a new biomarker for cardiovascular risk assessment and potential therapeutic target". Expert Rev. Mol. Diagn. 7 (5): 511–7. doi:10.1586/14737159.7.5.511. PMID 17892360. S2CID 23148887.
- Scott DL, White SP, Browning JL, Rosa J, Gelb M, Sigler P (1991). "Structures of free and inhibited human secretory phospholipase A2 from inflammatory exudate". Science. 254 (5034): 1007–10. Bibcode:1991Sci...254.1007S. doi:10.1126/science.1948070. PMID 1948070.
- Pruzanski W, Bogoch E, Stefanski E, Wloch M, Vadas P (1991). "Enzymatic activity and distribution of phospholipase A2 in human cartilage". Life Sci. 48 (25): 2457–62. doi:10.1016/0024-3205(91)90381-K. PMID 2046470.
- Wery JP, Schevitz RW, Clawson DK, Bobbitt JL, Dow ER, Gamboa G, Goodson Jr T, Hermann RB, Kramer RM (1991). "Structure of recombinant human rheumatoid arthritic synovial fluid phospholipase A2 at 2.2 A resolution". Nature. 352 (6330): 79–82. Bibcode:1991Natur.352...79W. doi:10.1038/352079a0. PMID 2062381. S2CID 4238043.
- Seilhamer JJ, Randall TL, Johnson LK, Heinzmann C, Klisak I, Sparkes RS, Lusis AJ (1989). "Novel gene exon homologous to pancreatic phospholipase A2: sequence and chromosomal mapping of both human genes". J. Cell. Biochem. 39 (3): 327–37. doi:10.1002/jcb.240390312. PMID 2708461. S2CID 40949411.
- Kanda A, Ono T, Yoshida N, Tojo H, Okamoto M (1989). "The primary structure of a membrane-associated phospholipase A2 from human spleen". Biochem. Biophys. Res. Commun. 163 (1): 42–8. doi:10.1016/0006-291X(89)92096-2. PMID 2775276.
- Seilhamer JJ, Pruzanski W, Vadas P, Plant S, Miller JA, Kloss J, Johnson LK (1989). "Cloning and recombinant expression of phospholipase A2 present in rheumatoid arthritic synovial fluid". J. Biol. Chem. 264 (10): 5335–8. doi:10.1016/S0021-9258(18)83549-9. PMID 2925608.
- Kramer RM, Hession C, Johansen B, Hayes G, McGray P, Chow EP, Tizard R, Pepinsky RB (1989). "Structure and properties of a human non-pancreatic phospholipase A2". J. Biol. Chem. 264 (10): 5768–75. doi:10.1016/S0021-9258(18)83616-X. PMID 2925633.
- Lai CY, Wada K (1989). "Phospholipase A2 from human synovial fluid: purification and structural homology to the placental enzyme". Biochem. Biophys. Res. Commun. 157 (2): 488–93. doi:10.1016/S0006-291X(88)80275-4. PMID 3202859.
- Hara S, Kudo I, Matsuta K, Miyamoto T, Inoue K (1989). "Amino acid composition and NH2-terminal amino acid sequence of human phospholipase A2 purified from rheumatoid synovial fluid". J. Biochem. 104 (3): 326–8. doi:10.1093/oxfordjournals.jbchem.a122467. PMID 3240982.
- Schevitz RW, Bach NJ, Carlson DG, Chirgadze N, Clawson D, Dillard R, Draheim S, Hartley L, Jones N (1995). "Structure-based design of the first potent and selective inhibitor of human non-pancreatic secretory phospholipase A2". Nat. Struct. Biol. 2 (6): 458–65. doi:10.1038/nsb0695-458. PMC 7097651. PMID 7664108.
- Ancian P, Lambeau G, Mattéi MG, Lazdunski M (1995). "The human 180-kDa receptor for secretory phospholipases A2. Molecular cloning, identification of a secreted soluble form, expression, and chromosomal localization". J. Biol. Chem. 270 (15): 8963–70. doi:10.1074/jbc.270.15.8963. PMID 7721806.
- MacPhee M, Chepenik KP, Liddell RA, Nelson KK, Siracusa LD, Buchberg AM (1995). "The secretory phospholipase A2 gene is a candidate for the Mom1 locus, a major modifier of ApcMin-induced intestinal neoplasia". Cell. 81 (6): 957–66. doi:10.1016/0092-8674(95)90015-2. PMID 7781071. S2CID 16493676.
- Minami T, Tojo H, Shinomura Y, Matsuzawa Y, Okamoto M (1993). "Purification and characterization of a phospholipase A2 from human ileal mucosa". Biochim. Biophys. Acta. 1170 (2): 125–30. doi:10.1016/0005-2760(93)90061-d. PMID 8399335.
- Sartipy P, Johansen B, Camejo G, Rosengren B, Bondjers G, Hurt-Camejo E (1996). "Binding of human phospholipase A2 type II to proteoglycans. Differential effect of glycosaminoglycans on enzyme activity". J. Biol. Chem. 271 (42): 26307–14. doi:10.1074/jbc.271.42.26307. PMID 8824283.
This article incorporates text from the United States National Library of Medicine, which is in the public domain.