Chemotherapy-induced acral erythema
| Chemotherapy-induced acral erythema | |
|---|---|
| Other names | Palmar-plantar erythrodysesthesia, palmoplantar erythrodysesthesia, hand-foot syndrome |
 | |
| Pictures of hands on capecitabine | |
| Specialty | Dermatology |
Chemotherapy-induced acral erythema, also known as palmar-plantar erythrodysesthesia or hand-foot syndrome is reddening, swelling, numbness and desquamation (skin sloughing or peeling) on palms of the hands and soles of the feet (and, occasionally, on the knees, elbows, and elsewhere) that can occur after chemotherapy in patients with cancer. Hand-foot syndrome is also rarely seen in sickle-cell disease. These skin changes usually are well demarcated. Acral erythema typically disappears within a few weeks after discontinuation of the offending drug.[1][2]
Signs and symptoms[edit]

The symptoms can occur anywhere between days to months after administration of the offending medication, depending on the dose and speed of administration.[3][4] The patient first experiences tingling and/or numbness of the palms and soles. This is followed 2-4 days later by bright redness, which is symmetrical and sharply defined.[5]
In severe cases this may be followed by burning pain and swelling, blistering and ulceration, peeling of the skin.[6] Healing occurs without scarring unless there has been skin ulceration or necrosis (skin loss/death). With each subsequent cycle of chemotherapy, the reaction will appear more quickly, be more severe and will take longer to heal.[5]
Causes[edit]
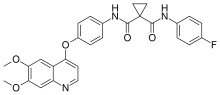
Acral erythema is a common adverse reaction to cytotoxic chemotherapy drugs, particularly cabozantinib, cytarabine, doxorubicin, and fluorouracil and its prodrug capecitabine.[3]
Targeted cancer therapies, especially the tyrosine kinase inhibitors sorafenib and sunitinib, have also been associated with a high incidence of acral erythema. However, acral erythema due to tyrosine kinase inhibitors seems to differ somewhat from acral erythema due to classic chemotherapy drugs.[7]
Pathogenesis[edit]
The cause of Palmar-plantar erythrodysesthesia (PPE) is unknown. Existing hypotheses are based on the fact that only the hands and feet are involved and posit the role of temperature differences, vascular anatomy, differences in the types of cells (rapidly dividing epidermal cells and eccrine glands).[citation needed]
In the case of PPE caused by PLD, the following mechanism has been demonstrated: sweat deposits and spreads the drug on the skin surface; then the drug penetrates into the stratum corneum like an external agent; palms and soles have high density of sweat glands, and their stratum corneum is approximately 10 times thicker than the rest of the body, and becomes an efficient long-term reservoir for the penetrating PLD, which was deposited on the skin before.[8]
Diagnosis[edit]
Painful red swelling of the hands and feet in a patient receiving chemotherapy is usually enough to make the diagnosis. The problem can also arise in patients after bone marrow transplants, as the clinical and histologic features of PPE can be similar to cutaneous manifestations of acute (first three weeks) graft-versus-host disease. It is important to differentiate PPE, which is benign, from the more dangerous graft-versus-host disease. As time progresses, patients with graft-versus-host disease progress to have other body parts affected, while PPE is limited to hands and feet. Serial biopsies every 3 to 5 days can also be helpful in differentiating the two disorders.[9]
Prevention[edit]
The cooling of hands and feet during chemotherapy may help prevent PPE.[3][10] Support for this and a variety of other approaches to treat or prevent acral erythema comes from small clinical studies, although none has been proven in a randomised controlled clinical trial of sufficient size.[11]
Modifying some daily activities to reduce friction and heat exposure to your hands and feet for a period of time following treatment (approximately one week after IV medication, much as possible during the time you are taking oral medication such as capecitabine)."Hand-Foot Syndrome". Chemocare. Archived from the original on 2021-01-26. Retrieved 2020-05-27.</ref>[12]
Treatment[edit]
The main treatment for acral erythema is discontinuation of the offending drug, and symptomatic treatment to provide analgesia, lessen edema, and prevent superinfection. However, the treatment for the underlying cancer of the patient must not be neglected. Often, the discontinued drug can be substituted with another cancer drug or cancer treatment.[13][14]
Symptomatic treatment can include wound care, elevation, and pain medication. Various emollients (creams) are recommended to keep skin moist. Corticosteroids and pyridoxine have also been used to relieve symptoms.[15] Other studies do not support the conclusion. A number of additional remedies are listed in recent medical literature.[16] Among them henna and 10% uridine ointment which went through clinical trial.[17]
Prognosis[edit]
Hand-foot invariably recurs with the resumption of chemotherapy. Long-term chemotherapy may also result in reversible palmoplantar keratoderma. Symptoms resolve 1–2 weeks after cessation of chemotherapy.[6] The range is 1-5 weeks, so it has recovered by the time the next cycle is due. Healing occurs without scarring unless there has been skin ulceration or necrosis. With each subsequent cycle of chemotherapy, the reaction will appear more quickly, be more severe and will take longer to heal.[5]
History[edit]
Hand-foot syndrome was first reported in association with chemotherapy by Zuehlke in 1974.[18] Synonyms for acral erythema (AE) include: hand-foot syndrome, palmar-plantar erythrodysesthesia, peculiar AE, chemotherapy-induced AE, toxic erythema of the palms and soles, palmar-plantar erythema, and Burgdorf's reaction. Common abbreviations are HFS and PPE.
References[edit]
- ^ James, William; Berger, Timothy; Elston, Dirk (2005). Andrews' Diseases of the Skin: Clinical Dermatology. (10th ed.). Saunders. ISBN 0-7216-2921-0.: 132
- ^ Rosenbeck L, Kiel PJ (2011). "Images in clinical medicine. Palmar-plantar rash with cytarabine therapy". N. Engl. J. Med. 364 (3): e5. doi:10.1056/NEJMicm1006530. PMID 21247311.
- ^ a b c Baack BR, Burgdorf WH (1991). "Chemotherapy-induced acral erythema". J. Am. Acad. Dermatol. 24 (3): 457–61. doi:10.1016/0190-9622(91)70073-b. PMID 2061446. S2CID 9626443.
- ^ Demirçay Z, Gürbüz O, Alpdoğan TB, Yücelten D, Alpdoğan O, Kurtkaya O, Bayik M (1997). "Chemotherapy-induced acral erythema in leukemic patients: a report of 15 cases". International Journal of Dermatology. 36 (8): 593–8. doi:10.1046/j.1365-4362.1997.00040.x. PMID 9329890. S2CID 25775290.
- ^ a b c "Hand-foot syndrome". DermNet NZ.
- ^ a b Apisarnthanarax N, Duvic MM (2003). "Acral Erythema". Holland-Frei Cancer Medicine (6th ed.). American Association for Cancer Research – via NCBI.
- ^ Lacouture ME, Reilly LM, Gerami P, Guitart J (2008). "Hand foot skin reaction in cancer patients treated with the multikinase inhibitors sorafenib and sunitinib". J. Ann. Oncol. 19 (11): 1955–61. doi:10.1093/annonc/mdn389. PMID 18550575.
- ^ Lademann, Juergen; Martschick, Anja; Kluschke, Franziska; Richter, Heike; Fluhr, Joachim W.; Patzelt, Alexa; Jung, Sora; Chekerov, Radoslav; Darvin, Maxim E.; Haas, Norbert; Sterry, Wolfram; Zastrow, Leonhard; Sehouli, Jalid (2014). "Efficient prevention strategy against the development of a palmar-plantar erythrodysesthesia during chemotherapy". Skin Pharmacology and Physiology. 27 (2): 66–70. doi:10.1159/000351801. ISSN 1660-5535. PMID 23969763. S2CID 33022244.
- ^ Crider MK, Jansen J, Norins AL, McHale MS (1986). "Chemotherapy-induced acral erythema in patients receiving bone marrow transplantation". Arch Dermatol. 122 (9): 1023–7. doi:10.1001/archderm.122.9.1023. PMID 3527075.
- ^ Zimmerman GC, Keeling JH, Burris HA, Cook G, Irvin R, Kuhn J, McCollough ML, Von Hoff DD (1995). "Acute cutaneous reactions to docetaxel, a new chemotherapeutic agent". Arch Dermatol. 131 (2): 202–6. doi:10.1001/archderm.1995.01690140086015. PMID 7857119.
- ^ von Moos, Roger; Thuerlimann, Beat J. K.; Aapro, Matti; Rayson, Daniel; Harrold, Karen; Sehouli, Jalid; Scotte, Florian; Lorusso, Domenica; Dummer, Reinhard; Lacouture, Mario E.; Lademann, Jürgen; Hauschild, Axel (April 2008). "Pegylated liposomal doxorubicin-associated hand-foot syndrome: recommendations of an international panel of experts". European Journal of Cancer. 44 (6): 781–790. doi:10.1016/j.ejca.2008.01.028. ISSN 0959-8049. PMID 18331788.
- ^ "Hand-Foot Syndrome: A Side Effect of Treatment". 18 December 2020 – via breastcancer.org.
- ^ Cutaneous complications of conventional chemotherapy agents. Payne AS, Savarese DMF. In: UpToDate [Textbook of Medicine]. Massachusetts Medical Society, and Wolters Kluwer publishers. 2010.
- ^ Gressett SM, Stanford BL, Hardwicke F (Sep 2006). "Management of hand-foot syndrome induced by capecitabine". Journal of Oncology Pharmacy Practice. 12 (3). Sage: 131–41. doi:10.1177/1078155206069242. PMID 17022868. S2CID 22230787.
- ^ Vukelja SJ, Baker WJ, Burris HA, Keeling JH, Von Hoff D (1993). "Pyridoxine therapy for palmar-plantar erythrodysesthesia associated with taxotere". J. Natl. Cancer Inst. 85 (17): 1432–3. doi:10.1093/jnci/85.17.1432. PMID 8102408.
- ^
- Juergen Barth (March 2004). "Letter to the editor - 5-FU induced palmar-plantar erythrodyesthesia – a hospital pharmacy developed "antidot"". Journal of Oncology Pharmacy Practice. Vol. 10, no. 57.
- Yucel, Idris; Guzin, Gonullu (2008). "Topical henna for capecitabine induced hand-foot syndrome". Investigational New Drugs. 26 (2): 189–192. doi:10.1007/s10637-007-9082-3. ISSN 0167-6997. PMID 17885735. S2CID 7737832.
- Hartinger, J.; Veselý, P.; Matoušková, E.; Argalacsová, S.; Petruželka, L.; Netíková, I. (2012). "Local treatment of hand-foot syndrome with uridine/thymidine: in vitro appraisal on a human keratinocyte cell line HaCaT". The Scientific World Journal. 2012: 421325. doi:10.1100/2012/421325. ISSN 1537-744X. PMC 3417181. PMID 22919318.
- Hand-Foot Syndrome in cancer patients: concepts, assessment and management of symptoms. 2013.[full citation needed]
- Ilyas, Saher; Wasif, Komal; Saif, Muhammad Wasif (2014). "Topical henna ameliorated capecitabine-induced hand-foot syndrome". Cutaneous and Ocular Toxicology. 33 (3): 253–255. doi:10.3109/15569527.2013.832280. ISSN 1556-9535. PMID 24021017. S2CID 36514303.
- ^ Irena Netikova; Agnes Petska; Juergen Barth (2009). "Recent clinical studies with uridine cream" (PDF). EJOP. Oncology Pharmacy Practice. 3 (2): 22–23. Archived from the original (PDF) on 2016-03-04. Retrieved 2015-08-26.
- ^ Zuehlke RL (1974). "Erythematous eruption of the palms and soles associated with mitotane therapy". Dermatologica. 148 (2): 90–2. doi:10.1159/000251603. PMID 4276191.
Further reading[edit]
- Farr, Katherina Podlekareva; Safwat, Akmal (2011). "Palmar-Plantar Erythrodysesthesia Associated with Chemotherapy and Its Treatment". Case Reports in Oncology. 4 (1): 229–235. doi:10.1159/000327767. PMC 3085037. PMID 21537373.
- Hand-Foot Syndrome or Palmar-Plantar Erythrodysesthesia (1 & 2)
