Talk:Greenhouse effect
| This is the talk page for discussing improvements to the Greenhouse effect article. This is not a forum for general discussion of the article's subject. |
Article policies
|
| Find sources: Google (books · news · scholar · free images · WP refs) · FENS · JSTOR · TWL |
| Archives: Index, 1, 2, 3, 4, 5, 6, 7, 8Auto-archiving period: 30 days |
| This article is written in American English, which has its own spelling conventions (color, defense, traveled) and some terms that are used in it may be different or absent from other varieties of English. According to the relevant style guide, this should not be changed without broad consensus. |
| This It is of interest to multiple WikiProjects. | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| The contentious topics procedure applies to this page. This page is related to climate change, which has been designated as a contentious topic. Editors who repeatedly or seriously fail to adhere to the purpose of Wikipedia, any expected standards of behaviour, or any normal editorial process may be blocked or restricted by an administrator. Editors are advised to familiarise themselves with the contentious topics procedures before editing this page. |
| This page is not a forum for general discussion about Greenhouse effect. Any such comments may be removed or refactored. Please limit discussion to improvement of this article. You may wish to ask factual questions about Greenhouse effect at the Reference desk. |
Simplified caption for first image?
[edit]I don't see why we need to effectively repeat the text inside the first image as the caption for that image. That's particularly true as the first paragraph of the article is also a synopsis of the greenhouse effect. Here is how things currently appear:

What I would like to do is change the caption to something brief and informative, as shown here:

Thoughts, particularly RCraig09? Efbrazil (talk) 16:32, 8 May 2024 (UTC)
- Option B is not informative at all; it's essentially a criticism of the chart itself. Option A is noticeably more concise than the lead's text. —RCraig09 (talk) 16:40, 8 May 2024 (UTC)
- The main point is that it is redundant with the text in the image itself. The image text says this:
- Some sunlight that hits Earth is reflected back into space, while the rest becomes heat. Greenhouse gases absorb and redirect heat radiated by Earth, insulating it from heat loss to space.
- This caption is really redundant with that:
- the greenhouse effect occurs when greenhouse gases first allow sunlight to pass through the atmosphere and heat the planet, but then absorb and redirect some of the outgoing longwave radiation (heat) that the planet emits.
- The caption I offered wasn't meant as a critique, just a pointer to other image people might find useful for showing how the greenhouse effect works. I'd also be fine just saying "Simplified diagram of how the greenhouse effect works", or other suggestions that aren't redundant. Thoughts? Efbrazil (talk) 16:48, 8 May 2024 (UTC)
- Option A is reader-centric but Option B is editor-centric. Although Option A tells the story sequentially and I favor it over Option B which points lay readers to the bowels of Commons, ...we could have NO caption for that particular graphic. That would work for me. —RCraig09 (talk) 19:30, 8 May 2024 (UTC)
- Cool, no caption works for me! I'll make the edit... Efbrazil (talk) 20:48, 8 May 2024 (UTC)
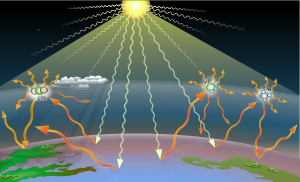
Greenhouse gases trap some of the heat that results when sunlight heats the Earth's surface. Three important greenhouse gases are shown symbolically in this image: carbon dioxide, water vapor, and methane. - Hmmm, I don't know but I think I'd prefer an image caption. I quite liked Option B, actually. The wikilink for "other options" could go somewhere else as well (but I don't mind if it goes to Commons). But I also wonder if that chosen image is really suitable. Compare with the lead image at greenhouse gas (on the right) which I think is a lot better. Wondering if we should maybe use the same image here (even though I usually argue that the lead image should be unique for each Wikipedia article). Just putting out some food for thought. EMsmile (talk) 09:14, 9 May 2024 (UTC)

- I was also looking for a good symbolic image for the German Wikipedia to visualize the greenhouse effect. I did not find one, so I created a new image. I had the idea that maybe the difference between the presence of and the absence of greenhouse gases could be a good visual way to explain the effect. In the German article I have added a caption with 3 sentences to describe each of the 3 sections. But the image is also self-explanatory and could work without captions. Physikinger (talk) 22:24, 11 June 2024 (UTC)
- I am sorry, but this image is confusing for the casual viewer-- so many arrows! Only one sun? Two planets or three? Also, it hurts my eyes. The current image has a lot of text. Text is for article content, not so much images. I created the image mentioned by EMsmile. I did my best to make it visually interesting, informative, and easily grasped with a minimum of text. I am also biased against renditions of the sun as a golden marble, and against depictions of Earth as having sapphire blue oceans and emerald green continents everywhere. Lastly, the red arrows are "bouncing" off of nothing in that image. Why not show them bouncing off of molecules in the air, the way they actually are? A loose necktie (talk) 13:16, 29 June 2024 (UTC)
- First of all, I really like your picture. It's a very nice and inspiring SVG artwork, and I'm using it as well in the German greenhouse effect Wikipedia article. But there are some points why I decided not to use it as the thumbnail image of the article:
- The molecules and text are so small that I found it less suitable as a symbolic thumbnail, which appears quite small in Google searches and when hovering over wiki links.
- There was also a discussion about a video https://www.youtube.com/watch?v=oqu5DjzOBF8 about a common misconception that heat radiation does not bounce between the sky and the ground, but rather most of the time between molecules. It's more a diffusive energy transport.
- In many drawings like yours, the atmosphere is shown as a layer like the roof of a greenhouse, which gives a false impression.
- With my image I tried to improve these points. I don't really understand your criticism. It's primarily a symbolic image, trying to convey an abstract idea of what the essence of the effect is. With the arrows I am trying to express that the mean free paths are very short in the presence of the greenhouse gases and that the surface cooling becomes less efficient.
- But don't get me wrong. I don't want to convince anyone to use my image. I just wanted to give a hint that there is another drawing. Physikinger (talk) 18:02, 11 July 2024 (UTC)
- First of all, I really like your picture. It's a very nice and inspiring SVG artwork, and I'm using it as well in the German greenhouse effect Wikipedia article. But there are some points why I decided not to use it as the thumbnail image of the article:
- I am sorry, but this image is confusing for the casual viewer-- so many arrows! Only one sun? Two planets or three? Also, it hurts my eyes. The current image has a lot of text. Text is for article content, not so much images. I created the image mentioned by EMsmile. I did my best to make it visually interesting, informative, and easily grasped with a minimum of text. I am also biased against renditions of the sun as a golden marble, and against depictions of Earth as having sapphire blue oceans and emerald green continents everywhere. Lastly, the red arrows are "bouncing" off of nothing in that image. Why not show them bouncing off of molecules in the air, the way they actually are? A loose necktie (talk) 13:16, 29 June 2024 (UTC)
- Cool, no caption works for me! I'll make the edit... Efbrazil (talk) 20:48, 8 May 2024 (UTC)
- Option A is reader-centric but Option B is editor-centric. Although Option A tells the story sequentially and I favor it over Option B which points lay readers to the bowels of Commons, ...we could have NO caption for that particular graphic. That would work for me. —RCraig09 (talk) 19:30, 8 May 2024 (UTC)
Unsourced opinion
| ||
|---|---|---|
Semi-protected edit request on 23 June 2024[edit]
The scientific errors are numerous, based In part on outdated information from the early 19th Century, and a lack of knowledge of more recent theoretical and experimental work up to the modern experiments done at CERN Lab's CLOUD Project in Geneva Switzerland. Past errors. In 1829 Fourier published his Greenhouse Theory based upon his incorrect understanding of how a greenhouse works. As we know a greenhouse is a box in which air is trapped. As it absorbs the sun's heat (in the form of infrared radiation (IR)), the warmed air is prevented from rising up into the sky (as a hot air balloon does), keeping the interior of the greenhouse warmer than the outside air. Fourie, however, believed that glass blocked out infrared radiation. This is an idiocy as Herschel discovered IR by using a glass prism to separate the IR from the visible light, allowing him to separately measure it and prove its existence. If glass blocked IR then Herschel would never have measured it. Fourie never realized that Herschel's use of a glass prison to separate out IR proved that glass did not block IR. Thus, he asked himself Why does a greenhouse get warm if the Sun's IR cannot pass through the glass? He falsely held that short-wave radiation (visible light) must be changing into IR when it strikes the interior of the greenhouse. As he believed that IR cannot pass through the glass, it becomes trapped inside the greenhouse and thus warms the greenhouse interior. This belief in visible light changing into IR continued until modern times until NASA satellite measurements showed that the IR radiation emanating off the Earth's surface was equal in wavelength and amplitude to that measured by NASA ground stations as hitting the Earth's surface. There is no more heat leaving the earth than is arriving. If Visible light was changing into IR then, given that on average a visible light photon has 1,000 times more energy than the average IR photon, there must be at least 1,000 times more heat leaving the earth than arriving at its surface, and this is not happening. In 1856, Eunice Foote tested the interaction of rarified air, hydrogen, nitrogen, oxygen, carbon dioxide, and moist air with the Sun's heat. Identical glass bottles were filled with a mercury thermometer, and with as pure a gas as possible for her to make. The bottles were placed in the direct light of the sun and the temperatures of the gases were measured by her. Her measurements showed little to no effect on the temperature of the non-thermally reactive gases, the hydrogen, oxygen, and nitrogen, but she noted a significant rise in the moist air and a greater rise in the carbon dioxide's temperature. This experiment showed that the Sun's IR heat passed unaffected through the hydrogen, oxygen, and nitrogen gas, but that the water vapor and Carbon Dioxide prevented some of the Sun's IR heat from passing through them. Some of the Sun's IR heat was trapped inside the bottle, reducing the amount of heat that passed out of the bottle towards the table below reducing its temperature. Forty years later in his paper "On the Influence of Carbonic Acid in the Air upon the Temperature of the Ground" [Philosophical Magazine and Journal of Science, Series 5, Volume 41, April 1896, pages 237-276]. Svante Arrhenius presents his calculations of the effect of CO2 and water vapor on IR radiation. He writes on page 240: “The intensity of radiation for any group of rays should always diminish with increasing quantity(emphasis mine), of aqueous vapour or carbonic acid traversed.” While on page 244 of the work he gives his exacting calculations writing: “If a ray of heat, corresponding to the angle of deviation 39 degrees 45 minutes passes through the unit of carbonic acid, it decreases in intensity in the proportion 1: 0.934 (log= -0.0296), the corresponding value for the unit of water-vapour is 1: 0.775 (log= -0.11105).” Over the next 67 years, Gilbert Plass, Kondrat'ev, Kaplan, Sasamori, Yamamoto, and Fritz Moller, experimentally tested Arrhenius calculations and found them to be correct. In Moller's work entitled; "On the Influence of Changes in the Carbon Dioxide Concentration in Air on the Radiation Balance of the Earth’s Surface and on the Climate” [Journal of Geophysical Research, Volume 68, Number 31, July 1, 1963]. In Table One, Moller charts his experimental findings and those of Plass, Kondrat'ev, Kaplan, Sasamori, and Yamamoto. Plass's experiments showed that when he reduced the CO2 concentration in his test chamber from 300 ppm to 150 ppm, the heat emanating from the earth below the test chamber rose 12.5 mcal/cm2. When it was increased from 300 ppm to 600 ppm the heat radiating off the earth below the test chamber fell -11.9 mcal/cm2. The same results were shown by Kaplan, Kondrat'ev, Yamamoto, and Sasamori. Unlike Foote, however, these tests showed the effect of Water Vapor was far higher than CO2's effect on IR. This is in agreement with Arrhenius's calculation of a larger effect than CO2 on IR. I cannot download the table referenced, so you will have to google the paper for yourself. Since 2012 CERN Labs in Geneva Switzerland has been using their Proton Synchrotron to do atmospheric tests to determine the effect of Cosmic Radiation on Cloud formation, leading to changes in the downward flow of Sun IR heat. The IPCC in its first report on CO2's effect on Earth's surface temperature, stated that 1.4 watts per meter squared of heat since the end of the Little Ice Age and the beginning of the Industrial Revolution could not be explained by those factors then proven to affect Earth's temperature and so must be caused by the increases in the CO2 concentrations since them. This claim ignored all scientific research in atmospheric done since the publication of Arrhenius' work. CERN reports that its experiments have proven that 1.2 watts per meter squared of the warming since the end of the Little Ice Age and the beginning of the Industrial Revolution was caused by changes in cloud cover due to changes in the Cosmic Radiation entering the Earth's upper atmosphere, not by CO2. CERN has firmly established that Comsic Radiation is the leading factor in Earth's surface Temperature. CERN's experiments show that increased levels of cosmic radiation in the upper atmosphere lead to significant increases in cloud formation and an increase in their whiteness, and fluffiness blocking out more Sun heat and cooling the Earth. The Sun's magnetosphere is the primary protector of the Earth from cosmic radiation. When the Sun enters a Solar Minimum, (like the Little Ice Age), its magnetosphere weakens and allows more cosmic radiation into the upper atmosphere where it seeds clouds. Conversely, when the Sun exits a Solar Minimum, its magnetosphere increases in strength lessening the cosmic radiation able to enter the upper atmosphere. This drop in cosmic radiation leads to fewer clouds that are less white and fluffy and so more Sun heat reaches the Earth. The entire idea of man-made climate change is based upon the false belief that there is more IR heat leaving the Earth than is arriving, due to the false belief that visible light is changing into IR heat. In addition, early Natural Philosophers, like Arrhenius, believed that the IR portion of the Sun was minuscule compared to the visible light's portion. Thus, while they understood that the CO2 and water vapor must also be blocking out incoming Sun heat from reaching the Earth cooling it, they felt it was a small effect that could be disregarded when measured against the much greater heat being kept in. Today we know that IR constitutes 52% of the sun's spectrum, and the atmosphere blocks out 174F degrees of Solar heat. Today, proponents of man-made CO2 warming constantly cite Schwarzchild's Equation as support for their position. Scharzchild's Equation does prove that increased levels of CO2 will make the atmosphere more insulating to outgoing heat, but it also keeps out more Sun heat cooling the Earth. The Stephan-Boltzmann Law tells us that if CO2 levels rise enough so as to make the atmosphere 1% more insulating, it will both keep in 1% more heat and keep out 1% more heat cooling the Earth by 1%. A 1% drop in Earth's surface temperature due to an increase in the CO2 level in the atmosphere will cause a 4% drop in radiated IR heat off the Earth's surface. Thus the Greenhouse effect will fall by 3%! The 1% increase in the insulating value of the atmosphere will cause a -4% drop in radiated heat off the Earth's surface, creating a 3% net drop in the Greenhouse Effect cooling the Earth. We cannot increase the Greenhouse Effect by blocking out more Sun IR heat with higher CO2 concentrations. That would lessen the Earth's surface Tempeartuer as Arrhenius calculated and Plas and the others proved experimentally. NASA has also proven this heat blocking is occurring further supporting Arrhenius. As CO2 levels in the troposphere have increased in the decades past, NASA satellites have measured an increase in the temperature of the Troposphere. The Troposphere is absorbing more Sun IR heat and preventing it from reaching the Earth below. Arrhenius was correct in saying that more CO2 or water vapor will keep out more Sun heat. All those claiming man-made greenhouse warming do so by refusing to face experimental results showing CO2 keeps out Sun heat and cools the earth. It is 250 Degrees above the atmosphere, while only 76.4 degrees average temperature below it. This is undeniable, the "greenhouse gases" cool the Earth by 174 degrees! This cooling would be impossible if man-made CO2 warming was real. The Earth's temperature would be greater than the total amount of heat the Earth receives from the Sun if the CO2 climate change theory was factual. It is time to end the political use of Wikipedia and provide the public with the truth, which is why this site exists.Bud Williams (talk) 04:13, 23 June 2024 (UTC)[1] Bud Williams (talk) 04:13, 23 June 2024 (UTC)
References
|
The section "Effects of pressure"
[edit]I don't really understand the section "Effects of pressure": do we really need it? Or is it in the right position? It's under "bodies other than Earth" but then it does talk about the situation on Earth. I wonder if it's perhaps too detailed for this high-level article? EMsmile (talk) 14:35, 7 August 2024 (UTC)
Circle
[edit]The greenhouse effect occurs when greenhouse gases in a planet's atmosphere This is a circular explanation. You should never explain a term with itself. Better is 'ir active gases'. 2003:E5:272D:4A00:6511:D243:C8D4:3A09 (talk) 19:12, 13 December 2024 (UTC)
- This is an encyclopedia, not a dictionary. Here, the term greenhouse gas is wikilinked to its own article. The term "IR active gases" is non-standard. —RCraig09 (talk) 19:45, 13 December 2024 (UTC)
Eunice Newton Foote
[edit]What has she measured? Differences in thermal conductivity? All gases showed significant differences in their physical parameters (density, thermal conductivity). Changed convection? Anything is possible. But not the greenhouse effect. That cannot be measured in a laboratory experiment. 2003:E5:272D:4A00:6511:D243:C8D4:3A09 (talk) 19:24, 13 December 2024 (UTC)
- We're talking about radiant heat (Thermal radiation), not physical-contact thermal conductivity or convection. The GHE is measurable in the laboratory. —RCraig09 (talk) 19:43, 13 December 2024 (UTC)
- Wikipedia articles that use American English
- C-Class level-4 vital articles
- Wikipedia level-4 vital articles in Physical sciences
- C-Class vital articles in Physical sciences
- C-Class Environment articles
- Top-importance Environment articles
- C-Class Climate change articles
- Top-importance Climate change articles
- WikiProject Climate change articles
- C-Class Weather articles
- High-importance Weather articles
- C-Class Climate articles
- High-importance Climate articles
- WikiProject Weather articles





