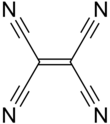
Tetracyanoethylene
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Ethenetetracarbonitrile | |||
| Other names
TCNE
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.010.527 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C2(CN)4 | |||
| Molar mass | 128.094 g·mol−1 | ||
| Density | 1.35 g/cm3 | ||
| Melting point | 199 °C (390 °F; 472 K) | ||
| Boiling point | 130 to 140 °C (266 to 284 °F; 403 to 413 K) 0.1 mmHg (sublimes)[1] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Tetracyanoethylene (TCNE) is organic compound with the formula C2(CN)4. It is a colorless solid, although samples are often off-white. It is an important member of the cyanocarbons.
Synthesis and reactions[edit]
TCNE is prepared by brominating malononitrile in the presence of potassium bromide to give the KBr-complex, and dehalogenating with copper.[1]
Oxidation of TCNE with hydrogen peroxide gives the corresponding epoxide, which has unusual properties.[2]
In the presence of base, TCNE reacts with malononitrile to give salts of pentacyanopropenide:[3]
- C2(CN)4 + CH2(CN)2 → [(NC)2C=C(CN)−C(CN)2]− + CN− + 2 H+
Redox chemistry[edit]
TCNE is an electron acceptor. Cyano groups have low energy π* orbitals, and the presence of four such groups, with their π systems (conjugated) to the central C=C double bond, gives rise to an electrophilic alkene. TCNE is reduced at −0.27 V vs ferrocene/ferrocenium:[4]
- C2(CN)4 + e− → [C2(CN)4]−
Because of its ability to accept an electron, TCNE has been used to prepare numerous charge-transfer salts.[5]
The central C=C distance in TCNE is 135 pm.[6] Upon reduction, this bond elongates to 141–145 pm, depending on the counterion.[7]
Safety[edit]
TCNE hydrolyzes in moist air to give hydrogen cyanide and should be handled accordingly.[1]
References[edit]
- ^ a b c Carboni, R. A. (1959). "Tetracyanoethylene". Organic Syntheses. 39: 64. doi:10.15227/orgsyn.039.0064.
- ^ Linn, W. J. (1969). "Tetracyanoethylene Oxide". Organic Syntheses. 49: 103. doi:10.15227/orgsyn.049.0103.
- ^ Middleton, W. J.; Wiley, D. W. (1961). "Tetramethylammonium 1,1,2,3,3-Pentacyanopropenide". Org. Synth. 41: 99. doi:10.15227/orgsyn.041.0099.
- ^ Connelly, N. G.; Geiger, W. E. (1996). "Chemical Redox Agents for Organometallic Chemistry". Chemical Reviews. 96 (2): 877–910. doi:10.1021/cr940053x. PMID 11848774.
- ^ Chittipeddi, Sailesh; Cromack, K. R.; Miller, Joel S.; Epstein, A. J. (1987-06-22). "Ferromagnetism in Molecular Decamethylferrocenium Tetracyanoethenide (DMeFc TCNE)". Physical Review Letters. 58 (25): 2695–2698. Bibcode:1987PhRvL..58.2695C. doi:10.1103/physrevlett.58.2695. ISSN 0031-9007. PMID 10034821.
- ^ Becker, P.; Coppens, P.; Ross, F. K. (1973). "Valence electron distribution in cubic tetracyanoethylene by the combined use of x-ray and neutron diffraction". Journal of the American Chemical Society. 95 (23): 7604–7609. doi:10.1021/ja00804a010.
- ^ Bock, H.; Ruppert, K. (1992). "Structures of charge-perturbed or sterically overcrowded molecules. 16. The cesium tetracyanoethylenide radical salt". Inorganic Chemistry. 31 (24): 5094–5099. doi:10.1021/ic00050a032.