User:Elcap/Pseudocapacitance
Basics and construction, draft for supercapacitor
[edit]Basics
[edit]Basic design
[edit]
Electrochemical capacitors (supercapacitors) basically consists out of two electrodes separated by an ion permeable membrane (separator), and an electrolyte connecting electrically the both electrodes. By applying a voltage to the capacitor an electric double layer at both electrodes is formed, which has a positive or negative layer of ions deposited in a mirror image on the opposite electrode.
Capacitance distribution
[edit]The two electrodes form a series circuit of two individual capacitors C1 and C2. The total capacitance Ctotal is given by the formula
Supercapacitors can be constructed with either symmetric or asymmetric electrodes. Symmetry implies that both electrodes have the same capacitance value.
That means, if C1 = C2 than Ctotal = 0.5 • C1. For symmetric capacitors the total capacitance value equals half the value of a single electrode.
For asymmetric capacitors one of the electrodes has higher capacitance value than the other. If C1 >> C2 than Ctotal ≈ C2. Asymmetric electrodes imply that total capacitance can equal that of a single electrode, potentially doubling the total
Storage principles
[edit]Electrocemical capacitors uses the double-layer effect to store electric energy. This double-layer has no conventional solid dielectric which separates the charges. The capacitance values of electrochemical capacitors are determined by two new and different high-capacity storage principles in the electric double-layer on their electrodes:
- Double-layer capacitance, electrostatic storage of the electrical energy achieved by separation of charge in a Helmholtz double layer.[1]
- Pseudocapacitance, ((Electrochemistry|electrochemical)) storage of the electrical energy achieved by faradiac redox reactions with charge-transfer.[2]
The amount of charge stored per unit voltage in an electrochemical capacitor is primarily a function of the electrode size but the amount of capacitance of each storage principle can vary extremely. Double-layer capacitance and pseudocapacitance both contribute to the total capacitance value of an electrochemical capacitorThe amount of capacitance of each storage principle in an electrochemical capacitor can vary extremely. Double-layer capacitance and pseudocapacitance both contribute inseparable to the total capacitance value of an electrochemical capacitor..[3] Both capacitances are only separable by measurement techniques.
This new storage principles gives a capacitor with a capacitance value in the "Farad" range.
Electrostatic double-layer capacitance
[edit]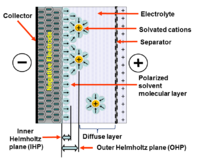
Every electrochemical capacitor has two electrodes, mechanically separated by a separator, which are electrically connected to each other via the electrolyte. The electrolyte is a mixture of positive and negative ions dissolved in a solvent such as water. At each of the two electrodes surfaces originates an area in which the liquid electrolyte contacts the conductive metallic surface of the electrode. This interface forming a common boundary among two different phases of matter, such as an insoluble solid electrode surface and an adjacent liquid electrolyte. In this interface occurs a very special phenomenon of the double layer effect.[4]
Applying a voltage to an electro-chemical capacitor both electrodes in the capacitor generates electrical double-layers. These double layers consist out of two layers of ions. One layer is in the surface lattice structure of the electrode. The other layer, with opposite polarity, emerges from dissolved and solvated ions in the electrolyte. The two layers are separated by a monolayer of solvent molecules, e. g. for water as solvent by water molecules. The monolayer forms the inner Helmholtz plane (IHP). It adheres by physical adsorption on the surface of the electrode and separates the oppositely polarized ions from each other, becoming a molecular dielectric. The forces that cause the adhesion are not chemical bonds but physical forces. Chemical bonds persist within of the adsorbed molecules, but they are polarized.
The amount of charge in the electrode is matched by the magnitude of counter-charges in outer Helmholtz plane (OHP). This double-layer phenomena store electrical charges as in a conventional capacitor. The double-layer charge forms a static electric field in the molecular layer of the solvent molecules in the IHP that corresponds to the strength of the applied voltage.

The double-layer serves approximately like the dielectric layer in a conventional capacitor, but with the thickness of a single molecule. Therefore to calculate the capacitance the standard formula for conventional plate capacitors can be used. This capacitance can be calculated with:[5]
- .
The capacitance C is greatest in capacitors made from materials with a high permittivity ε, large electrode plate surface areas A and reciprocal to the distance d between plates.
If the electrolyte solvent is water then the influence of the high field strength creates permittivity ε of 6 (instead of 80 without an applied electric field). Because activated carbon electrodes have an extremely large surface area in the range of 10 to 40 µF/cm2 and the extremely thin double-layer distance is on the order of a few ångströms (0.3-0.8 nm), the double-layer capacitors have much higher capacitance values than conventional capacitors.[1][2]
The amount of charge stored per unit voltage in an electrochemical capacitor is primarily a function of the electrode size. The electrostatic storage of energy in the double-layers is linear with respect to the stored charge, and correspond to the concentration of the adsorbed ions. But deviating from conventional capacitors, where usually the charge is transferred via electrons, the capacitance of the double-layer capacitors depends from the limited moving speed of ions in the electrolyte and the resistive porous structure of the electrodes. Capacitance values of supercapacitors depends strongly on the measuring time. Charging and discharging electric double-layers in principle is unlimited. No chemical changes take place. Lifetimes of real supercapacitors only are limited by electrolyte evaporation effects.
Electrochemical Pseudocapacitance
[edit]
Applying a voltage at the electrochemical capacitor terminals moves the polarized ions or charged atoms in the electrolyte to the opposite polarized electrode and forms a double-layer, separated by a single layer of solvent molecules. Pseudocapacitance can originate when specifically adsorbed cations out of the electrolyte pervade the double-layer. This pseudocapacitance stores electrical energy by means of reversible faradaic redox reactions on the surface of suitable electrodes in an electrochemical capacitor with a electric double-layer.[6][1][7] Pseudocapacitance is accompanied with an electron charge-transfer between electrolyte and electrode coming from a de-solvated and adsorbed ion whereby only one electron per charge unit is participating. This faradaic charge transfer originates by a very fast sequence of reversible redox, intercalation or electrosorption processes. The adsorbed ion has no chemical reaction with the atoms of the electrode. No chemical bonds arise.[8] Only a charge-transfer take place.
The electrons involved in the faradaic processes are transferred to or from valence electron states (orbitals) of the redox electrode reagent. They enter the negative electrode and flow through the external circuit to the positive electrode where a second double-layer with an equal number of anions has formed. But these anions don’t accept the electrons. They remain on the electrode's surface in the charged state, and the electrons remain in the strongly ionized and "electron hungry" transition-metal ions of the electrode. This kind of pseudocapacitance has a linear function within narrow limits and is determined by the potential-dependent degree of surface coverage of the adsorbed anions. The storage capacity of the pseudocapacitance is limited by the finite quantity of reagent of available surface. A faradaic pseudocapacitance still only occurs together with a static double-layer capacitance. Pseudocapacitance and double-layer capacitance both contribute indivisible to the total capacitance value of the chemical capacitor. The amount of pseudocapacitance depends on the surface area, material and structure of the electrodes.

Since all the pseudocapacitance reactions take place only with de-solvated ions, which are much smaller than solvated ion with their solvating shell, the pseudocapacitance can be much higher than the double-layer capacitance for the same electrode surface. Therefore the pseudocapacitance may exceed the value of double-layer capacitance for the same surface area by factor 100, depending on the nature and the structure of the electrode.[6]
The ability of electrodes to accomplish pseudocapacitance effects by redox reactions, intercalation or electrosorption strongly depends on the chemical affinity of electrode materials to the ions adsorbed on the electrode surface as well as on the structure and dimension of the electrode pores. Materials exhibiting redox behavior for use as electrodes in pseudocapacitors are transition-metal oxides like ruthenium (RuO2), iridium (IrO2), or manganese (MnO2) inserted by doping in the conductive electrode material such as active carbon, as well as conducting polymers such as polyaniline or derivatives of polythiophene covering the electrode material.
Pseudocapacitance may also originate from the structure and especially from the pore size of the electrodes. The tailored sizes of pores in nano-structured carbon electrodes like carbide-derived carbons (CDCs) or carbon nanotubes (CNTs) for electrodes can be referred to as intercalated pores which can be entered by de-solvated ions from the electrolyte solution and originates pseudocapacitance.[12][13][14]
The amount of electric charge stored in a pseudocapacitance is linearly proportional to the applied voltage. The unit of pseudocapacitance is farad.
Potential distribution
[edit]
Conventional capacitors consist out of two electrodes which are separated by a dielectric material. In conventional capacitors such as ceramic capacitors and film capacitors, the electric charge of a loaded capacitor is stored in a static electric field that permeates the dielectric between the electrodes. The electric field originates by the separation of charge carriers and the strength of the electric field correlates with the potential between the two electrodes. It drops over the dielectric. The total energy increases with the amount of stored charge and the potential between the plates. The maximum potential between the plates, the maximal voltage, is limited by the dielectric's breakdown field strength.

This static storage also applies for electrolytic capacitors in which most of the potential decreases over the anode's thin oxide layer. The electrolyte as cathode may be somewhat resistive so that for "wet" electrolytic capacitors, a small amount of the potential decreases over the electrolyte. For electrolytic capacitors with solid conductive polymer electrolyte this voltage drop is negligible.
Conventional capacitors are also called electrostatic capacitors. The potential (voltage) of a charged capacitor correlates linearly with the stored charge.
Different from conventional capacitors electrochemical capacitors (supercapacitors) basically consists out of two electrodes separated by an ion permeable membrane (separator), and electrically connected via an electrolyte. In this double-layer electrodes a mixture of a double-layer and pseudocapacitance is stored. If both electrodes have approximately the same resistance (#Leakage current), the potential of the capacitor decreases symmetrically over both double-layers, whereby a voltage drop across the ESR of the electrolyte is achieved. The maximum potential across the capacitor, the maximal voltage, is limited by the electrolytes decomposition voltage.
Both the electrostatic and electrochemical storage of energy in electrochemical capacitors are linear with respect to the stored charge, just as in conventional capacitors. The voltage between the capacitors terminals is linear with respect to the amount of stored energy. This linear voltage gradient differs from rechargeable electrochemical batteries, in which the voltage between the terminals remains independent of the amount of stored energy, providing a relatively constant voltage.
Supercapacitor types
[edit]
Supercapacitor is the generic term for the family of electrochemical capacitors. They store, as described above, its electric energy with the two different storage principles, the static double-layer capacitance and electrochemical pseudocapacitance. The distribution of the amounts of both capacitances per capacitor depends on the material and structure of the electrodes. Based on this the supercapacitor family are divided into three types: [2] [3]
- Double-layer capacitors – with activated carbon electrodes or derivates with much higher electrostatic double-layer capacitance than electrochemical pseudocapacitance
- Pseudocapacitors – with transition metal oxide or conducting polymer electrodes with a high amount of electrochemical pseudocapacitance
- Hybrid capacitors – capacitors with asymmetric electrodes one of which exhibits mostly electrostatic and the other mostly electrochemical capacitance, such as lithium-ion capacitors
Construction
[edit]Construction details
[edit]- Styles of supercapacitors with activated carbon electrodes
-
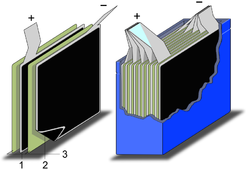
Schematic construction of a wound supercapacitors
1.Terminals, 2.Safety vent, 3.Sealing disc, 4.Aluminum can, 5.Positive pole, 6.Separator, 7.Carbon electrode, 8.Collector, 9.Carbon electrode, 10.Negative pole -
Schematic construction of a supercapacitor with stacked electrodes
1.Positive electrode, 2.Negative electrode, 3.Separator
Supercapacitors are constructed with two metal foils (current collectors), each coated with an electrode material e.g. activated carbon. The collectors serves as the power connection between the electrode material and the external terminals of the capacitor. Specifically of the electrode material is the structure having a very large surface. E. g. in this example activated carbon is electrochemically etched, so that the surface of the material is about a factor 100,000 larger than the smooth surface. The both electrodes are separated by a ion permeable membrane (separator) used as insulator to protect the electrodes against direct contact forcing short circuits. This construction is subsequently rolled or folded into a cylindrical or rectangular shape and can be stacked in an aluminum can or an adaptable rectangular housing. Then the cell is impregnated with a liquid or viscous electrolyte organic or aqueous type, or may be of solid state. The electrolyte, an ionic conductor enters the pores of the electrodes and serves as conductive connection between the electrodes across the separator. Last but not least the housing will be hermetically closed to ensure stable behavior over the specified life time.
This construction gives an electrochemical capacitor with a capacitance value in the "Farad" range.
Styles
[edit]References
[edit]- ^ a b c Marin S. Halper, James C. Ellenbogen (March 2006). "Supercapacitors: A Brief Overview" (PDF). MITRE Nanosystems Group. Retrieved 2013-05-14.
- ^ a b Cite error: The named reference
Namisnykwas invoked but never defined (see the help page). - ^ Cite error: The named reference
Frackowiak1was invoked but never defined (see the help page). - ^ "The electrical double layer". 2011. Retrieved 20 January 2014.
- ^ Cite error: The named reference
Srinivasanwas invoked but never defined (see the help page). - ^ a b B. E. Conway (1999), Electrochemical Supercapacitors: Scientific Fundamentals and Technological Applications (in German), Berlin: Springer, pp. 1–8, ISBN 0306457369 see also Brian E. Conway in Electrochemistry Encyclopedia: ELECTROCHEMICAL CAPACITORS Their Nature, Function, and Applications
- ^ E. Frackowiak, F. Beguin: Carbon Materials For The Electrochemical Storage Of Energy In Capacitors. In: CARBON. 39, 2001, S. 937–950 (PDF) E. Frackowiak, K. Jurewicz, S. Delpeux, F. Béguin: Nanotubular Materials For Supercapacitors. In: Journal of Power Sources. Volumes 97–98, Juli 2001, S. 822–825, doi:10.1016/S0378-7753(01)00736-4.
- ^ Garthwaite, Josie (12 July 2011). "How ultracapacitors work (and why they fall short)". Earth2Tech. GigaOM Network. Retrieved 23 April 2013.
Double-layer capacitance - draft for own article
[edit]Double-layer capacitance store electrical energy by means of an electrical double layer effect (EDL efffect).[1][2] This electrical phenomenon appears at the interface between a metallic conductive electrode and an adjacent liquid electrolyte in a supercapacitor. Along this boundary between two different phases two layers of ions with opposite polarity will be arise if voltage applied. These two layers of ions are separated by a single layer of solvent molecules adhere at the surface of the electrode serves like a dielectric in a conventional capacitor.
The amount of electric charge stored in a double-layer capacitance is linearly proportional to the applied voltage and depends primarily on the electrode surface. The unit of double-layer capacitance is farad.
History
[edit]- Development of the double layer and pseudocapacitance model see Double layer (interfacial)
- Development of the electrochemical components see Supercapacitors
DL Capacitance functionality
[edit]
Through the description of the electrical phenomena at an interface between a metallic conductive electrode and a liquid electrolyte, Helmholtz laid the theoretical foundations of a double layer. This phenomenon will be used in every electrochemical capacitors to store electrical energy.
Every electrochemical capacitor has two electrodes, mechanically separated by a separator, which are electrically connected to each other via the electrolyte. The electrolyte is a mixture of positive and negative ions dissolved in a solvent such as water. At each of the two electrodes surfaces originates an area in which the liquid electrolyte contacts the conductive metallic surface of the electrode. This interface forms a common boundary among two different phases of matter, such as an insoluble solid electrode surface and an adjacent liquid electrolyte. In this interface occurs a very special phenomenon of the double layer effect.[1]</ref>[2]
Applying a voltage to this arrangement both electrodes generates two layers of polarized ions. One layer of ions is in the surface lattice structure of the electrode. The other layer, with opposite polarity, originates from dissolved and solvated ions distributed in the electrolyte and has moved in direction of the polarized electrode. These two layers of polarized ions are separated by a monolayer of solvent molecules, f. e. for water as solvent by water molecules. The molecular monolayer forms the inner Helmholtz plane (IHP). It adheres by physical adsorption on the surface of the electrode and separates the oppositely polarized ions from each other, becoming a molecular dielectric. The forces that cause the adhesion are not chemical bonds but physical forces. Chemical bonds persist within of the adsorbed molecules, but they are polarized.
The amount of charge in the electrode is matched by the magnitude of counter-charges in the outer Helmholtz plane (OHP). This is the aria close to the IHP in which the polarized electrolyte ions are collected. This separating of two layers of polarized ions through the double-layer phenomena store electrical charges as in a conventional capacitor. The double-layer charge forms a static electric field in the molecular IHP layer of the solvent molecules that corresponds to the strength of the applied voltage.
The "thickness" of a charged layer in the metallic electrode, i.e., the average extension perpendicular to the surface, is about 0.1 nm. It mainly depends on the electron density because the atoms in solid electrodes are stationary. In the electrolyte, the thickness depends on the size of the molecules of the solvent and of the movement and concentration of ions in the solvent. It ranges from 0.1 to 10 nm, and is described by the Debye length. The sum of the thicknesses is the total thickness of a double layer.
The very small thickness of the IHP creates an extremely strong electric field E over the separating solvent molecules. At a potential difference of, for example, U = 2 V and a molecular thickness of d = 0.4 nm, the electric field strength will be
To compare this figure with values from other capacitor types requires an estimation for electrolytic capacitors, the capacitors with the thinnest dielectric under the conventional capacitors. The voltage proof of aluminum oxide, the dielectric layer of aluminum electrolytic capacitors, is approximately 1.4 nm/V. For a 6.3 V capacitor therefore the layer is 8.8 nm. The electric field is 6.3 V/8.8 nm = 716 kV/mm, around 7 times lower than in the double-layer. The field strength of some 5000 kV/mm is unrealizable in conventional capacitors. No conventional dielectric material could prevent charge carrier breakthrough. In a double-layer capacitor the chemical stability of the solvent's molecular bonds prevents breakthrough.[3]
The forces that cause the adhesion of solvent molecules in the IHP are not chemical bonds but physical forces. Chemical bonds exist within the adsorbed molecules, but they are polarized.
The magnitude of the electric charge that can accumulate in the layers corresponds to the concentration of the adsorbed ions and the electrodes surface. Up to the electrolyte's decomposition voltage, this arrangement behaves like a capacitor in which the stored electrical charge is linearly dependent on the voltage.

The double-layer is like the dielectric layer in a conventional capacitor, but with the thickness of a single molecule. Using the early Helmholtz model to calculate the capacitance the model predicts a constant differential capacitance Cd independent from the charge density, even depending on the dielectric constant ε and the charge layer separation δ.
If the electrolyte solvent is water then the influence of the high field strength creates a permittivity ε of 6 (instead of 80 without an applied electric field) and the layer separation δ ca. 0.3 nm, the Helmholtz model predicts a differential capacitance value of about 18 F/cm2.[4] This value can be used to calculate capacitance values using the standard formula for conventional plate capacitors if only the surface of the electrodes is known. This capacitance can be calculated with:
- .
The capacitance C is greatest in components made from materials with a high permittivity ε, large electrode plate surface areas A and a small distance d between plates. Because activated carbon electrodes have a very high surface area in the range of 10 to 40 µF/cm2 and an extremely thin double-layer distance which is on the order of a few ångströms (0.3-0.8 nm), it is understandable why supercapacitors have the highest capacitance values among the capacitors.[5][6]
In real produced supercapacitors with a high amount of double-layer capacitance the capacitance value not alone depends on electrode surface and DL distance. Additional several other parameters such as electrode material and structure, electrolyte mixture, and amount of pseudocapacitance gives rise to capacitance value.[1]
Because an electrochemical capacitor is composed out of two electrodes electric charge in the Helmholtz layer at one electrode is mirrored (with opposite polarity) in the second Helmholtz layer at the second electrode. Therefore the total capacitance value of a double-layer capacitor is the result of two capacitors connected in series. If both electrodes have approximately the same capacitance value, as in symmetrical supercapacitors, the total value is roughly half that of one electrode.
DL construction
[edit]For real electrochemical capacitors using double-layer capacitance see Supercapacitor#Constuction
DL materials
[edit]For real electrochemical capacitors using double-layer capacitance see Supercapacitor#Materials
Application
[edit]For applications of real electrochemical capacitors using double-layer capacitance see Supercapacitor#Applications
Literature
[edit]- Héctor D. Abruña; Yasuyuki Kiya; Jay C. Henderson (2008), "Batteries and electrochemical capacitors" (PDF), Physics Today (in German), no. 12, pp. 43–47
- F. Béguin, E. Raymundo-Piñero, E. Frackowiak, Carbons for Electrochemical Energy Storage and Conversion Systems, Chapter 8. Electrical Double-Layer Capacitors and Pseudocapacitors, CRC Press 2009, Pages 329–375, Print ISBN 978-1-4200-5307-4, eBook ISBN 978-1-4200-5540-5, DOI0.1201/9781420055405-c8
- J. O'M. Bockris; M. A. V. Devanathan; K. Muller (1963), "On the Structure of Charged Interfaces", Proceedings of the Royal Society (in German), vol. 274, no. 1356, pp. 55–79, doi:10.1098/rspa.1963.0114, S2CID 94958336
- B. E. Conway (1999), Electrochemical Supercapacitors: Scientific Fundamentals and Technological Applications (in German), Berlin: Springer, ISBN 0306457369
- K. W. Leitner, M. Winter, J. O. Besenhard, Composite supercapacitor electrodes, Journal of Solid State Electrochemistry, Publisher Springer-Verlag, Volume 8, Issue 1, pp 15–16, Date 2003-12-01, DOI 10.1007/s10008-003-0412-x, Print ISSN1432-8488, Online ISSN1433-0768, |url=http://link.springer.com/article/10.1007%2Fs10008-003-0412-x?LI=true |title=Composite supercapacitor electrodes - Springer |publisher=Link.springer.com |date=2003-12-01 |accessdate=2013-05-24
- Yu. M. Volfkovich, T. M. Serdyuk, Electrochemical Capacitors, Russian Journal of Electrochemistry, September 2002, Volume 38, Issue 9, pp 935–959, 2002-09-01, DOI 10.1023/A:1020220425954, Print ISSN 1023-1935, Online ISSN 1608-3342, Publisher Kluwer Academic Publishers-Plenum Publishers |url=http://www.springerlink.com/content/k8715uk524h6h12w/ |title=Electrochemical Capacitors - Springer |publisher=Springerlink.com |date=2002-09-01 |accessdate=2013-05-24}}
- Jiujun Zhang; Lei Zhang; Hansan Liu; Andy Sun; Ru-Shi Liu (2011), Electrochemical Technologies for Energy Storage and Conversion, Band 1 (in German), Weinheim: Wiley-VCH, pp. 317–376, ISBN 978-3-527-32869-7
{{citation}}: Unknown parameter|comment=ignored (help)
References
[edit]- ^ a b c Z. Stojek, The Electrical Double Layer and Its Structure, [1]
- ^ a b "The electrical double layer". 2011. Retrieved 2014-01-20.
- ^ Daniel Gräser, Christoph Schmid: Supercap, Grundlagen - Eigenschaften – Anwendungen. Berner Fachhochschule, Semesterarbeit in Technologie und Deutsch (PDF).
- ^ S. Srinivasan, Fuel Cells, From Fundamentals to Applications, Springer eBooks, 2006, ISBN 978-0-387-35402-6,[2] Download CHAPTER 2, ELECTRODE/ELECTROLYTE INTERFACES: STRUCTURE AND KINETICS OF CHARGE TRANSFER (pdf, 769 kB) [3]
- ^ Marin S. Halper, James C. Ellenbogen (March 2006). Supercapacitors: A Brief Overview (PDF) (Technical report). MITRE Nanosystems Group. Retrieved 2014-01-20.
{{cite tech report}}: Cite has empty unknown parameter:|1=(help) - ^ Adam Marcus Namisnyk. A SURVEY OF ELECTROCHEMICAL SUPERCAPACITOR TECHNOLOGY (PDF) (Technical report). Retrieved 2014-01-20.
Pseudocapacitance - draft for own article
[edit]
Pseudocapacitance stores electrical energy electrochemically by means of reversible faradaic redox reactions on the surface of suitable electrodes in an electrochemical capacitor with a electric double-layer.[1][2][3] Pseudocapacitance is accompanied with an electron charge-transfer between electrolyte and electrode coming from a de-solvated and adsorbed ion whereby only one electron per charge unit is participating. This faradaic charge transfer originates by a very fast sequence of reversible redox, elctrosorption or intercalation processes. The adsorbed ion has no chemical reaction with the atoms of the electrode. No chemical bonds arise.[4] Only a charge-transfer take place.
A faradaic pseudocapacitance still only occurs together with a static double-layer capacitance. Pseudocapacitance and double-layer capacitance both contribute indivisible to the total capacitance value of the chemical capacitor. The amount of pseudocapacitance depends on the surface area, material and structure of the electrodes. The pseudocapacitance may exceed the value of double-layer capacitance for the same surface area by factor 100, depending on the nature and the structure of the electrode.[1]
The amount of electric charge stored in a pseudocapacitance is linearly proportional to the applied voltage. The unit of pseudocapacitance is farad.
History
[edit]- Development of the double layer and pseudocapacitance model see Double layer (interfacial)
- Development of the electrochemical components see Supercapacitors
Redox reactions
[edit]Differences
[edit]Rechargeable batteries
[edit]Redox reactions in batteries with faradaic charge-transfer between an electrolyte and the surface of an electrode were characterized decades ago. These chemical processes are associated with chemical reactions of the electrode materials usually with attendant phase changes. Although these chemical processes are relatively reversible, battery charge/discharge cycles often irreversibly produce unreversed chemical reaction products of the reagents. Accordingly, the cycle-life of rechargeable batteries is usually limited. Further, the reaction products lowers power density. Additionally, the chemical processes are relatively slow, extending charge/discharge times.
Electro-chemical capacitors
[edit]
A fundamental difference between redox reactions in batteries and in electrochemical capacitors (Supercapacitors) is that in the latter, the reactions are very a fast sequence of reversible processes with electron transfer without any phase changes of the electrode molecules. They does not involve making or breaking chemical bonds. The de-solvated atoms or ions contributing the pseudocapacitance simply cling[4] to the atomic structure of the electrode and charges are distributed on surfaces by physical adsorption processes. Compared with batteries, supercapacitor faradaic processes are much faster and much more stable over the time because they leave no or fewer reaction products leading to a degradation of capacitance.
This behavior is the essence of the new class of capacitance, termed “pseudocapacitance”.
Pseudocapacitive processes lead to a charge dependent linear capacitive behavior as well as the accomplishing non-faradaic double-layer capacitance in contrast to batteries, which have nearly a charge-independent behavior. Pseudocapacitance and double-layer capacitance both contribute indivisible to the total capacitance value of a supercapacitor like the both sides of the same coin. The amount of pseudocapacitance depends on the surface area, material and structure of the electrodes. The pseudocapacitance may exceed the value of double-layer capacitance for the same surface area by factor 100, depending on the nature and the structure of the electrode.[1]
Capacitance functionality
[edit]
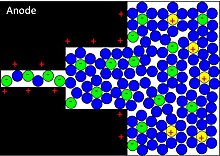
Applying a voltage at the capacitor terminals moves the polarized ions or charged atoms in the electrolyte to the opposite polarized electrode. Between the surfaces of the electrodes and the adjacent electrolyte an electric double-layer will be generated. One layer of ions on the surface atoms of the electrode and the second layer of adjacent polarized and solvated ions in the electrolyte which have moved to the opposite polarized electrode. The two ion layers are separated by a layer of single solvent molecules of the electrolyte. Between the two electrical separated layers a static electric field has formed which results in a double-layer capacitance. Accompanied with the electric double-layer some de-solvated ions out of the electrolyte pervade the separating solvent layer and will be adsorbed by the surface atoms of the electrode. They will be specifically adsorbed and deliver they charge to the electrode.
With other words: In a Helmholtz double-layer the ions in the electrolyte may also act as electron donors transferring with a charge-transfer electrons to the atoms of the electrode resulting in a faradaic current. This faradaic charge transfer originates by a very fast sequence of reversible redox reactions, electrosorptions or intercalation processes between electrolyte and the electrode surface is called pseudocapacitance.[5]
Depending on the electrode's structure or surface material, pseudocapacitance can originate when specifically adsorbed cations pervade the double-layer, proceeding in several one-electron stages. The electrons involved in the faradaic processes are transferred to or from valence-electron states (orbitals) of the redox electrode reagent. They enter the negative electrode and flow through the external circuit to the positive electrode where a second double-layer with an equal number of anions has formed. But these anions don’t accept the electrons. They remain on the electrode's surface in the charged state, and the electrons remain in the strongly ionized and "electron hungry" transition-metal ions of the electrode. This kind of pseudocapacitance has a linear function within narrow limits and is determined by the potential-dependent degree of surface coverage of the adsorbed anions. The storage capacity of the pseudocapacitance is limited by the finite quantity of reagent or of available surface.
Description of the systems that give rise to pseudocapacitance:[5]
- Redox system: Ox + ze‾ ⇌ Red and O‾
2 + H
1 ⇌ in lattice - Intercalation system: Liˡ in "Ma
2" - Electrosorption, underpotential deposition of metal adatoms: M10−
꞊ + S + ze‾ ⇌ SM (S = surface lattice sites) or H
ˡ e‾ + S ⇌ SH
All three types of electrochemical processes giving rise to pseudocapacitance have been utilized in supercapacitors.[5][6]
When discharging pseudocapacitance, the charge transfer is reversed and the ions or atoms leave the double-layer and distribute randomly into the electrolyte.
Pseudocapacitive materials
[edit]The ability of electrodes to accomplish pseudocapacitance effects by redox reactions of electroactive species, electrosorption of H or metal ad-atoms or intercalation strongly depends on the chemical affinity of electrode materials to the ions adsorbed on the electrode surface as well as on the structure and dimension of the electrode pores. Materials exhibiting redox behavior for use as electrodes in pseudocapacitors are transition-metal oxides inserted by doping in the conductive electrode material such as active carbon, as well as conducting polymers such as polyaniline or derivatives of polythiophene covering the electrode material.
Transition metal oxides
[edit]The best researched and understood by the research of B. E. Conway [1][7] are electrodes out of transition metal oxides for high amount of pseudocapacitance. Many oxides of transition metals like ruthenium (RuO
2), iridium (IrO
2), iron (Fe
3O
4), manganese (MnO
2) or sulfides such as titanium sulfide (TiS
2) or their combinations are able to generate many faradaic electron–transferring reactions combined with low conducting resistance.[8] Ruthenium dioxide in combination with H
2SO
4 electrolyte provides one of the best examples of pseudocapacitance. Charge/discharge takes place with electron charge transfer or removal and occurs over a window of about 1.2 V per electrode. In addition for these transition metal electrodes, its reversibility is excellent, with a cycle life over several hundred-thousand cycles.
Here the pseudocapacitance originates out of a coupled, reversible redox reaction with several oxidation steps with overlapping potential. The electrons mostly come from the valence orbitals of the electrode. The electron transfer reaction is very fast, and can be accompanied with high currents.
The electron transfer reaction take place according to:
- where [9]
During charging and discharging in this charge-transfer transition H+ protons are incorporated into or removed from the crystal lattice of ruthenium. This generates storage of electrical energy without chemical transformation. The OH groups are deposited as a molecular layer on the electrode surface and remain in the region of the Helmholtz layer. Since the measurable voltage from the redox reaction is proportional to the charged state, the reaction behaves like a capacitor rather than a battery, whose voltage is largely independent of the state of charge.
Conducting polymers
[edit]Another type of material with a high amount of pseudocapacitance is electron-conducting polymers. Conductive polymers electrodes include polyaniline, polythiophene, polypyrrole and polyacetylene have a lower reversibility of the redox reaction processes with faradaic charge transfer than electrodes with transition metal oxides and suffer from a limited stability during cycling.[10] Such electrodes employ electrochemical doping or dedoping of the polymers with anions and cations. The greatest capacitance and power density have the n/p-type polymer configuration, with one negatively charged (n-doped) and one positively charged (p-doped) electrode.
Pseudocapacitive structures
[edit]Pseudocapacitance may also originate from the structure and especially from the pore size of the electrodes. The use of carbide-derived carbons (CDCs) or carbon nanotubes /CNTs for electrodes provides a network of very small pores formed by nanotube entanglement. These nanoporous materials have diameters in the range of <2 nm that can be referred to as intercalated pores. Solvated ions in the electrolyte can’t enter these small pores but de-solvated ions which have reduced their ion dimensions are able to enter, resulting in larger ionic packing density and increased charge storage. The tailored sizes of pores in nano-structured carbon electrodes can maximize ion confinement, increasing specific capacitance by faradaic H
2 adsorption treatment. Occupation of these pores by de-solvated ions from the electrolyte solution occurs according to (faradaic) intercalation.[11][12][13]
Verification of pseudocapacitance
[edit]
The properties of pseudocapacitance can be expressed in a cyclic voltammogram. For an ideal double-layer capacitor the sign of the current changes immediately after reversing the potential and the shape of the voltammetry is rectangular. For this electrostatic energy storage the current is independent on potential of the electrode. For double-layer capacitors with resistive losses, the shape changes into a parallelogram. For electrodes with faradaic pseudocapacitance the electrical charge stored in the capacitor is strongly dependent on the potential. Therefore the voltammetry characteristics deviate from the parallelogram, caused by a delay while reversing the potential, coming from kinetic processes during charging.[14][15]
Applications
[edit]Pseudocapacitance arises in supercapacitors
Literature
[edit]- Héctor D. Abruña; Yasuyuki Kiya; Jay C. Henderson (2008), "Batteries and electrochemical capacitors" (PDF), Physics Today (in German), no. 12, pp. 43–47
- F. Béguin, E. Raymundo-Piñero, E. Frackowiak, Carbons for Electrochemical Energy Storage and Conversion Systems, Chapter 8. Electrical Double-Layer Capacitors and Pseudocapacitors, CRC Press 2009, Pages 329–375, Print ISBN 978-1-4200-5307-4, eBook ISBN 978-1-4200-5540-5, DOI0.1201/9781420055405-c8
- J. O'M. Bockris; M. A. V. Devanathan; K. Muller (1963), "On the Structure of Charged Interfaces", Proceedings of the Royal Society (in German), vol. 274, no. 1356, pp. 55–79, doi:10.1098/rspa.1963.0114, S2CID 94958336
- B. E. Conway (1999), Electrochemical Supercapacitors: Scientific Fundamentals and Technological Applications (in German), Berlin: Springer, ISBN 0306457369
- K. W. Leitner, M. Winter, J. O. Besenhard, Composite supercapacitor electrodes, Journal of Solid State Electrochemistry, Publisher Springer-Verlag, Volume 8, Issue 1, pp 15–16, Date 2003-12-01, DOI 10.1007/s10008-003-0412-x, Print ISSN1432-8488, Online ISSN1433-0768, |url=http://link.springer.com/article/10.1007%2Fs10008-003-0412-x?LI=true |title=Composite supercapacitor electrodes - Springer |publisher=Link.springer.com |date=2003-12-01 |accessdate=2013-05-24
- Volkmar M. Schmidt (2003), Elektrochemische Verfahrenstechnik. Grundlagen, Reaktionstechnik, Prozessoptimierung (in German), Weinheim: Wiley-VCH, pp. 539–639, ISBN 3-527-29958-0
{{citation}}: Unknown parameter|comment=ignored (help) - Yu. M. Volfkovich, T. M. Serdyuk, Electrochemical Capacitors, Russian Journal of Electrochemistry, September 2002, Volume 38, Issue 9, pp 935–959, 2002-09-01, DOI 10.1023/A:1020220425954, Print ISSN 1023-1935, Online ISSN 1608-3342, Publisher Kluwer Academic Publishers-Plenum Publishers |url=http://www.springerlink.com/content/k8715uk524h6h12w/ |title=Electrochemical Capacitors - Springer |publisher=Springerlink.com |date=2002-09-01 |accessdate=2013-05-24}}
- Jiujun Zhang; Lei Zhang; Hansan Liu; Andy Sun; Ru-Shi Liu (2011), Electrochemical Technologies for Energy Storage and Conversion, Band 1 (in German), Weinheim: Wiley-VCH, pp. 317–376, ISBN 978-3-527-32869-7
{{citation}}: Unknown parameter|comment=ignored (help)
References
[edit]- ^ a b c d B. E. Conway (1999), Electrochemical Supercapacitors: Scientific Fundamentals and Technological Applications (in German), Berlin: Springer, pp. 1–8, ISBN 0306457369 see also Brian E. Conway in Electrochemistry Encyclopedia: ELECTROCHEMICAL CAPACITORS Their Nature, Function, and Applications
- ^ Marin S. Halper, James C. Ellenbogen (March 2006). Supercapacitors: A Brief Overview (PDF) (Technical report). MITRE Nanosystems Group. Retrieved 2014-01-20.
{{cite tech report}}: Cite has empty unknown parameter:|1=(help) - ^ E. Frackowiak, F. Beguin: Carbon Materials For The Electrochemical Storage Of Energy In Capacitors. In: CARBON. 39, 2001, S. 937–950 (PDF) E. Frackowiak, K. Jurewicz, S. Delpeux, F. Béguin: Nanotubular Materials For Supercapacitors. In: Journal of Power Sources. Volumes 97–98, Juli 2001, S. 822–825, doi:10.1016/S0378-7753(01)00736-4.
- ^ a b Garthwaite, Josie (12 July 2011). "How ultracapacitors work (and why they fall short)". Earth2Tech. GigaOM Network. Retrieved 23 April 2013.
- ^ a b c B.E. Conway, W.G. Pell, Double-layer and pseudocapacitance types of electrochemical capacitors and their applications to the development of hybrid components
- ^ B. E. Conway, V. Birss, J. Wojtowicz, The role and the utilization of pseudocapacitance for energy storage by supercapacitors, Journal of Power Sources, Volume 66, Issues 1–2, May–June 1997, Pages 1–14
- ^ B. E. Conway (Mai 1991), "Transition from 'Supercapacitor' to 'Battery' Behavior in Electrochemical Energy Storage", Journal of the Electrochemical Society (in German), vol. 138, no. 6, pp. 1539–1548, doi:10.1149/1.2085829
{{citation}}: Check date values in:|date=(help)CS1 maint: date and year (link) - ^ M. Jayalakshmi, K. Balasubramanian, Simple Capacitors to Supercapacitors - An Overview, Int. J. Electrochem. Sci., 3 (2008) 1196 – 1217, PDF
- ^ P. Simon, Y.Gogotsi, Materials for electrochemical capacitors, nature materials, VOL 7, NOVEMBER 2008
- ^ Yu.M. Volfkovich, A.A. Mikhailin, D.A. Bograchev, V.E. Sosenkin and V.S. Bagotsky, Studies of Supercapacitor Carbon Electrodes with High Pseudocapacitance, A. N. Frumkin Institute of Physical Chemistry and Electrochemistry, Russian Academy of Sciences, Moscow, Russia, Dr. Ujjal Kumar Sur (Ed.), ISBN 978-953-307-830-4
- ^ A.G. Pandolfo, A.F. Hollenkamp, Carbon properties and their role in supercapacitors, Journal of Power Sources 157 (2006) 11–27
- ^ B.P. Bakhmatyuk, B.Ya. Venhryn, I.I. Grygorchak, M.M. Micov and S.I. Mudry, INTERCALATION PSEUDO-CAPACITANCE IN CARBON SYSTEMS OF ENERGY STORAGE
- ^ P. Simon, A. Burke, Nanostructured carbons: Double-Layer capacitance and more
- ^ Elzbieta Frackowiak, Francois Beguin, PERGAMON, Carbon 39 (2001) 937–950, Carbon materials for the electrochemical storage of energy in Capacitors
- ^ Why does an ideal capacitor give rise to a rectangular cyclic voltammogram