User:Mr. Ibrahem/Ibandronic acid
 | |
| Clinical data | |
|---|---|
| Trade names | Boniva, Bonviva, Bondronat, others |
| Other names | Ibandronate sodium |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 0.6% |
| Protein binding | 90.9 to 99.5% (concentration-dependent) |
| Metabolism | Nil |
| Elimination half-life | 10 to 60 hours |
| Excretion | Kidney |
| Chemical and physical data | |
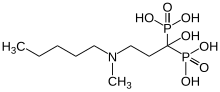
| Formula | C9H23NO7P2 |
| Molar mass | 319.231 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Ibandronic acid, also known as ibandronate, is a medication used treat of osteoporosis, high calcium due to cancer, and bone metastases from breast cancer.[1][2] It may be taken by mouth or injection into a vein.[1]
Common side effects include heart burn, low calcium, weakness, headache, and fever.[2] Other side effects may include anaphylaxis, esophagitis, femur fracture, and osteonecrosis of the jaw.[2][3] It is a bisphosphonate and works by stopping bone breakdown by cells known as osteoclasts.[1][2]
Ibandronic acid was patented in 1986 by Boehringer Mannheim and approved for medical use in 1996.[4] It is available as a generic medication.[1] In the United Kingdom a 150 mg pill cost the NHS about £4.50 as of 2021.[1] This amount in the United States is about 12 USD.[5]
References
[edit]- ^ a b c d e BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 769. ISBN 978-0857114105.
- ^ a b c d "Bondronat". Archived from the original on 11 January 2021. Retrieved 24 November 2021.
- ^ "Ibandronate Monograph for Professionals". Drugs.com. Archived from the original on 11 December 2018. Retrieved 24 November 2021.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 523. ISBN 9783527607495. Archived from the original on 2021-03-18. Retrieved 2020-12-06.
- ^ "Ibandronate Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 24 November 2021.
