Cubane
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Cubane[1] | |||
| Systematic IUPAC name
Pentacyclo[4.2.0.02,5.03,8.04,7]octane | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C8H8 | |||
| Molar mass | 104.15 g/mol | ||
| Appearance | Transparent[2] crystalline solid | ||
| Density | 1.29 g/cm3 | ||
| Melting point | 133.5 °C (272.3 °F; 406.6 K)[3] | ||
| Boiling point | 161.6 °C (322.9 °F; 434.8 K)[3] | ||
| Related compounds | |||
Related hydrocarbons
|
Cuneane Dodecahedrane Tetrahedrane Prismane Prismane C8 | ||
Related compounds
|
Octafluorocubane Octanitrocubane Octaazacubane Mirex | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Cubane is a synthetic hydrocarbon compound with the formula C8H8. It consists of eight carbon atoms arranged at the corners of a cube, with one hydrogen atom attached to each carbon atom. A solid crystalline substance, cubane is one of the Platonic hydrocarbons and a member of the prismanes. It was first synthesized in 1964 by Philip Eaton and Thomas Cole.[4] Before this work, Eaton believed that cubane would be impossible to synthesize due to the "required 90 degree bond angles".[5][6] The cubic shape requires the carbon atoms to adopt an unusually sharp 90° bonding angle, which would be highly strained as compared to the 109.45° angle of a tetrahedral carbon. Once formed, cubane is quite kinetically stable, due to a lack of readily available decomposition paths. It is the simplest hydrocarbon with octahedral symmetry.
Having high potential energy and kinetic stability makes cubane and its derivative compounds useful for controlled energy storage. For example, octanitrocubane and heptanitrocubane have been studied as high-performance explosives. These compounds also typically have a very high density for hydrocarbon molecules. The resulting high energy density means a large amount of energy can be stored in a comparably smaller amount of space, an important consideration for applications in fuel storage and energy transport. Furthermore, their geometry and stability make them suitable isosteres for benzene rings.[7]
Synthesis
[edit]The classic 1964 synthesis starts with the conversion of 2-cyclopentenone to 2-bromocyclopentadienone:[4][8]
Allylic bromination with N-bromosuccinimide in carbon tetrachloride followed by addition of molecular bromine to the alkene gives a 2,3,4-tribromocyclopentanone. Treating this compound with diethylamine in diethyl ether causes elimination of two equivalents of hydrogen bromide to give the diene product.
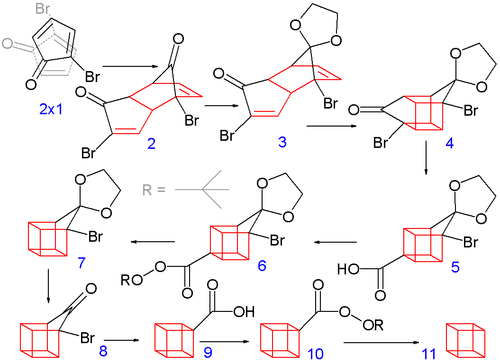
The construction of the eight-carbon cubane framework begins when 2-bromocyclopentadienone undergoes a spontaneous Diels-Alder dimerization. One ketal of the endo isomer is subsequently selectively deprotected with aqueous hydrochloric acid to 3.
In the next step, the endo isomer 3 (with both alkene groups in close proximity) forms the cage-like isomer 4 in a photochemical [2+2] cycloaddition. The bromoketone group is converted to ring-contracted carboxylic acid 5 in a Favorskii rearrangement with potassium hydroxide. Next, the thermal decarboxylation takes place through the acid chloride (with thionyl chloride) and the tert-butyl perester 6 (with tert-butyl hydroperoxide and pyridine) to 7; afterward, the acetal is once more removed in 8. A second Favorskii rearrangement gives 9, and finally another decarboxylation gives, via 10, cubane (11).
A more approachable laboratory synthesis of disubstituted cubane involves bromination of the ethylene ketal of cyclopentanone to give a tribromocyclopentanone derivative. Subsequent steps involve dehydrobromination, Diels-Alder dimerization, etc.[9][10]

The resulting cubane-1,4-dicarboxylic acid is used to synthesize other substituted cubanes. Cubane itself can be obtained nearly quantitatively by photochemical decarboxylation of the thiohydroxamate ester (the Barton decarboxylation).[11]
Derivatives
[edit]The synthesis of the octaphenyl derivative from tetraphenylcyclobutadiene nickel bromide by Freedman in 1962 pre-dates that of the parent compound. It is a sparingly soluble colourless compound that melts at 425–427 °C.[3][12][13][14] A hypercubane, with a hypercube-like structure, was predicted to exist in a 2014 publication.[15][16] Two isomers of cubene have been synthesized, and a third analyzed computationally. The alkene in ortho-cubene is exceptionally reactive due to its pyramidalized geometry. At the time of its synthesis, this was the most pyramidalized alkene to have been made.[17] The meta-cubene isomer is even less stable, and the para-cubene isomer probably only exists as a diradical rather than an actual diagonal bond.[18]
In 2022, both heptafluorocubane and octafluorocubane were synthesized.[19] Octafluorocubane is of theoretical interest because of its unusual electronic structure,[20] which is indicated by its susceptibility to undergo reduction to a detectable anion C
8F−
8, with a free electron trapped inside the cube, in effect making it the world's smallest box.[21]
Cubylcubanes and oligocubanes
[edit]Cubene (1,2-dehydrocubane) and 1,4-cubanediyl(1,4-dehydrocubane) are enormously strained compounds which both undergo nucleophilic addition very rapidly, and this has enabled chemists to synthesize cubylcubane. X-ray diffraction structure solution has shown that the central cubylcubane bond is exceedingly short (1.458 Å), much shorter than the typical C-C single bond (1.578 Å). This is attributed to the fact that the exocyclic orbitals of cubane are s-rich and close to the nucleus.[22] Chemists at the University of Chicago extended and modified the sequence in a way that permits the preparation of a host of [n]cubylcubane oligomers.[23] The [n]cubylcubanes are rigid molecular rods with the particular promise at the time of making liquid crystals with exceptional UV transparency. As the number of linked cubane units increases, the solubility of [n]cubylcubane plunges; as a result, only limited chain length (up to 40 units) have been synthesized in solutions. The skeleton of [n]cubylcubanes is still composed of enormously strained carbon cubes, which therefore limit its stability. In contrast, researchers at Penn State University showed that poly-cubane synthesized by solid-state reaction is 100% sp3 carbon bonded with a tetrahedral angle (109.5°) and exhibits exceptional optical properties (high refractive index).[24]
Reactions
[edit]Cuneane may be produced from cubane by a metal-ion-catalyzed σ-bond rearrangement.[25][26]
With a rhodium catalyst, it first forms syn-tricyclooctadiene, which can thermally decompose to cyclooctatetraene at 50–60 °C.[27]
See also
[edit]References
[edit]- ^ Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 169. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
The retained names adamantane and cubane are used in general nomenclature and as preferred IUPAC names.
- ^ "Start".
- ^ a b c Biegasiewicz, Kyle; Griffiths, Justin; Savage, G. Paul; Tsanakstidis, John; Priefer, Ronny (2015). "Cubane: 50 years later". Chemical Reviews. 115 (14): 6719–6745. doi:10.1021/cr500523x. PMID 26102302.
- ^ a b Eaton, Philip E.; Cole, Thomas W. (1964). "Cubane". J. Am. Chem. Soc. 86 (15): 3157–3158. doi:10.1021/ja01069a041.
- ^ Teachers, University of New South Wales Summer School for Chemistry (1963). Approach to Chemistry: Lectures and Workshop Reports of the ... Summer School for Chemistry Teachers. The University. p. 98. "This compound was described only a few months ago and, curiously enough, it is quite easy to make, although only a year ago I would have predicted that it would be difficult, or even impossible, to synthesize."
- ^ Moore, John W.; Stanitski, Conrad L.; Jurs, Peter C. (2002). Chemistry: The Molecular Science. Harcourt College Publishers. p. 372. ISBN 978-0-03-032011-8. "This sharp bond angle creates severe bond strain in cubane, a compound thought previously impossible to synthesize because of the required 90° bond angles."
- ^ Wiesenfeldt, Mario P.; Rossi-Ashton, James A.; Perry, Ian B.; Diesel, Johannes; Garry, Olivia L.; Bartels, Florian; Coote, Susannah C.; Ma, Xiaoshen; Yeung, Charles S.; Bennett, David J.; MacMillan, David W. C. (June 2023). "General access to cubanes as benzene bioisosteres". Nature. 618 (7965): 513–518. doi:10.1038/s41586-023-06021-8. ISSN 1476-4687. PMC 10680098.
- ^ Eaton, Philip E.; Cole, Thomas W. (1964). "The Cubane System". J. Am. Chem. Soc. 86 (5): 962–964. doi:10.1021/ja01059a072.
- ^ Bliese, Marianne; Tsanaktsidis, John (1997). "Dimethyl Cubane-1,4-dicarboxylate: A Practical Laboratory Scale Synthesis". Australian Journal of Chemistry. 50 (3): 189. doi:10.1071/C97021.
- ^ Fluorochem, Inc (July 1989). "Cubane Derivatives for Propellant Applications" (PDF). Archived (PDF) from the original on 2021-07-09.
- ^ Eaton, Philip E. (1992). "Cubane: Ausgangsverbindungen für die Chemie der neunziger Jahre und des nächsten Jahrhunderts". Angewandte Chemie (in German). 104 (11): 1447–1462. Bibcode:1992AngCh.104.1447E. doi:10.1002/ange.19921041105.
- ^ Freedman, H. H. (1961). "Tetraphenylcyclobutadiene Derivatives. II.1 Chemical Evidence for the Triplet State". J. Am. Chem. Soc. 83 (9): 2195–2196. doi:10.1021/ja01470a037.
- ^ Freedman, H. H.; Petersen, D. R. (1962). "Tetraphenylcyclobutadiene Derivatives. IV.1 "Octaphenylcubane"; A Dimer of Tetraphenylcyclobutadiene". J. Am. Chem. Soc. 84 (14): 2837–2838. doi:10.1021/ja00873a046.
- ^ Pawley, G. S.; Lipscomb, W. N.; Freedman, H. H. (1964). "Structure of the Dimer of tetraphenylcyclobutadiene". J. Am. Chem. Soc. 86 (21): 4725–4726. doi:10.1021/ja01075a042.
- ^ Pichierri, F. (2014). "Hypercubane: DFT-based prediction of an Oh-symmetric double-shell hydrocarbon". Chem. Phys. Lett. 612: 198–202. Bibcode:2014CPL...612..198P. doi:10.1016/j.cplett.2014.08.032.
- ^ "Hypercubane: DFT-based prediction of an Oh-symmetric double-shell hydrocarbon".
- ^ Eaton, Philip E.; Maggini, Michele (1988). "Cubene (1,2-dehydrocubane)". J. Am. Chem. Soc. 110 (21): 7230–7232. doi:10.1021/ja00229a057.
- ^ Minyaev, Ruslan M.; Minkin, Vladimir I.; Gribanova, Tatyana N. (2009). "2.3 A Theoretical Approach to the Study and Design of Prismane Systems". In Dodziuk, Helena (ed.). Strained Hydrocarbons. Wiley. p. 55. ISBN 9783527627141.
- ^ Sugiyama M, Akiyama M, Yonezawa Y, Komaguchi K, Higashi M, Nozaki K, Okazoe T (August 2022). "Electron in a cube: Synthesis and characterization of perfluorocubane as an electron acceptor". Science. 377 (6607): 756–759. Bibcode:2022Sci...377..756S. doi:10.1126/science.abq0516. PMID 35951682. S2CID 251515925.
- ^ Pichierri, F. Substituent effects in cubane and hypercubane: a DFT and QTAIM study. Theor Chem Acc 2017; 136: 114. doi:10.1007/s00214-017-2144-5
- ^ Krafft MP, Riess JG (August 2022). "Perfluorocubane-a tiny electron guzzler". Science. 377 (6607): 709. Bibcode:2022Sci...377..709K. doi:10.1126/science.adc9195. PMID 35951708. S2CID 251517529.
- ^ Gilardi, Richard.; Maggini, Michele.; Eaton, Philip E. (1 October 1988). "X-ray structures of cubylcubane and 2-tert-butylcubylcubane: short cage-cage bonds". Journal of the American Chemical Society. 110 (21): 7232–7234. doi:10.1021/ja00229a058. ISSN 0002-7863.
- ^ Eaton, Philip E. (1992). "Cubanes: Starting Materials for the Chemistry of the 1990s and the New Century". Angewandte Chemie International Edition in English. 31 (11): 1421–1436. doi:10.1002/anie.199214211. ISSN 1521-3773.
- ^ Huang, Haw-Tyng; Zhu, Li; Ward, Matthew D.; Wang, Tao; Chen, Bo; Chaloux, Brian L.; Wang, Qianqian; Biswas, Arani; Gray, Jennifer L.; Kuei, Brooke; Cody, George D.; Epshteyn, Albert; Crespi, Vincent H.; Badding, John V.; Strobel, Timothy A. (21 January 2020). "Nanoarchitecture through Strained Molecules: Cubane-Derived Scaffolds and the Smallest Carbon Nanothreads". Journal of the American Chemical Society. 142 (42): 17944–17955. doi:10.1021/jacs.9b12352. ISSN 0002-7863. PMID 31961671. S2CID 210870993.
- ^ Smith, Michael B.; March, Jerry (2001). March's Advanced Organic Chemistry (5th ed.). John Wiley & Sons. p. 1459. ISBN 0-471-58589-0.
- ^ Kindler, K.; Lührs, K. (1966). "Studien über den Mechanismus chemischer Reaktionen, XXIII. Hydrierungen von Nitrilen unter Verwendung von Terpenen als Wasserstoffdonatoren". Chem. Ber. 99: 227–232. doi:10.1002/cber.19660990135.
- ^ Cassar, Luigi; Eaton, Philip E.; Halpern, Jack (1970). "Catalysis of symmetry-restricted reactions by transition metal compounds. Valence isomerization of cubane". Journal of the American Chemical Society. 92 (11): 3515–3518. doi:10.1021/ja00714a075. ISSN 0002-7863.