Thionyl chloride

| |

| |

| |
| Names | |
|---|---|
| IUPAC name
Thionyl chloride
| |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.028.863 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1836 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| SOCl2 | |
| Molar mass | 118.97 g/mol |
| Appearance | Colourless liquid (yellows on ageing) |
| Odor | Pungent and unpleasant |
| Density | 1.638 g/cm3, liquid |
| Melting point | −104.5 °C (−156.1 °F; 168.7 K) |
| Boiling point | 74.6 °C (166.3 °F; 347.8 K) |
| Reacts | |
| Solubility | Soluble in most aprotic solvents: toluene, chloroform, diethyl ether. Reacts with protic solvents such as alcohols |
| Vapor pressure |
|
Refractive index (nD)
|
1.517 (20 °C)[2] |
| Viscosity | 0.6 cP |
| Structure | |
| pyramidal | |
| 1.44 D | |
| Thermochemistry | |
Heat capacity (C)
|
121.0 J/mol (liquid)[3] |
Std molar
entropy (S⦵298) |
309.8 kJ/mol (gas)[3] |
Std enthalpy of
formation (ΔfH⦵298) |
−245.6 kJ/mol (liquid)[3] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Very toxic, corrosive, releases HCl on contact with water |
| GHS labelling: | |
  
| |
| Danger | |
| H302, H314, H331 | |
| P261, P280, P305+P351+P338, P310 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
None[4] |
REL (Recommended)
|
C 1 ppm (5 mg/m3)[4] |
IDLH (Immediate danger)
|
N.D.[4] |
| Related compounds | |
Related Thionyl halides
|
|
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Thionyl chloride is an inorganic compound with the chemical formula SOCl2. It is a moderately volatile, colourless liquid with an unpleasant acrid odour. Thionyl chloride is primarily used as a chlorinating reagent, with approximately 45,000 tonnes (50,000 short tons) per year being produced during the early 1990s,[5] but is occasionally also used as a solvent.[6][7][8] It is toxic, reacts with water, and is also listed under the Chemical Weapons Convention as it may be used for the production of chemical weapons.
Thionyl chloride is sometimes confused with sulfuryl chloride, SO2Cl2, but the properties of these compounds differ significantly. Sulfuryl chloride is a source of chlorine whereas thionyl chloride is a source of chloride ions.
Production[edit]
The major industrial synthesis involves the reaction of sulfur trioxide and sulfur dichloride:[9] This synthesis can be adapted to the laboratory by heating oleum to slowly distill the sulfur trioxide into a cooled flask of sulfur dichloride.[10]
Other methods include syntheses from:
The second of the above five reactions also affords phosphorus oxychloride (phosphoryl chloride), which resembles thionyl chloride in many of its reactions. They may be separated by distillation, since thionyl chloride boils at a much lower temperature than phosphoryl chloride[citation needed].
Properties and structure[edit]
 |
SOCl2 adopts a trigonal pyramidal molecular geometry with Cs molecular symmetry. This geometry is attributed to the effects of the lone pair on the central sulfur(IV) center.
In the solid state SOCl2 forms monoclinic crystals with the space group P21/c.[11]
Stability[edit]
Thionyl chloride has a long shelf life, however "aged" samples develop a yellow hue, possibly due to the formation of disulfur dichloride. It slowly decomposes to S2Cl2, SO2 and Cl2 at just above the boiling point.[9][12] Thionyl chloride is susceptible to photolysis, which primarily proceeds via a radical mechanism.[13] Samples showing signs of ageing can be purified by distillation under reduced pressure, to give a colourless liquid.[14]

Reactions[edit]
Thionyl chloride is mainly used in the industrial production of organochlorine compounds, which are often intermediates in pharmaceuticals and agrichemicals. It usually is preferred over other reagents, such as phosphorus pentachloride, as its by-products (HCl and SO2) are gaseous, which simplifies purification of the product.
Many of the products of thionyl chloride are themselves highly reactive and as such it is involved in a wide range of reactions.
With water and alcohols[edit]
Thionyl chloride reacts exothermically with water to form sulfur dioxide and hydrochloric acid:
By a similar process it also reacts with alcohols to form alkyl chlorides. If the alcohol is chiral the reaction generally proceeds via an SNi mechanism with retention of stereochemistry;[15] however, depending on the exact conditions employed, stereo-inversion can also be achieved. Historically the use of SOCl2 with pyridine was called the Darzens halogenation, but this name is rarely used by modern chemists.

Reactions with an excess of alcohol produce sulfite esters, which can be powerful methylation, alkylation and hydroxyalkylation reagents.[16]
For example, the addition of SOCl2 to amino acids in methanol selectively yields the corresponding methyl esters.[17]
With carboxylic acids[edit]
Classically, it converts carboxylic acids to acyl chlorides:[18][19][20]
The reaction mechanism has been investigated:[21]
With nitrogen species[edit]
With primary amines, thionyl chloride gives sulfinylamine derivatives (RNSO), one example being N-sulfinylaniline. Thionyl chloride reacts with primary formamides to form isocyanides[22] and with secondary formamides to give chloroiminium ions; as such a reaction with dimethylformamide will form the Vilsmeier reagent.[23] By an analogous process primary amides will react with thionyl chloride to form imidoyl chlorides, with secondary amides also giving chloroiminium ions. These species are highly reactive and can be used to catalyse the conversion of carboxylic acids to acyl chlorides,[24] they are also exploited in the Bischler–Napieralski reaction as a means of forming isoquinolines.
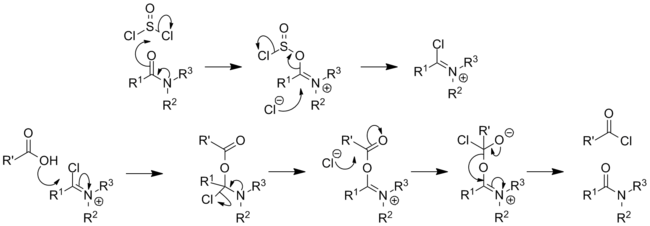
Primary amides will continue on to form nitriles if heated (Von Braun amide degradation).[25] Thionyl chloride has also been used to promote the Beckmann rearrangement of oximes.
With sulfur species[edit]
- Thionyl chloride will transform sulfinic acids into sulfinyl chlorides[26][27]
- Sulfonic acids react with thionyl chloride to produce sulfonyl chlorides.[28][29] Sulfonyl chlorides have also been prepared from the direct reaction of the corresponding diazonium salt with thionyl chloride.[30]
- Thionyl chloride can be used in variations of the Pummerer rearrangement.

With phosphorus species[edit]
Thionyl chloride converts phosphonic acids and phosphonates into phosphoryl chlorides. It is for this type of reaction that thionyl chloride is listed as a Schedule 3 compound, as it can be used in the "di-di" method of producing G-series nerve agents. For example, thionyl chloride converts dimethyl methylphosphonate into methylphosphonic acid dichloride, which can be used in the production of sarin and soman.
With metals[edit]
As SOCl2 reacts with water it can be used to dehydrate various metal chloride hydrates, such magnesium chloride (MgCl2·6H2O), aluminium chloride (AlCl3·6H2O), and iron(III) chloride (FeCl3·6H2O).[9] This conversion involves treatment with refluxing thionyl chloride and follows the following general equation:[31]
Other reactions[edit]
- Thionyl chloride can engage in a range of different electrophilic addition reactions. It adds to alkenes in the presence of AlCl3 to form an aluminium complex which can be hydrolysed to form a sulfinic acid. Both aryl sulfinyl chlorides and diaryl sulfoxides can be prepared from arenes through reaction with thionyl chloride in triflic acid[32] or the presence of catalysts such as BiCl3, Bi(OTf)3, LiClO4 or NaClO4.[33][34]
- In the laboratory, a reaction between thionyl chloride and an excess of anhydrous alcohol can be used to produce anhydrous alcoholic solutions of HCl.
- Thionyl chloride undergoes halogen exchange reactions to give other thionyl species.
- Reactions with fluorinating agents such as antimony trifluoride give thionyl fluoride:
- A reaction with hydrogen bromide gives thionyl bromide:
- Thionyl iodide can likewise be prepared by a reaction with potassium iodide, but is reported to be highly unstable.[35][36]
Batteries[edit]

Thionyl chloride is a component of lithium–thionyl chloride batteries,[37] where it acts as the positive electrode (in batteries: cathode) with lithium forming the negative electrode (anode); the electrolyte is typically lithium tetrachloroaluminate. The overall discharge reaction is as follows:
These non-rechargeable batteries had many advantages over other forms of lithium batteries such as a high energy density, a wide operational temperature range, and long storage and operational lifespans. However, their high cost, non-rechargeability, and safety concerns have limited their use. The contents of the batteries are highly toxic and require special disposal procedures; additionally, they may explode if shorted. The technology was used on the Sojourner Mars rover.
Safety[edit]
SOCl2 is highly reactive and can violently release hydrochloric acid upon contact with water and alcohols. It is also a controlled substance under the Chemical Weapons Convention, where it is listed as a Schedule 3 substance, since it is used in the manufacture of G-series nerve agents[citation needed] and the Meyer and Meyer–Clarke methods of producing sulfur-based mustard gases.[38]
History[edit]
In 1849, the French chemists Jean-François Persoz and Bloch, and the German chemist Peter Kremers (1827-?), independently first synthesized thionyl chloride by reacting phosphorus pentachloride with sulfur dioxide.[39][40] However, their products were impure: both Persoz and Kremers claimed that thionyl chloride contained phosphorus,[41] and Kremers recorded its boiling point as 100 °C (instead of 74.6 °C). In 1857, the German-Italian chemist Hugo Schiff subjected crude thionyl chloride to repeated fractional distillations and obtained a liquid which boiled at 82 °C and which he called Thionylchlorid.[42] In 1859, the German chemist Georg Ludwig Carius noted that thionyl chloride could be used to make acid anhydrides and acyl chlorides from carboxylic acids and to make alkyl chlorides from alcohols.[43]
See also[edit]
References[edit]
- ^ Thionyl chloride in Linstrom, Peter J.; Mallard, William G. (eds.); NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD) (retrieved 2014-05-11)
- ^ Patnaik, Pradyot (2003). Handbook of Inorganic Chemicals. New York, NY: McGraw-Hill. ISBN 0-07-049439-8.
- ^ a b c Lide, David R.; et al., eds. (1996). CRC Handbook of Chemistry and Physics (76th ed.). Boca Raton, FL: CRC Press. pp. 5–10. ISBN 0-8493-0476-8.
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0611". National Institute for Occupational Safety and Health (NIOSH).
- ^ Lauss, H.-D.; Steffens, W. "Sulfur Halides". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a25_623. ISBN 978-3527306732.
- ^ Oka, Kitaro (1981). "Some Applications of Thionyl Chloride in Synthetic Organic Chemistry". Synthesis. 1981 (9): 661–681. doi:10.1055/s-1981-29563. S2CID 94917739.
- ^ Calderazzo, Fausto; Dell'Amico, Daniela Belli (April 1981). "Syntheses of carbonyl halides of late transition elements in thionyl chloride as solvent. Carbonyl complexes of palladium(II)". Inorganic Chemistry. 20 (4): 1310–1312. doi:10.1021/ic50218a072.
- ^ Garber, E. B.; Pease, L. E. D.; Luder, W. F. (20 April 1953). "Titration of Aprotic Acids in Thionyl Chloride". Analytical Chemistry. 25 (4): 581–583. doi:10.1021/ac60076a012.
- ^ a b c Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 694. ISBN 978-0-08-037941-8.
- ^ Brauer, George (1963). "Handbook of Preparative Inorganic Chemistry" (PDF). Sciencemadness Library. pp. 382–383.
- ^ Mootz, D.; Merschenz-Quack, A. (15 May 1988). "Structures of thionyl halides: SOCl2 and SOBr2" (PDF). Acta Crystallographica Section C. 44 (5): 926–927. Bibcode:1988AcCrC..44..926M. doi:10.1107/S010827018800085X.
- ^ Brauer, Georg, ed. (1963). Handbook of Preparative Inorganic Chemistry. Vol. 1. Translated by Scripta Technica; Reed, F. (2nd ed.). New York, NY: Academic Press. p. 383. ISBN 978-0121266011.
- ^ Donovan, R. J.; Husain, D.; Jackson, P. T. (1969). "Spectroscopic and kinetic studies of the SO radical and the photolysis of thionyl chloride". Transactions of the Faraday Society. 65: 2930. doi:10.1039/TF9696502930.
- ^ Friedman, L.; Wetter, W. P. (1967). "Purification of thionyl chloride". Journal of the Chemical Society A: Inorganic, Physical, Theoretical: 36. doi:10.1039/J19670000036.
- ^ Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, p. 469, ISBN 978-0-471-72091-1
- ^ Van Woerden, H. F. (1963). "Organic Sulfites". Chemical Reviews. 63 (6): 557–571. doi:10.1021/cr60226a001.
- ^ Brenner, M.; Huber, W. (1953). "Herstellung von α-Aminosäureestern durch Alkoholyse der Methylester" [Manufacture of α-amino acid esters by alcoholysis of methyl esters]. Helvetica Chimica Acta (in German). 36 (5): 1109–1115. doi:10.1002/hlca.19530360522.
- ^ Clayden, Jonathan; Greeves, Nick; Warren, Stuart; Wothers, Peter (2001). Organic Chemistry (1st ed.). Oxford University Press. p. 295. ISBN 978-0-19-850346-0.
- ^ Allen, C. F. H.; Byers, J. R. Jr; Humphlett, W. J. (1963). "Oleoyl chloride". Organic Syntheses; Collected Volumes, vol. 4, p. 739.
- ^ Rutenberg, M. W.; Horning, E. C. (1963). "1-Methyl-3-ethyloxindole". Organic Syntheses; Collected Volumes, vol. 4, p. 620.
- ^ Clayden, Jonathan; Greeves, Nick; Warren, Stuart; Wothers, Peter (2001). Organic Chemistry (1st ed.). Oxford University Press. ISBN 978-0-19-850346-0.
- ^ Niznik, G. E.; Morrison, W. H., III; Walborsky, H. M. (1988). "1-d-Aldehydes from organometallic reagents: 2-methylbutanal-1-d". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 6, p. 751. - ^ Arrieta, A.; Aizpurua, J. M.; Palomo, C. (1984). "N,N-Dimethylchlorosulfitemethaniminium chloride (SOCl2-DMF) a versatile dehydrating reagent". Tetrahedron Letters. 25 (31): 3365–3368. doi:10.1016/S0040-4039(01)81386-1.
- ^ Clayden, J. (2001). Organic Chemistry. Oxford: Oxford University Press. pp. 276–296. ISBN 0-19-850346-6.
- ^ Krynitsky, J. A.; Carhart, H. W. (1963). "2-Ethylhexanonitrile". Organic Syntheses; Collected Volumes, vol. 4, p. 436.
- ^ Hulce, M.; Mallomo, J. P.; Frye, L. L.; Kogan, T. P.; Posner, G. H. (1990). "(S)-(+)-2-(p-toluenesulfinyl)-2-cyclopentenone: Precursor for enantioselective synthesis of 3-substituted cyclopentanones". Organic Syntheses; Collected Volumes, vol. 7, p. 495.
- ^ Kurzer, F. (1963). "p-Toluenesulfinyl chloride". Organic Syntheses; Collected Volumes, vol. 4, p. 937.
- ^ Weinreb, S. M.; Chase, C. E.; Wipf, P.; Venkatraman, S. (2004). "2-Trimethylsilylethanesulfonyl chloride (SES-Cl)". Organic Syntheses; Collected Volumes, vol. 10, p. 707.
- ^ Hazen, G. G.; Bollinger, F. W.; Roberts, F. E.; Russ, W. K.; Seman, J. J.; Staskiewicz, S. (1998). "4-Dodecylbenzenesulfonyl azides". Organic Syntheses; Collected Volumes, vol. 9, p. 400.
- ^ Hogan, P. J.; Cox, B. G. (2009). "Aqueous Process Chemistry: The Preparation of Aryl Sulfonyl Chlorides". Organic Process Research & Development. 13 (5): 875–879. doi:10.1021/op9000862.
- ^ Pray, A. R.; Heitmiller, R. F.; Strycker, S.; Aftandilian, V. D.; Muniyappan, T.; Choudhury, D.; Tamres, M. (1990). "Anhydrous Metal Chlorides". Inorganic Syntheses. Vol. 28. pp. 321–323. doi:10.1002/9780470132593.ch80. ISBN 978-0-470-13259-3.
- ^ Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, p. 697, ISBN 978-0-471-72091-1
- ^ Peyronneau, M.; Roques, N.; Mazières, S.; Le Roux, C. (2003). "Catalytic Lewis Acid Activation of Thionyl Chloride: Application to the Synthesis of Aryl Sulfinyl Chlorides Catalyzed by Bismuth(III) Salts". Synlett (5): 0631–0634. doi:10.1055/s-2003-38358.
- ^ Bandgar, B. P.; Makone, S. S. (2004). "Lithium/Sodium Perchlorate Catalyzed Synthesis of Symmetrical Diaryl Sulfoxides". Synthetic Communications. 34 (4): 743–750. doi:10.1081/SCC-120027723. S2CID 96348273.
- ^ Rao, M. R. Aswathanarayana (March 1940). "Thionyl iodide: Part I. Formation of thionyl iodide". Proceedings of the Indian Academy of Sciences - Section A. 11 (3): 185–200. doi:10.1007/BF03046547. S2CID 104552644.
- ^ Rao, M. R. Aswathanarayana (March 1940). "Thionyl iodide: Part II. Rate of decomposition and spectroscopic". Proceedings of the Indian Academy of Sciences - Section A. 11 (3): 201–205. doi:10.1007/BF03046548. S2CID 104752226.
- ^ Gangadharan, R.; Namboodiri, P.N.N.; Prasad, K.V.; Viswanathan, R. (January 1979). "The lithium—thionyl chloride battery — a review". Journal of Power Sources. 4 (1): 1–9. Bibcode:1979JPS.....4....1G. doi:10.1016/0378-7753(79)80032-4.
- ^ Institute of Medicine (1993). Chapter 5: Chemistry of Sulfur Mustard and Lewisite. The National Academies Press. ISBN 0-309-04832-X.
{{cite book}}:|work=ignored (help) - ^ See:
- Persoz; Bloch (1849). "Des composés binaires formés par les métalloïdes, et, en particulier, de l'action du chloride phosphorique sur les acides sulfureux, sulfurique, phosphorique, chromique, etc" [Binary compounds formed by metalloids and in particular the action of phosphorus pentachloride on sulfurous acid, sulfuric acid, phosphoric acid, chromic acid, etc.]. Comptes rendus (in French). 28: 86–88.
- Persoz; Bloch (1849). "Addition à une Note précédente concernant les combinaisons du chloride phosphorique avec les acides phosphorique, sulfurique et sulfureux" [Addition to a preceding note concerning compounds of phosphorus pentachloride with phosphoric acid, sulfuric acid, and sulfurous acid]. Comptes rendus (in French). 28: 389.
- ^ Kremers, P. (1849). "Ueber schwefligsaures Phosphorchlorid" [On sulfurous phosphorus pentachloride]. Annalen der Chemie und Pharmacie (in German). 70 (3): 297–300. doi:10.1002/jlac.18490700311.
- ^ The German chemist Georg Ludwig Carius noted that, when the reaction mixture that produced thionyl chloride was distilled, the crude mixture initially released substantial quantities of gas, so that phosphoryl chloride (POCl3) was carried into the receiver. Carius, L. (1859). "Ueber die Chloride des Schwefels und deren Derivate" [On sulfur chloride and its derivatives]. Annalen der Chemie und Pharmacie (in German). 111: 93–113. doi:10.1002/jlac.18591110111. From p. 94: " … dabei ist jedoch die Vorsicht zu gebrauchen, … und nie reines Chlorthionyl erhalten wird." ( … however, during that [i.e., the fractional distillation], caution must be used, [so] that one carefully avoids a concentration of hydrogen chloride or excess sulfurous acid in the liquid that is to be distilled, as otherwise, by the evolution of gas that occurs at the start of the distillation, much phosphoryl chloride is transferred and pure thionyl chloride is never obtained.)
- ^ Schiff, Hugo (1857). "Ueber die Einwirkung des Phosphorsuperchlorids auf einige anorganische Säuren" [On the reaction of phosphorus pentachloride with some inorganic acids]. Annalen der Chemie und Pharmacie (in German). 102: 111–118. doi:10.1002/jlac.18571020116. The boiling point of thionyl chloride which Schiff observed, appears on p. 112. The name Thionylchlorid is coined on p. 113.
- ^ Carius, L. (1859). "Ueber die Chloride des Schwefels und deren Derivate" [On sulfur chloride and its derivatives]. Annalen der Chemie und Pharmacie (in German). 111: 93–113. doi:10.1002/jlac.18591110111. On p. 94, Carius notes that thionyl chloride can be " … mit Vortheil zur Darstellung wasserfreier Säuren verwenden." ( … used advantageously for the preparation of acid anhydrides.) Also on p. 94, Carius shows chemical equations in which thionyl chloride is used to transform benzoic acid (OC7H5OH) into benzoyl chloride (ClC7H5O) and to transform sodium benzoate into benzoic anhydride. On p. 96, he mentions that thionyl chloride will transform methanol into methyl chloride (Chlormethyl). Thionyl chloride behaves like phosphoryl chloride: from pp. 94-95: "Die Einwirkung des Chlorthionyls … die Reaction des Chlorthionyls weit heftiger statt." (The reaction of thionyl chloride with [organic] substances containing oxygen proceeds in general parallel to that of phosphoryl chloride; where the latter exerts an effect, thionyl chloride usually does so also, only in nearly all cases the reaction occurs far more vigorously.)














